39 energy diagram for hydrogen
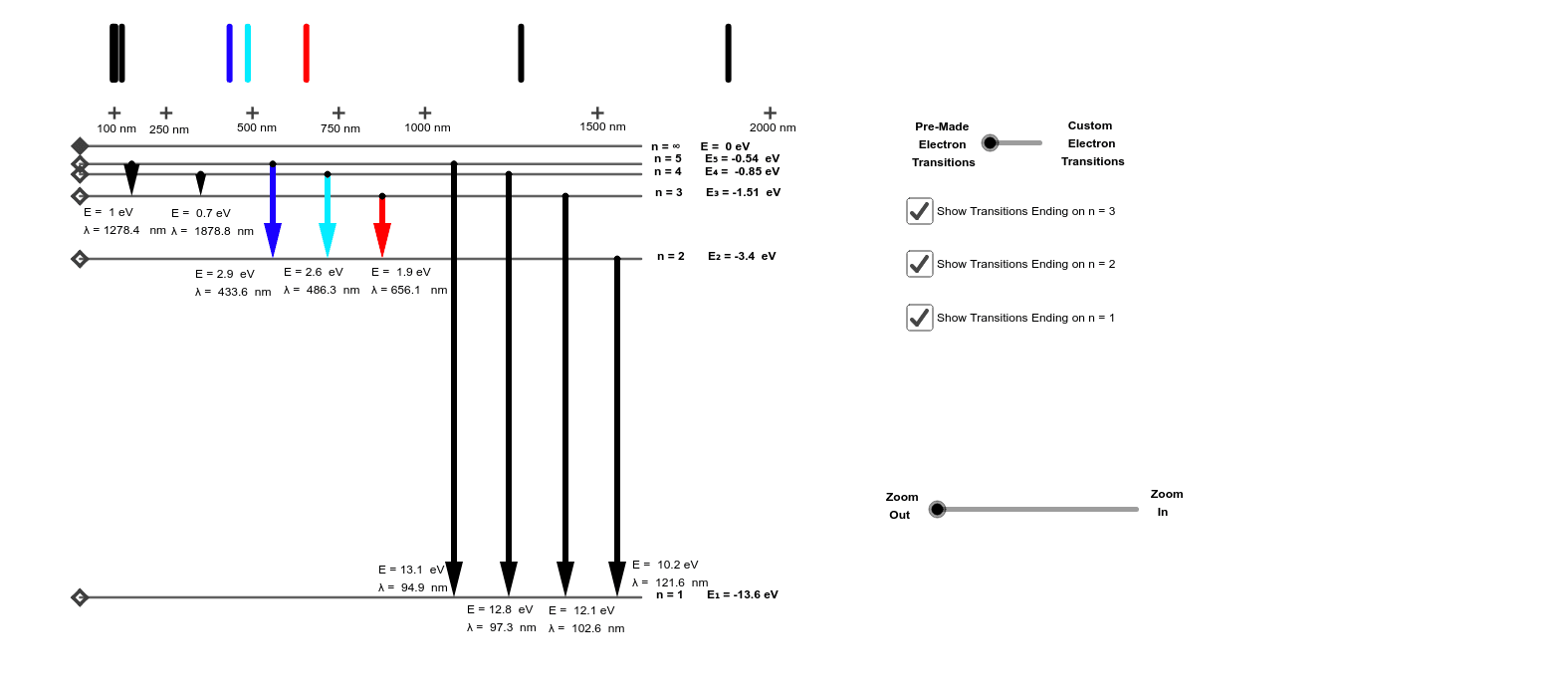
1 answerThe hydrogen atom is the simplest in the universe that consists of only a proton and an electron. The electron is at the ground state, which is the nearest ... The ionization energy of an atom is the energy required to remove the electron completely from the atom. (transition from ground state n = 0 to infinity n = ∞ ). For hydrogen, the ionization energy = 13.6eV. When an excited electron returns to a lower level, it loses an exact amount of energy by emitting a photon.
Q. The energy level diagram for the reaction between sodium hydrogen carbonate and dilute hydrochloric acid is shown. Which correctly describes the type of reaction and the energy of the reactants and products?

Energy diagram for hydrogen
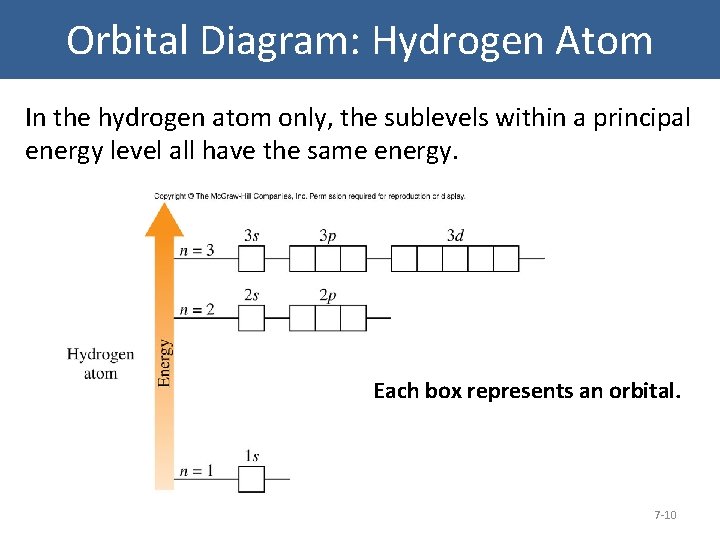
Energy level diagrams and the hydrogen atom. It's often helpful to draw a diagram showing the energy levels for the particular element you're interested in. The diagram for hydrogen is shown above. The n = 1 state is known as the ground state, while higher n states are known as excited states. If the electron in the atom makes a transition from ... Another question that though the internuclear distance at a particular point is constant yet potential energy keeps on increasing. I think the point where potential energy become zero we can say that attractive forces between electron and nucleus of bonding hydrogen atoms just equals the repulsive force between bonding hydrogen electrons. How Bohr's model of hydrogen explains atomic emission spectra. ... be illustrated using an energy level diagram, such as the example above showing electrons ...
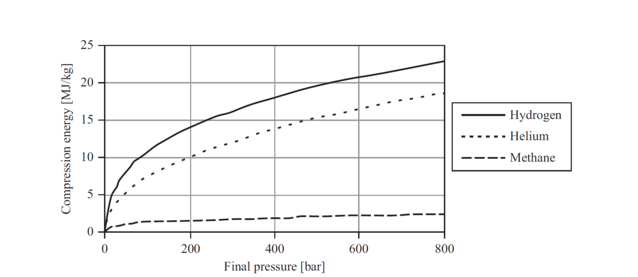
Energy diagram for hydrogen. The model solves for a total cost of energy in dollars per kWh and does not distinguish between the types of energy (electricity, heat, and hydrogen). The user sets the costs for two of the energy types; these values are subtracted from the total energy cost to calculate the cost of the third energy type. Il n'y a pas de texte répondant à ce style dans ce document.-6: Flammability Diagram for Hydrogen/Air/Nitrogen at 400°C and 1 bar For hydrogen, the minimum ignition energy is low at 0.017 mJ for mixtures with air (ISO, 2004), and even lower at 0.0012 mJ for mixtures with oxygen (Kuchta, 1986). Hydrogen has a such a low A Hydrogen atom has only one single electron revolving around the nucleus at the lowest energy level. When we heat a hydrogen atom, the electron gains energy ...1 answer · Top answer: Hint: Energy level diagram is the direct consequence of the principal quantum number - ‘n’. Energy diagram is necessary to determine the energy difference ... Problem: Consider the following portion of the energy-level diagram for hydrogen:n = 4 –0.1361 × 10 –18 Jn = 3 –0.2420 × 10 –18 Jn = 2 –0.5445 × 10 –18 Jn = 1 –2.178 × 10 –18 JFor which of the following transitions does the light emitted have the longest wavelength?A. n = 4 to n = 1B. n = 4 to n = 2C. n = 4 to n = 3D. n = 3 to n = 2E. n = 2 to n = 1
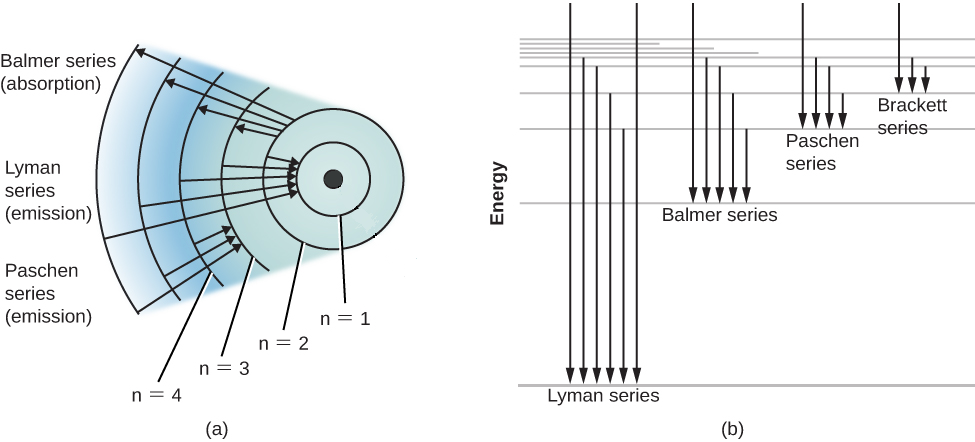
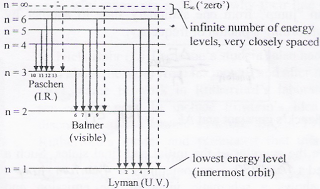
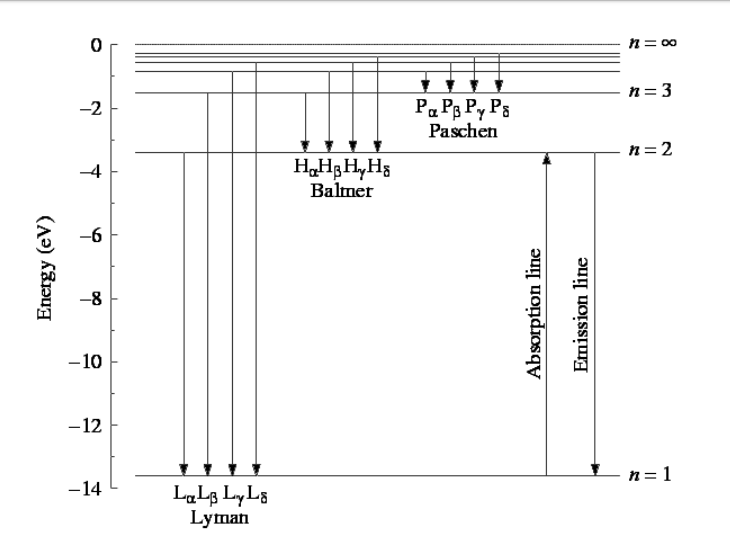
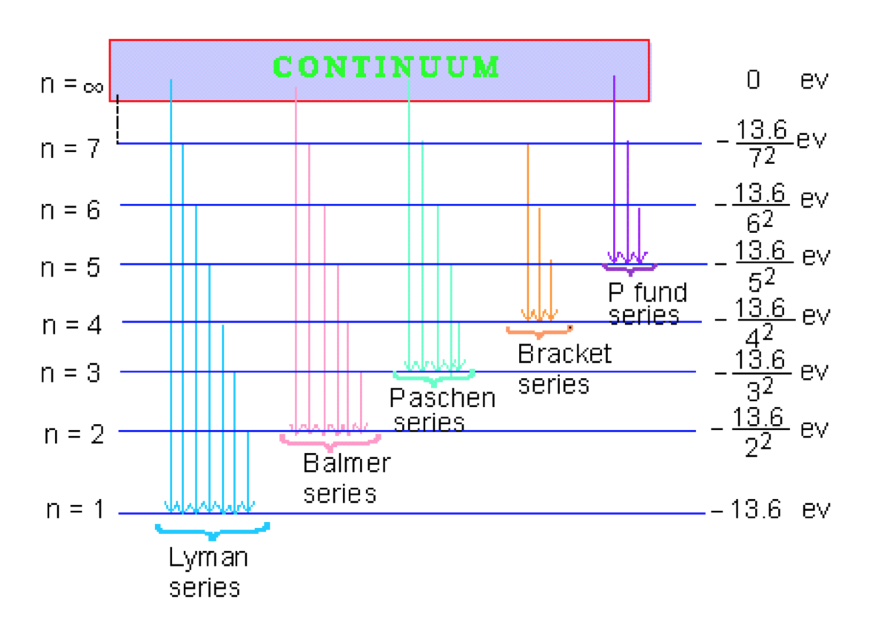
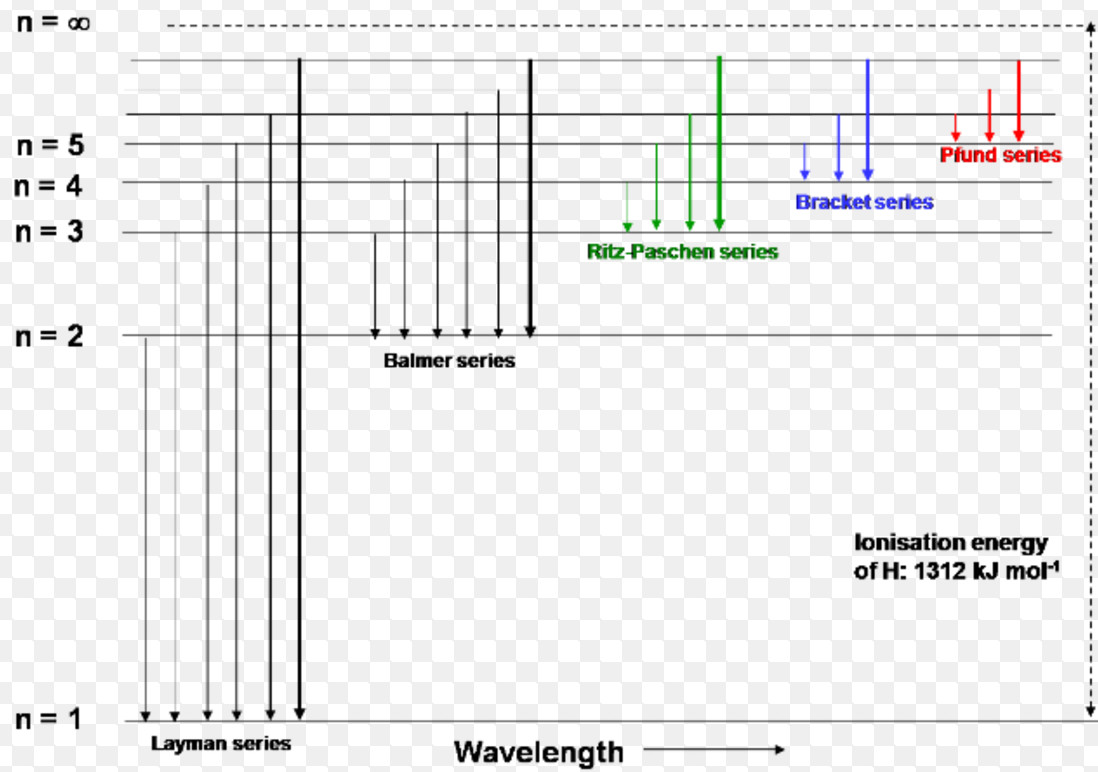
Click here to get an answer to your question ✍️ Draw a neat labelled energy level diagram of the Hydrogen atom.19 Nov 20191 answer · Top answer: Given figure shows energy level diagram for Hydrogen atom. 29 Mar 2021 — The electron energy level diagram for the hydrogen atom. He found that the four visible spectral lines corresponded to transitions from ... Hydrogen is not toxic but is a simple asphyxiate by the displacement of oxygen in the air. Under prolonged exposure to fire or intense heat the containers may rupture violently and rocket. Hydrogen is used to make other chemicals, in petroleum refining and in oxyhydrogen welding and cutting. The phase diagram of hydrogen is shown below the table. The diagram below shows the energy level diagram of a hydrogen atom. The associated spectrum to the diagram above is shown below. The transition labelled A in the top diagram gives the spectral line labelled B in the spectrum diagram. (a) (i) Show that the frequency of spectral line B is about 4.6 × 10 14 Hz. ΔE = 3.40-1.51 = 1.89 eV.
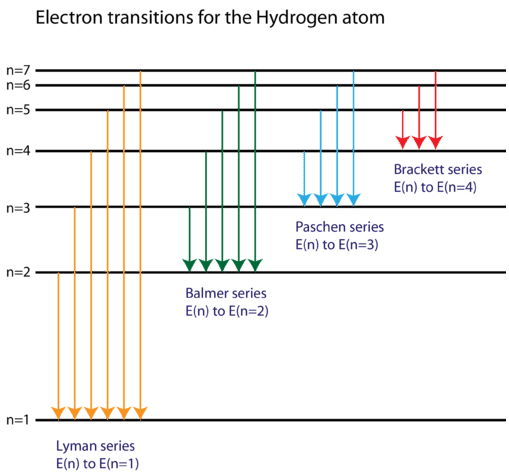
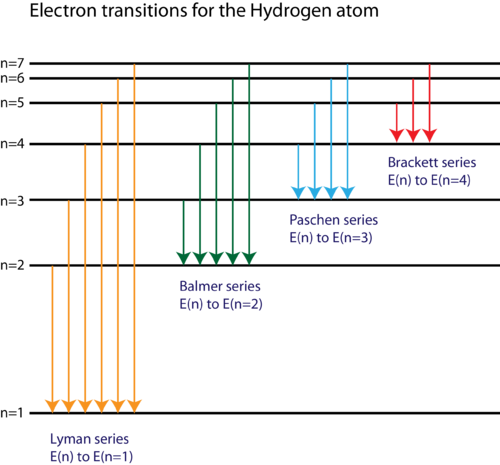
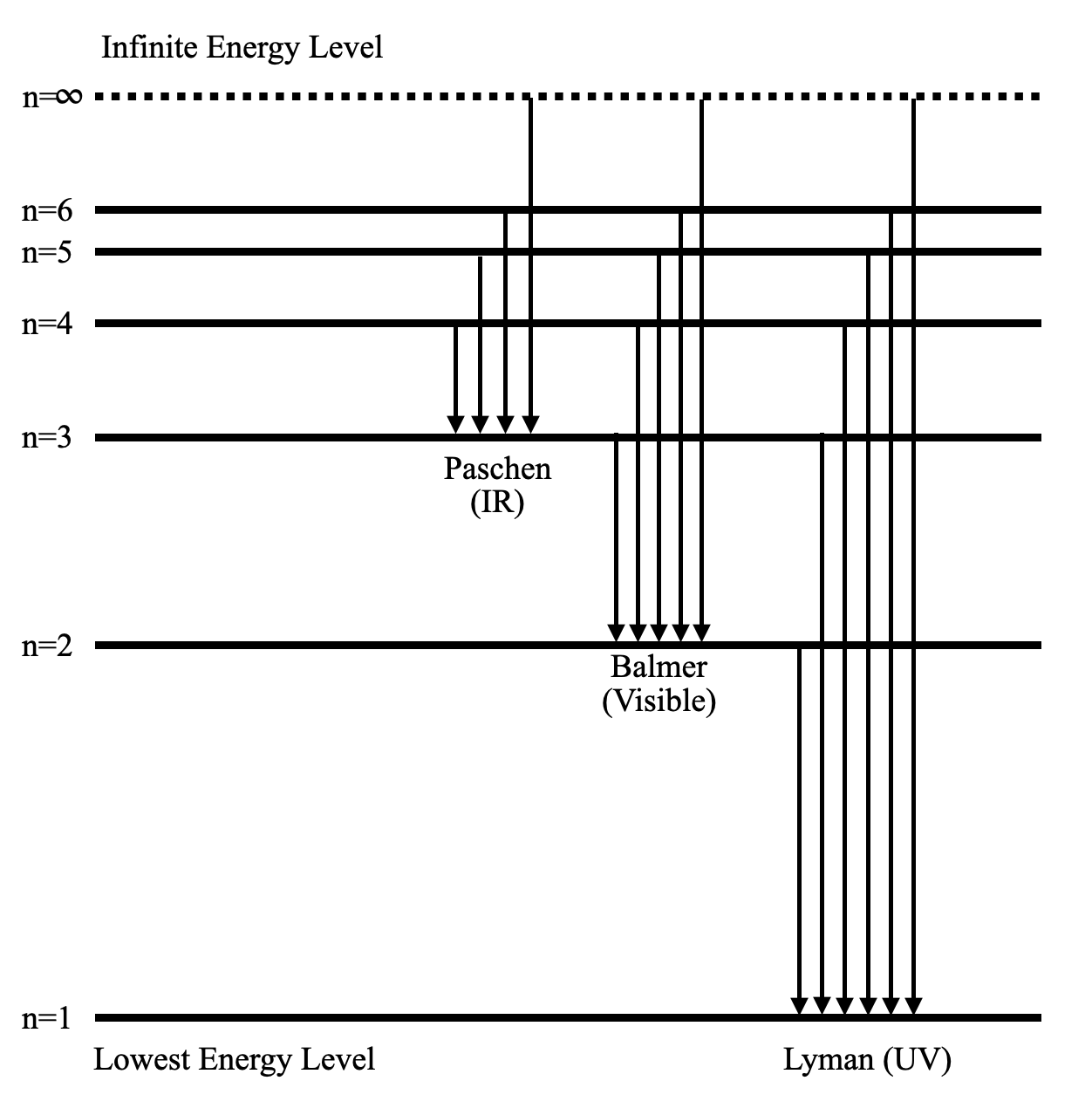
The energy level diagram of the hydrogen atom is given below: The transition of electrons from a higher energy level (n>1) to the ground level (n=1) gives rise to the Lyman series in hydrogen spectra. How Bohr's model of hydrogen explains atomic emission spectra. ... be illustrated using an energy level diagram, such as the example above showing electrons ... Another question that though the internuclear distance at a particular point is constant yet potential energy keeps on increasing. I think the point where potential energy become zero we can say that attractive forces between electron and nucleus of bonding hydrogen atoms just equals the repulsive force between bonding hydrogen electrons. Energy level diagrams and the hydrogen atom. It's often helpful to draw a diagram showing the energy levels for the particular element you're interested in. The diagram for hydrogen is shown above. The n = 1 state is known as the ground state, while higher n states are known as excited states. If the electron in the atom makes a transition from ...

Draw A Neat Labelled Energy Level Diagram For H Atom Showing The Transitions Explain The Series Of Spectral Lines For H Atom Whose Fixed Inner Orbit Numbers Are 3 And 4 Respectively

Hydrogen Emission Spectrum Spectroscopy Successive Ionisation Energy Patterns Related To Sub Shells And Group Of Periodic Table Gce A Level Revision Notes

The Figure Shows An Energy Level Diagram For The Hydrogen Atom Several Transition Are Marked As I Ii Iii The Diagram Is Only Indicative And Not To Scale Img Src Https D10lpgp6xz60nq Cloudfront Net Physics Images
On An Energy Level Diagram Of Hydrogen Show By A Downward Or An Upward Arrow A Transition Which Results In Sarthaks Econnect Largest Online Education Community

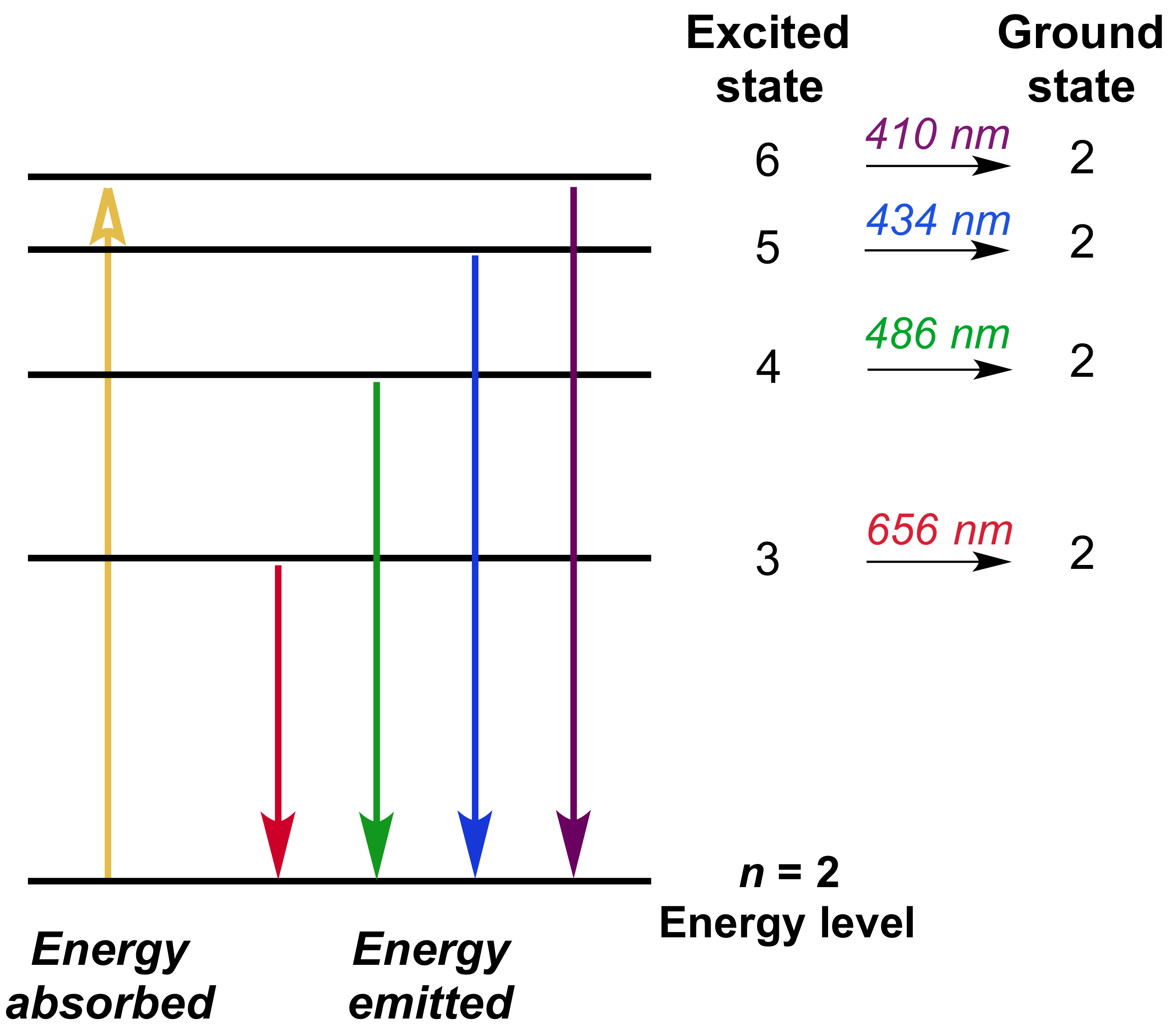
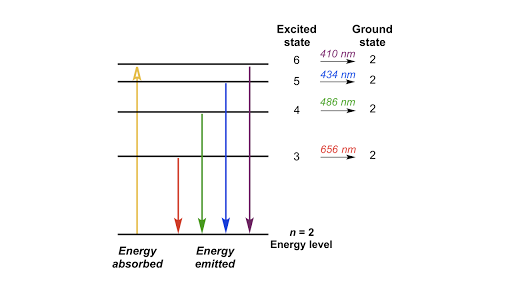
Draw A Partial Energy Level Diagram For Hydrogen All Wavelengths Are Ending At The N 2 State And The Energy Of The N 2 State Is 545 Aj Wavelengths Nm 411 26 434 77 487 10 658 42 Study Com
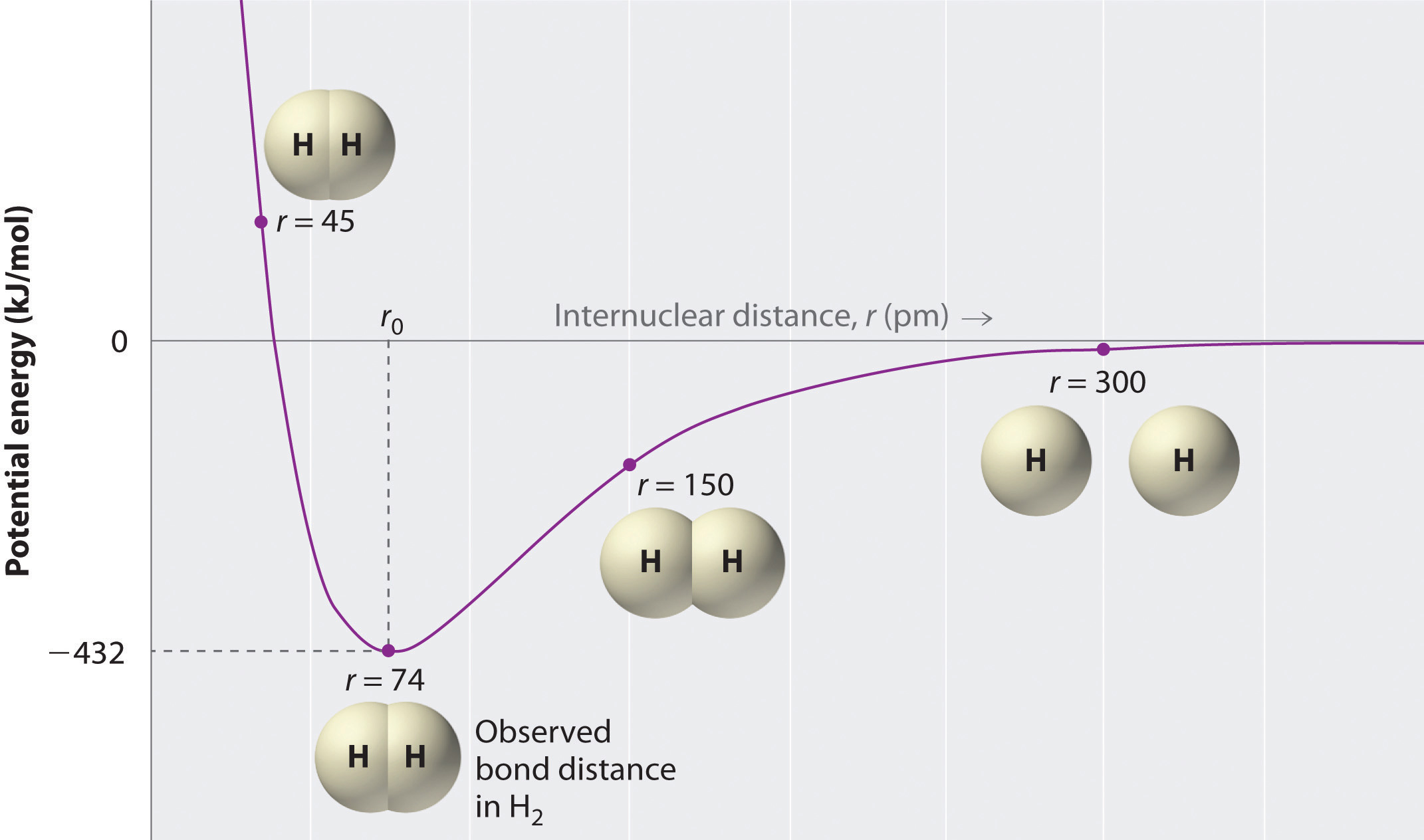
Two Hydrogen Atoms Interact To Form A Hydrogen Molecule Classify The Following Statements That Describe The Stages Of Bond Formation In A Hydrogen Molecule According To The Predominant Force Existing Between The

Hydrogen Based Autonomous Energy Supply System H2one Products And Technical Services Hydrogen Energy Toshiba Energy Systems Solutions Corporation

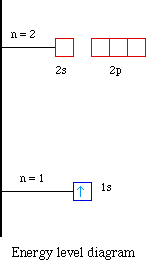
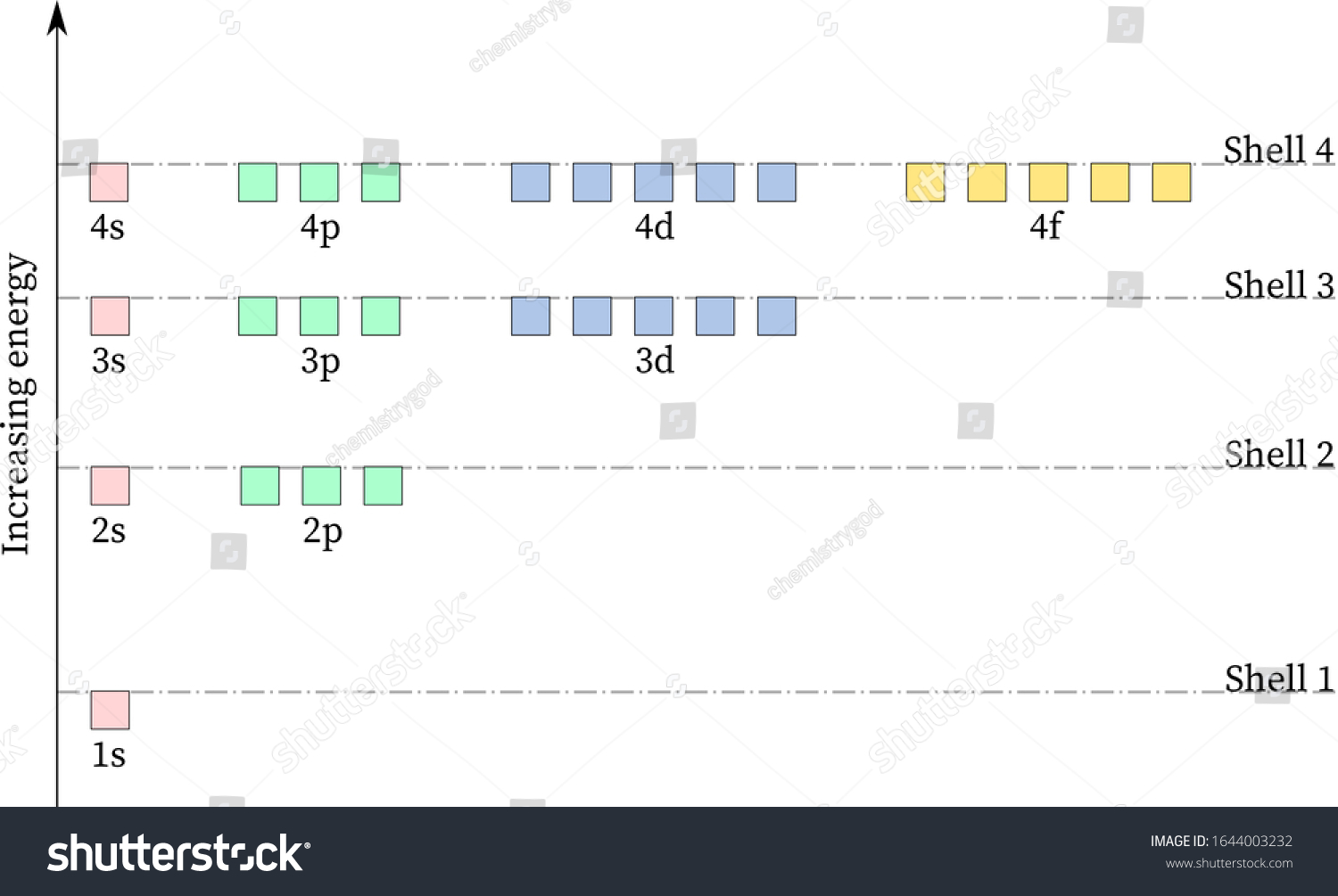
Molecular Orbital Diagram Atomic Orbital Antibonding Molecular Orbital Molecular Orbital Theory Molecular Orbital Angle Text Chemistry Png Pngwing

Hydrogen Emission Spectrum Spectroscopy Successive Ionisation Energy Patterns Related To Sub Shells And Group Of Periodic Table Gce A Level Revision Notes

Bohr S Theory Of The Hydrogen Atom Belhaven University Physical Sciences For A Sustainable Society Openstax Cnx
How Would You Determine The Ionization Energy Of A Hydrogen Atom In Kj Mol If The Electron Is In Its Ground State Socratic










0 Response to "39 energy diagram for hydrogen"
Post a Comment