41 consider the sugar-water phase diagram
Chemistry Q&A Library Consider the sugar - water phase diagram How much sugar will dissolve in 1000 g water at 80°C? If the saturated liquid solution in part (a) is cooled to 20°C, some of the sugar will precipitate out as a solid. 10.1 Consider the sugarwater phase diagram of Figure 10.1. (a) How much sugar will dissolve in 1000 g of water at 80C (176F)?. (b) If the saturated liquid ...
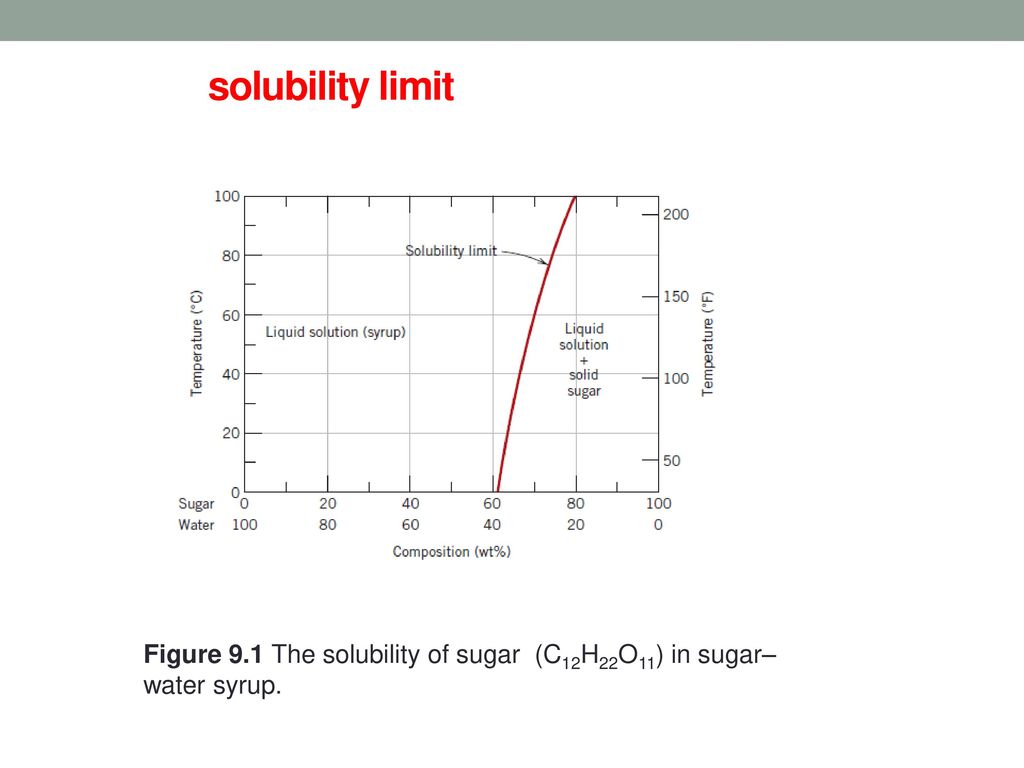
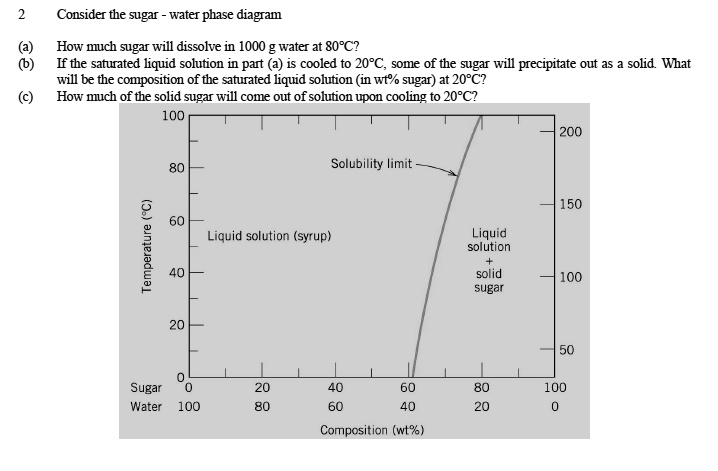
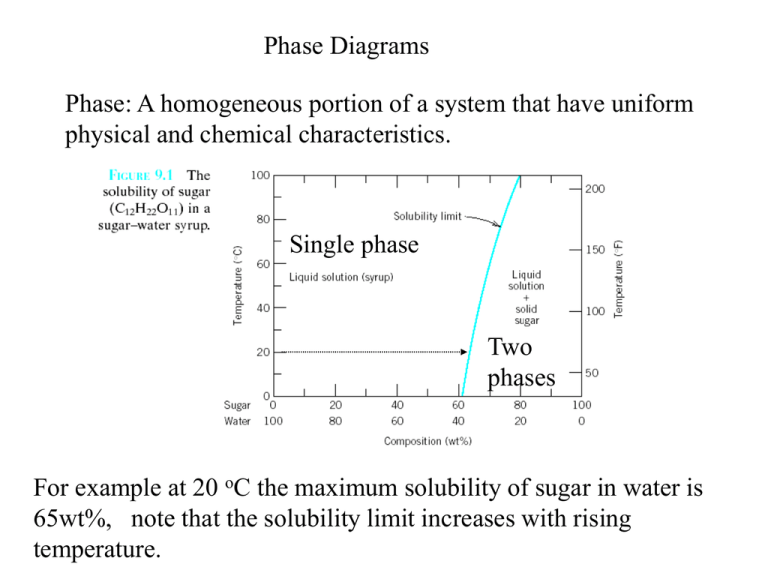
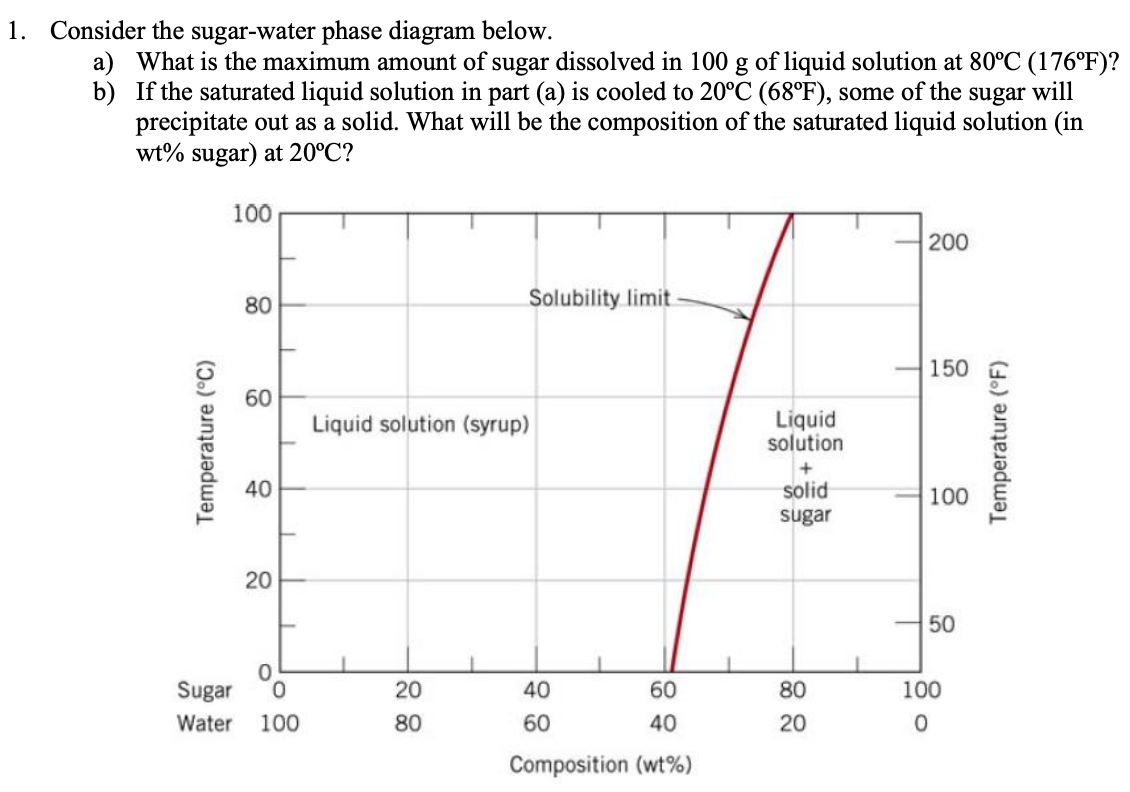
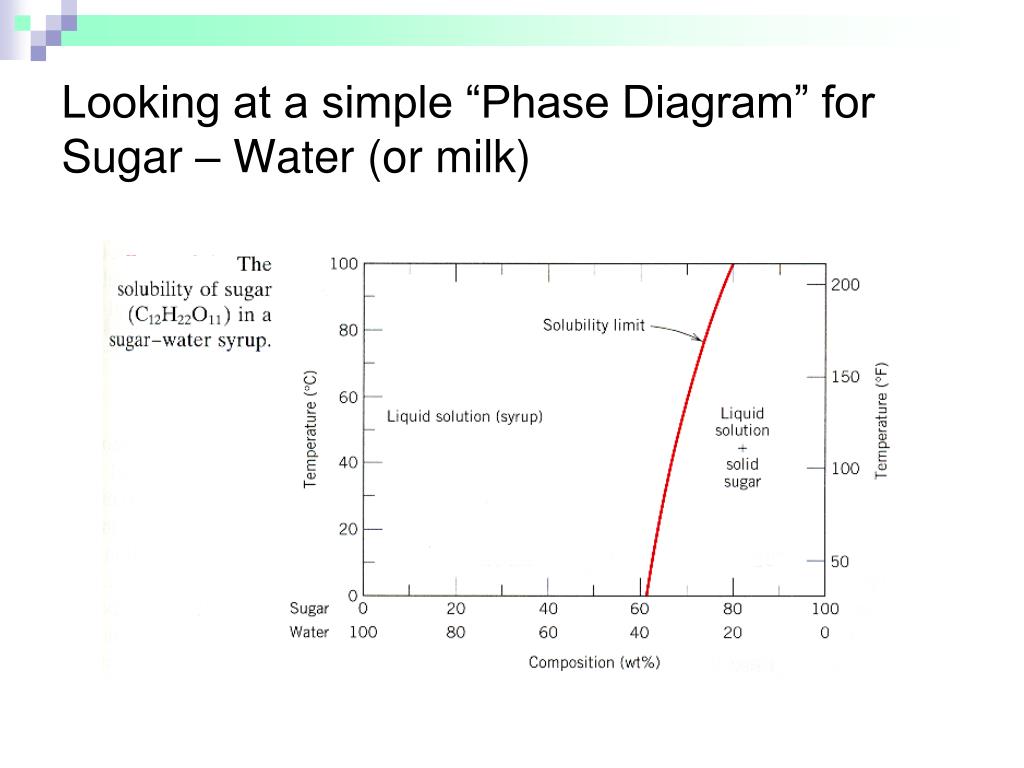
PHASE DIAGRAMS PROBLEM SOLUTIONS Solubility Limit 9.1 Consider the sugar–water phase diagram of Figure 9.1. (a) How much sugar will dissolve in 1500 g water at 90 °C (194 °F)? (b) If the saturated liquid solution in part (a) is cooled to 20 °C (68 °F), some of the sugar will precipitate out as a solid.
Consider the sugar-water phase diagram
Question 9.1 Consider the sugar–water phase diagram of Figure 9.1. (a) How much sugar will dissolve in 1500 g water at 90°C (194°F)?. 9.1 Consider the sugar–water phase diagram of Figure 9.1. (a) How much sugar will dissolve in 1500 g water at 90°C (194°F)? (b) If the saturated liquid solution in part (a) is cooled to 20°C (68°F), some of the sugar will precipitate out as a solid. What will be the composition of the saturated liquid solution (in wt% sugar) at 20°C? 14:440:407 ch9 Question 9.1 Consider the sugar–water phase diagram of Figure 9.1. (a) How much sugar will dissolve in 1500 g water at 90°C (194°F)? ...
Consider the sugar-water phase diagram. 10.1 Consider the sugar–water phase diagram of Figure 10.1. (a) How much sugar will dissolve in 1500 g water at 90 ?C (194?F)? (b) If the saturated liquid ... Transcribed image text: Consider the sugar-water phase diagram, Animated Figure 10.1: (a) How much sugar will dissolve in 1000 g of water at 70°C? i 5050 g (b) If the saturated liquid solution in part (a) is cooled to 20°C, some of the sugar will precipitate out as a solid, because as seen in the figure, the composition of the saturated liquid solution drops to 64 wt% sugar. 18 Jan 2021 — 1 Answer to Consider the sugar–water phase diagram of Figure. (a) How much sugar will dissolve in 1500 g water at 90(C (194(F)?1 answer · Top answer: Solution: a) Given Data: Mass of water \(m_w=1500\:g\) Temperature \(T=90^0C\) Now from the given suger-water phase diagram the concentration of sugar ... 9.1 Consider the sugar-water phase diagram of Figure 9.1. (a) How much sugar will dissolve in 1500 g of water at 90°C (194°F)? (b) If the saturated liquid solution in part (a) is cooled to 20°C (68°F), some of the sugar will precipitate out as a solid.
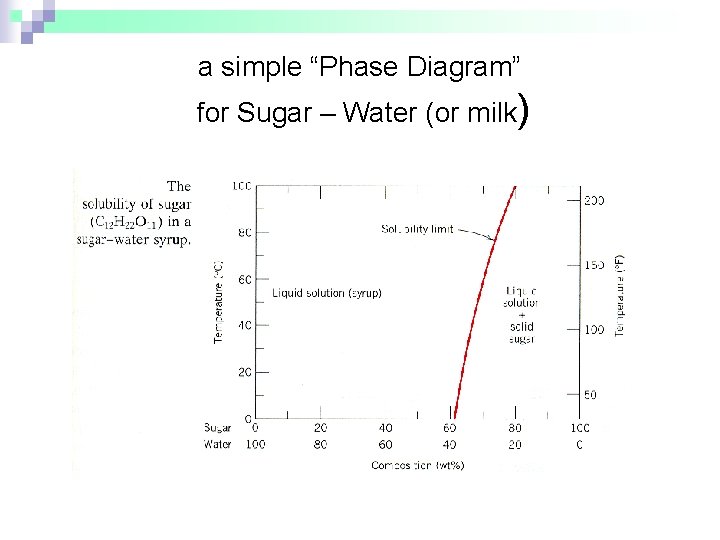
Ex: Phase Diagram: Water-Sugar System. Question: What is the. solubility limit at 20C? Answer: 65wt% sugar. If C. o < 65wt% sugar: sugar. If C. o > 65wt% sugar: syrup + sugar. • Solubility limit increases with T: e.g., if T = 100C, solubility limit = 80wt% sugar. Pure Sugar. Temperature (°C) 0 20 40 60 80 100 Co=Composition (wt% sugar) L ... Answer: 65 wt% sugar . If Co < 65 wt% sugar: syrup If Co > 65 wt% sugar: syrup + sugar. 65 Sucrose/Water Phase Diagram Pure Sugar Temperature (°C) 0 20 40 60 80 100 Co =Composition (wt% sugar) L (liquid solution i.e., syrup) Solubility Limit L (liquid) + S (solid 20 sugar) 40 60 80 100 Pure Water Adapted from Fig. 9.1, Callister 7e. Consider the sugar–water phase diagram below, (a) How much sugar will dissolve in 1000 g of water at 80°C (176°F)? (b) If the saturated liquid solution in part (a) is cooled to 20°C (68°F), some of the sugar will precipitate out as a solid. Transcribed image text: Consider the sugar-water phase diagram, Animated Figure 9.1: (a) How much sugar will dissolve in 1000 g of water at 80°C? lg [The tolerance is +/-10%.] (b) If the saturated liquid solution in part (a) is cooled to 20°C, some of the sugar will precipitate out as a solid, because as seen in the figure, the composition of the saturated liquid solution drops to 64 wt% sugar.
Science; Chemistry; Chemistry questions and answers; Consider the sugar-water phase diagram, Animated Figure 10.1 (a) How much sugar will dissolve in 1000 g of water at 70°C? e (b) If the saturated liquid solution in part (a) is cooled to 20°C, some of the sugar will precipitate out as a solid, because as seen in the figure, the composition of the saturated liquid solution drops to 64 wt% sugar. 14:440:407 ch9 Question 9.1 Consider the sugar–water phase diagram of Figure 9.1. (a) How much sugar will dissolve in 1500 g water at 90°C (194°F)? ... 9.1 Consider the sugar–water phase diagram of Figure 9.1. (a) How much sugar will dissolve in 1500 g water at 90°C (194°F)? (b) If the saturated liquid solution in part (a) is cooled to 20°C (68°F), some of the sugar will precipitate out as a solid. What will be the composition of the saturated liquid solution (in wt% sugar) at 20°C? Question 9.1 Consider the sugar–water phase diagram of Figure 9.1. (a) How much sugar will dissolve in 1500 g water at 90°C (194°F)?.

Consider A Sugar Water Phase Diagram A How Much Sugar Will Dissolve In 1000 G Of Water At 90 O C B If The Saturated Liquid Solution In Part A Is Cooled To
Solved Consider The Sugar Water Phase Diagram How Nuch Sugar Wll Dissolve In 1000 Water 80pc If The Saturated Liquid Solution In Part Is Cooled To 20pc Some Of The Sugar Will

Chapter 9 Phase Diagram Hfang2 Mece3345 Lectures Materials Science Chapter 9 1 Chapter 9 Phase Diagram Pdf Document
Solved View Policies Current Attempt In Progress Consider The Sugar Water Phase Diagram Animated Figure 9 1 A How Much Sugar Will Dissolve In 1 Course Hero
Polymers Free Full Text Application Of Differential Scanning Calorimetry Dsc And Modulated Differential Scanning Calorimetry Mdsc In Food And Drug Industries Html


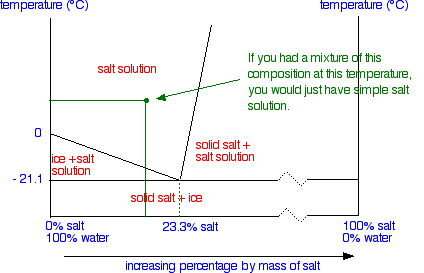

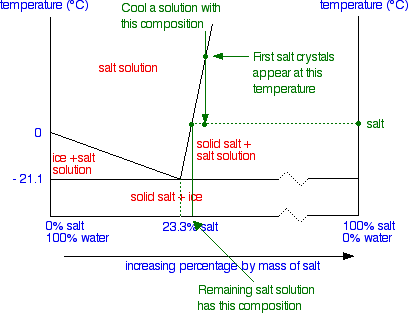
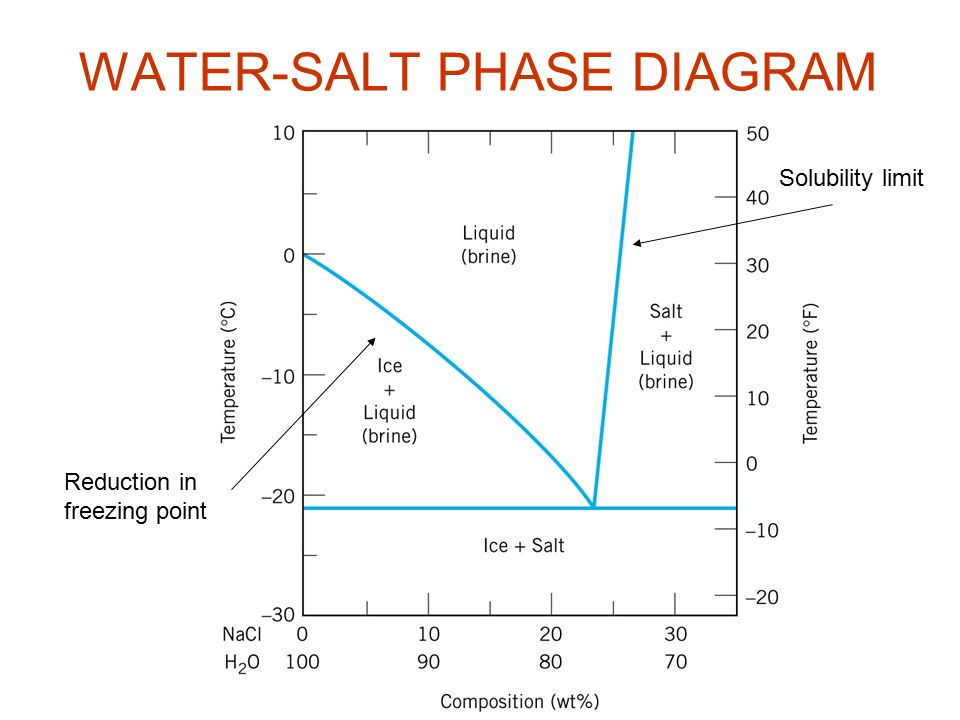


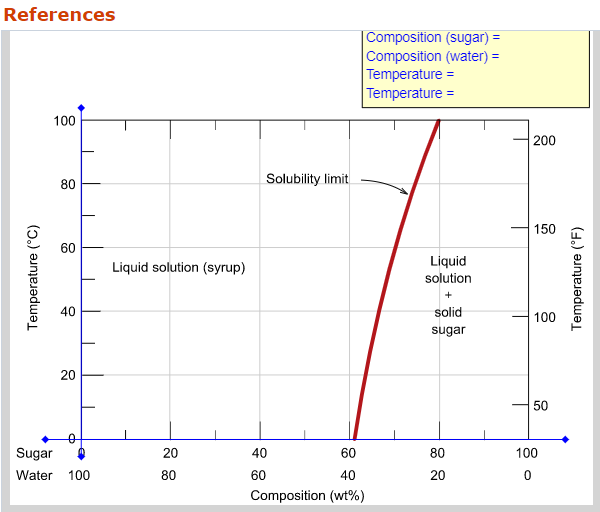
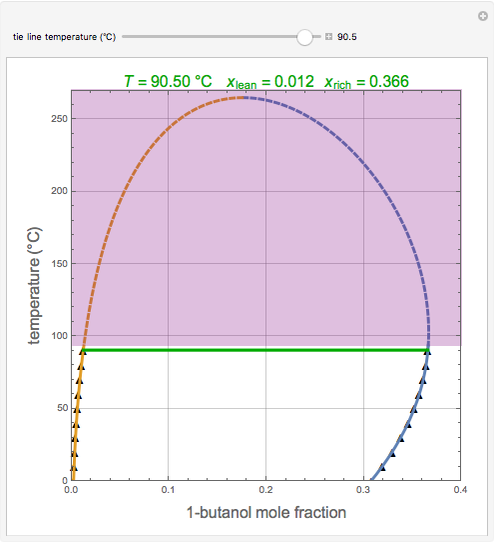








0 Response to "41 consider the sugar-water phase diagram"
Post a Comment