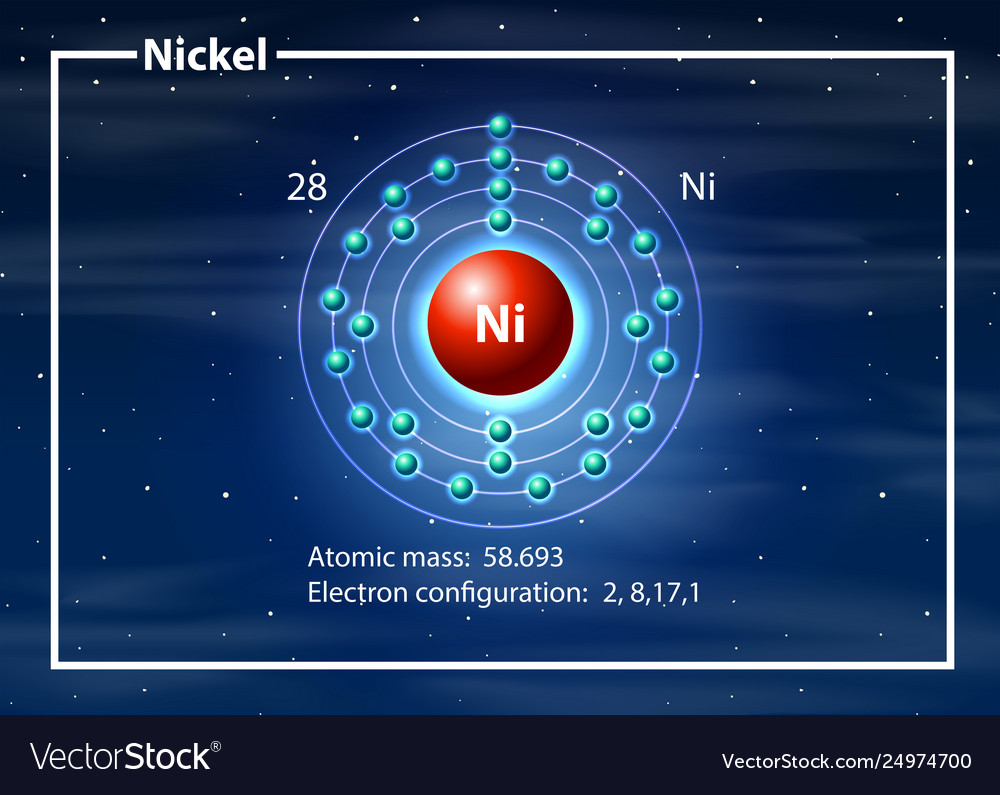
37 nickel electron dot diagram
Nickel(II) hydroxide. NICKEL HYDROXIDE. 11113-74-9. CCRIS 429. EINECS 234-348-1 The nickel obtained from another ore, nickeliferous limonite, is contaminated with iron. ... Deduce the Lewis (electron dot) structure of the carbonate ion, giving the shape and the oxygen-carbon-oxygen bond angle. - trigonal planar - 120.
Answer (1 of 2): Instead of acting like an entitled egomaniac who thinks he's the smartest guy in the room like the other guy did, I'll provide an actual answer. Since nickel is a transition element, you have to manually write out its electron configuration and figure out how many electrons the l...

Nickel electron dot diagram
2. Electron Configuration 02 (gives the most information) Is 2s (quicker to draw than orbital filling diagrams) 1 s2 2s2 2p4 3. Electron Dot shows only the valence (outer energy level) electrons EX. Oxygen atom 1 . Write orbital filling diagrams, electron configurations, and electron dot diagrams for the following elements. LKrl 10 Table. a ... Electron Configurations are an organized means of documenting the placement of electrons based upon the energy levels and orbitals groupings of the periodic table.. The electron configuration for the first 10 elements. H #1s^1# He #1s^2# Li #1s^2 2s^1# Be #1s^2 2s^2# B #1s^2 2s^2 2p^1# C #1s^2 2s^2 2p^2# N #1s^2 2s^2 2p^3# O #1s^2 2s^2 2p^4# F #1s^2 2s^2 2p^5# Structure and bonding. In nickel tetracarbonyl, the oxidation state for nickel is assigned as zero. The formula conforms to 18-electron rule.The molecule is tetrahedral, with four carbonyl (carbon monoxide) ligands. Electron diffraction studies have been performed on this molecule, and the Ni-C and C-O distances have been calculated to be 1.838(2) and 1.141(2) angstroms respectively.
Nickel electron dot diagram. Feb 21, 2017 — Tellurium has got electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p4, that means it has got 6 valence electrons (in the last - 5th - shell, ...2 answers · Top answer: Instead of acting like an entitled egomaniac who thinks he’s the smartest guy in the room ...Does it matter where you put the dots on a Lewis ...1 answerMay 5, 2020What is the Lewis dot structure for br2? - electrons ...2 answersSep 19, 2016More results from www.quora.com C) Electron X absorbs energy when it changes to a lower energy level. D) Electron X neither emits nor absorbs energy when it changes energy level. 30.Electron X can change to a higher energy level or a lower energy level. Which statement is true of electron X? A) decreases B) increases C) remains the same 31.As an electron in a hydrogen atom ... In the case of Nickel the abbreviated electron configuration is [Ar] 3d8 4s2. Nevertheless, check the complete configuration and other interesting facts about Nickel that most people don't know. Nickel Overview Nickel Complete Electron Configuration 1s2 2s2 2p6 3s2 3p6 4 s2 3 d8 Abbreviated Electron Configuration [Ar] 3d8 4s2 Sources The effects of nickel on hepatic glutathione and the enzymes glutathione reductase, glutathione-peroxidase, glutathione-S-transferase, and gamma-glutamyl-transpeptidase were investigated in female albino rats.Three groups of rats were sacrificed 16, 24, or 72 hr after a subcutaneous injection of nickel as nickel chloride at a level of 200 micromol/kg. . Four groups of rats were sacrificed 16 ...
The Lewis Structure, or Lewis Dot Diagram, shows the bonding between atoms of a molecule and any electrons that may exist. The Lewis Structure for Li is Li with one dot to the right of the element. Lewis Structure Examples. The Lewis electron dot structures of a few molecules are illustrated in this subsection. 1. Lewis Structure of CO2. The central atom of this molecule is carbon. Oxygen contains 6 valence electrons which form 2 lone pairs. Since it is bonded to only one carbon atom, it must form a double bond. This Dot diagram basically represents the numbers of valence electrons for the atoms. It uses the representative symbol of Nickel (Ni) to draw the valence electrons around it. So, the numbers of dots show are exactly the numbers of valence electrons. You can draw the Lewis dot diagram for any chemical element. This photo about: Electron Dot Diagram for Ammonia, entitled as Chem Supplemental Instruction Dean Students Fice Nickel Lewis Dot Electron Dot Diagram For Ammonia - also describes Chem Supplemental Instruction Dean Students fice Nickel Lewis Dot and labeled as: ], with resolution 2523px x 3266px
Jan 26, 2021 — Valence electrons are the electrons which are located in the outer shell or orbit. There are 28 electrons in the nickel in the 4 orbits and the ... 10. Write the short hand electron configuration for element 19, element 53, element 88. Which families do they belong to and give electron dot structure. 11. Specify properties of m, nm, sm. Given an unknown element with some general properties can you identify it as a metal, non-metal, or semimetal? 12. List the diatomic elements. 13. a. A step-by-step explanation of how to draw the Ni(NO3)2 Lewis Dot Structure.For Ni(NO3)2 we have an ionic compound and we need to take that into account when ... Electron dot diagrams are diagrams in which the valence electrons of an atom are shown as dots distributed around the element's symbol. A beryllium atom, with two valence electrons, would have the electron dot diagram below. Since electrons repel each other, the dots for a given atom are distributed evenly around the symbol before they are ...
There are two types of diagrams one is the Lewis diagram the other is the Electron dot diagram. To make the electron dot diagram you put the electron symbol and put a dot on one of the sides for ...
What is the abbreviated electron configuration for nickel ( atomic number 28)?: [Ar]4s2 3d8. ... In an electron-dot structure of an element, the dots are used to represent: The valence electrons. How many valence electrons are in the electron-dot structures for the elements in group 3A?: 3.
Nitrogen triiodide (NI3) lewis dot structure, molecular geometry, hybridization, polarity. Nitrogen triiodide appears as purple gas is an inorganic compound having the chemical formula NI3. It releases a purple cloud of iodine vapor when touched gently because of its extremely sensitive contact explosion property.
Iron Electron Configuration Png Download Nickel Electron Configuration Diagram Transparent Png 1880x1405 3903560 Pinpng
To write the configuration for the Nickel ions, first we need to write the electron configuration for just Nickel (Ni). We first need to find the number of ...
A Lewis electron dot diagram (or electron dot diagram or a Lewis diagram or a Lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. The number of dots equals the number of valence electrons in the atom. These dots are arranged to the right and left and above and below the ...
Answer (1 of 2): To draw lewis dot structures, you first must know the number of valence electrons the atom has. By writing Fe in a noble gas configuration, you get [Ar] 3d6 4s2. By looking at this, since iron is a transition metal, you can see that iron technically has 8 valence electrons, but w...
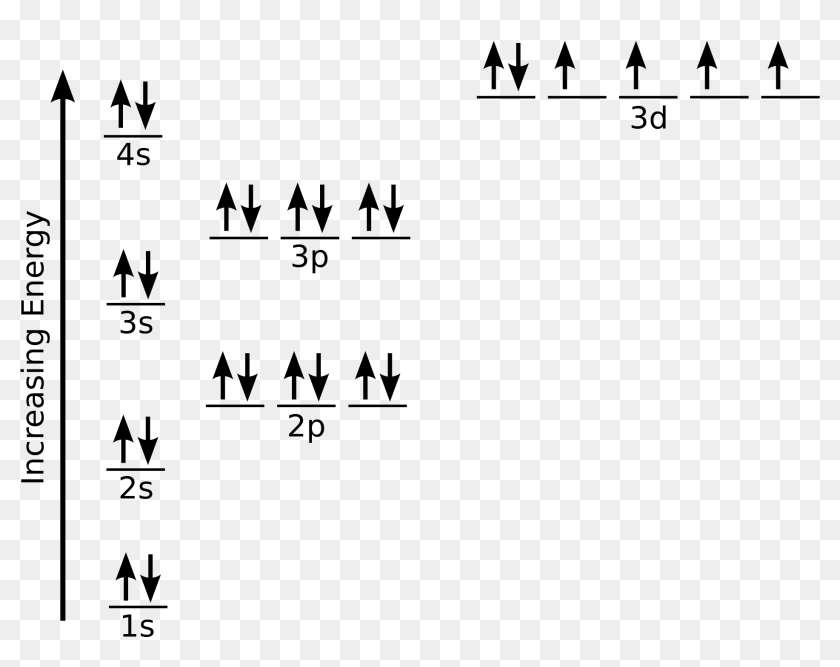
To help determine the electron configuration of nickel (Ni), we will visualize how the electrons are distributed in an Aufbau diagram below. Nickel has an atomic number of 28, which means a ...

1 Write Orbital Diagrams For Each Of These Ions A V5 B Cr3 C Ni2 D Fe3 2 Determine If The Ion Is Diamagnetic Or Paramagnetic A V5 B Cr3 C Ni2
(or electron dot diagram or a Lewis diagram or a Lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol ...
What is nickel's electron configuration? Chemistry Electron Configuration Electron Configuration. 1 Answer Sarita Rana Feb 9, 2017 Ni electronic configuration: [Ar], 3d8, 4s2. Explanation: Atomic Number of Nickel (Ni) = 28 Electronic configuration is 1s2, 2s2, 2p6, 3s2, 3p6, 4s2, 3d8 or [Ar], 3d8, 4s2 ...
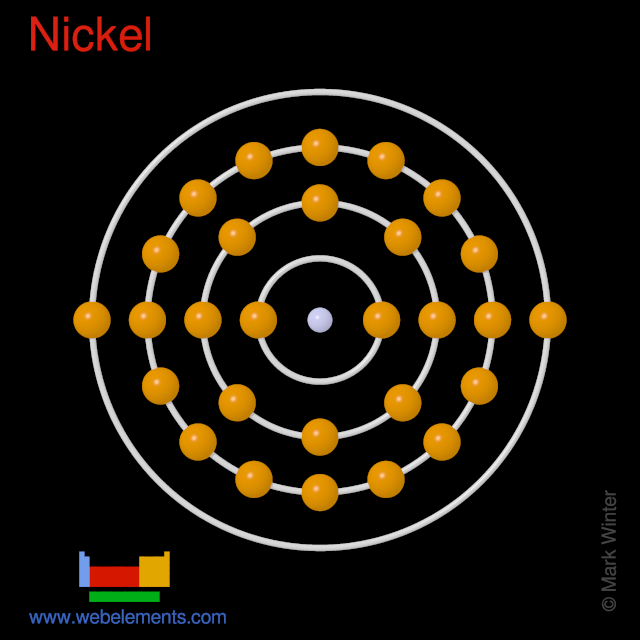
Element Nickel - Ni Comprehensive data on the chemical element Nickel is provided on this page; including scores of properties, element names in many languages, most known nuclides of Nickel. Common chemical compounds are also provided for many elements.
Draw the complete Lewis electron-dot diagram for ethyne in the appropriate cell in the table above. Which of the four molecules contains the shortest carbon-to-carbon bond? Explain. A Lewis electron-dot diagram of a molecule of ethanoic acid is given below. The carbon atoms in the molecule are labeled x and y, respectively.
The nickel atom has two electron configurations, [Ar] 3d 8 4s 2 and [Ar] 3d 9 4s 1, which are very close in energy - the symbol [Ar] refers to the argon-like core structure. There is some disagreement on which configuration has the lowest energy. Chemistry textbooks quote the electron configuration of nickel as [Ar] 4s 2 3d 8, which can also ...
MO Diagram for HF The AO energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. The F 2s is nonbonding. H-F nb σ σ* Energy H -13.6 eV 1s F -18.6 eV -40.2 eV 2s 2p So H-F has one σ bond and three lone electron pairs on fluorine
Lewis-Dot Diagrams •Lewis Dot Diagrams are a way to represent the valence electrons in an atom. -Element's symbol represents the nucleus and inner-level electrons -Dots represent the valence electrons 5.3 Electron Configuration. Lewis-Dot Diagrams •Dots are placed one at a time on the four
Structure and bonding. In nickel tetracarbonyl, the oxidation state for nickel is assigned as zero. The formula conforms to 18-electron rule.The molecule is tetrahedral, with four carbonyl (carbon monoxide) ligands. Electron diffraction studies have been performed on this molecule, and the Ni-C and C-O distances have been calculated to be 1.838(2) and 1.141(2) angstroms respectively.
Electron Configurations are an organized means of documenting the placement of electrons based upon the energy levels and orbitals groupings of the periodic table.. The electron configuration for the first 10 elements. H #1s^1# He #1s^2# Li #1s^2 2s^1# Be #1s^2 2s^2# B #1s^2 2s^2 2p^1# C #1s^2 2s^2 2p^2# N #1s^2 2s^2 2p^3# O #1s^2 2s^2 2p^4# F #1s^2 2s^2 2p^5#
2. Electron Configuration 02 (gives the most information) Is 2s (quicker to draw than orbital filling diagrams) 1 s2 2s2 2p4 3. Electron Dot shows only the valence (outer energy level) electrons EX. Oxygen atom 1 . Write orbital filling diagrams, electron configurations, and electron dot diagrams for the following elements. LKrl 10 Table. a ...
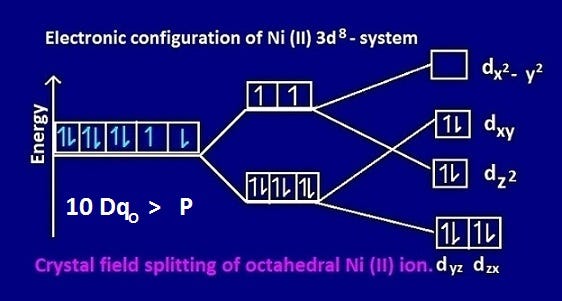
Why Is Ni Nh3 6 Cl2 Paramagnetic But Co Nh3 6 Cl3 Is Diamagnetic By Kakali Ghosh Teacher Blogger M Sc Chemistry Medium




















0 Response to "37 nickel electron dot diagram"
Post a Comment