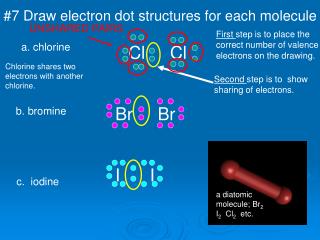
41 electron dot diagram for br2
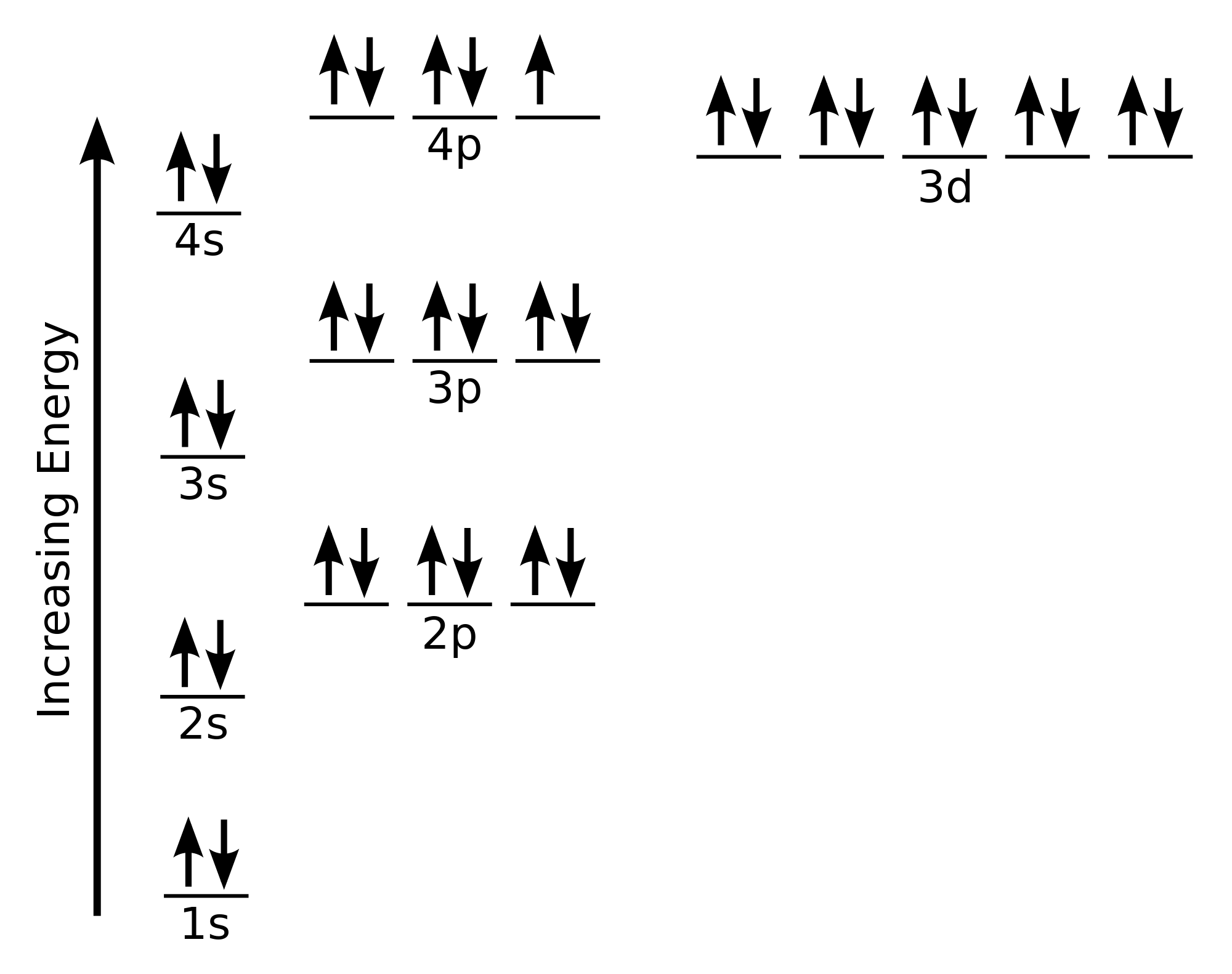
Bromine is an active ingredient in four products; two products with multiple active ingredients and two products as the sole active ingredient. The multiple active ingredient products control mold, mildew, fungi, insects, and odors in exposed surfaces of bedding, mattresses, textiles, drapes, upholstered furniture, rugs, carpets, and storage areas. To draw the Lewis electron dot diagram we picture in our minds the symbol for Mg in a box with all of its core electrons (i.e., 1s22s22p6). Then we place the valence electrons around the sides of the box with each side representing an orbital in the outermost energy level.
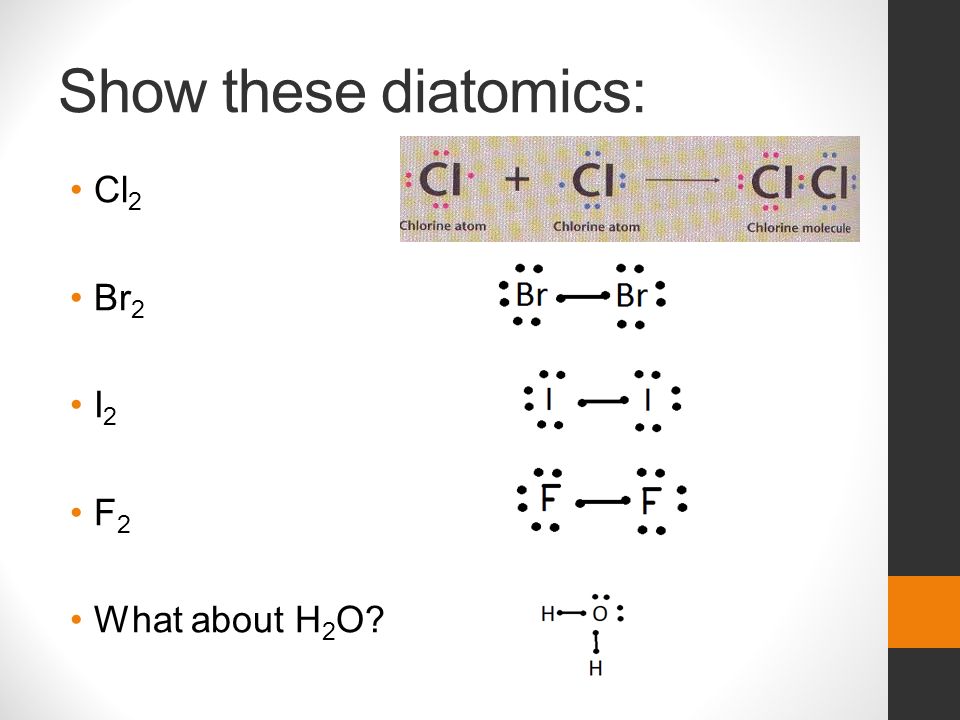
Draw the Lewis structure for Br2.... Draw the Lewis structure for SiO2.... Draw the Lewis structure for OCl2.... Draw the Lewis structure for FSiN.... Other sets by this creator. Economics Unit 11 Quiz. 15 terms. webbie75. Economics Test Units 6-10. 39 terms. webbie75.

Electron dot diagram for br2
A Lewis electron dot diagram (or electron dot diagram or a Lewis diagram or a Lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. Electrons exist outside of an atom’s nucleus and are found in principal energy levels that contain only up to a specific number of electrons. What is the bond between Br atoms in a Br2 molecule? The bond between Br atoms in an Br2 molecule is concealment and is formed by sharing of two valence ... To find the number of valence electrons in groups 13-18, you take the series number and subtract 10 from it. Therefore, if Bromine is in series 17, 17-10 is 7, so Bromine has 7 valence electrons. The electron dot diagram to the left depicts the number of valence electrons bromine has. The dots are arranged in a specific pattern to show how many ... Answer (1 of 2): First calculate the number of electrons required (ER) to give everybody an octet. That would be: ER =2*8= 16 Then calculate how many electrons you actually have. Look in the periodic table. Bromine is in column 17. Ten of that is the d block, so subtract 10. Gives 7, being the ...
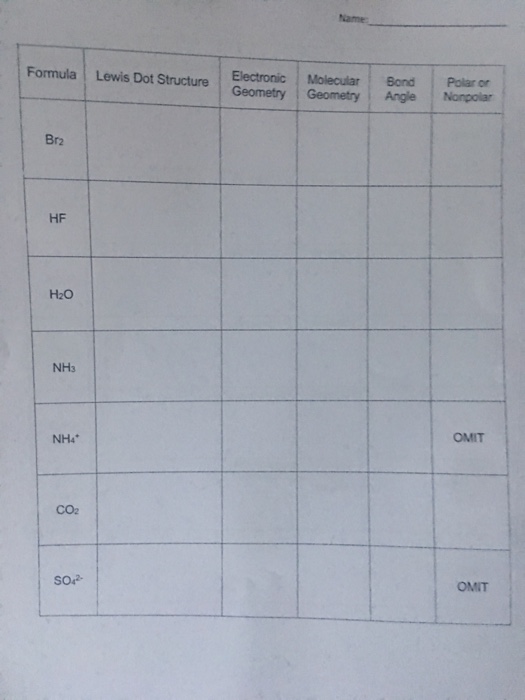
Electron dot diagram for br2. A. Draw Lewis structure for Br2 and HBr. Q1. B. What is the molecular geometr (shape of the molecule) for Br2 and HBr. Q1. C. State whether Br2 is polar or nonpolar. Q1. D. State whether HBr is polar or nonpolar. Q2. A. Draw Lewis structure for BBr Q2. B. What is the molecular geometry (shape of the molecule) for BBrs Q2. C. State whether BBr ... A step-by-step explanation of how to draw the BrCl Lewis Dot Structure.For the BrCl structure use the periodic table to find the total number of valence elec... In the box below, draw the electron-dot (Lewis) structure of carbon di0>äde. In the box below, draw a Lewis electron-dot diagram for a molecule of phosphorus trichloride, PC13 (c) ammonia 60) 61) 62) 65) In the box provided, draw a Lewis electron-dot diagram for a molecule of chlorine, Ch. in terms of electrons, why the bonding in NaCl is Ionic. Moving onto drawing Lewis dot structures of various compounds: Step 1: Count the total number of valence electrons in the compound. Use the periodic table to figure out how many valence electrons each individual element of the compound contributes then add them all up. Upgrade to remove ads.
What is the electron dot structure of magnesium bromide? The structure should have single bond between Mg with the two bromides, each contributing one electron. The rest of the valence electrons are around bromide as to fulfill the octet rule of 8 electrons for each bromide attached to the magnesium. A step-by-step explanation of how to draw the Br2 Lewis Dot Structure (Bromine gas).For the Br2 structure use the periodic table to find the total number of ... Get the free "Lewis structure" widget for your website, blog, Wordpress, Blogger, or iGoogle. Find more Chemistry widgets in Wolfram|Alpha. Answer to: Draw and explain the electron-dot diagram for SiCl2Br2. By signing up, you'll get thousands of step-by-step solutions to your homework...
A step-by-step explanation of how to draw the MgBr2 Lewis Dot Structure.For MgBr2 we have an ionic compound and we need to take that into account when we dra... There is only one element in bromine molecule. Bromine is a group VIIA element in the periodic table and contains seven electrons in its last shell. Now, we know how many electrons are there in valence shells of bromine atoms. valence electrons given by bromine atoms = 7 * 2 = 14. Total valence electrons = 14. What is the correct Lewis structure for Br2?:Br - Br: What is the correct Lewis structure for O2?:O = O: What is the correct Lewis structure for N2?: N ≡ N : The Lewis structure for carbon monoxide is This structures shows. 2 lone pairs and 3 bonding pairs. Which sequence below represents the proper order of increasing bond strength? Answer (1 of 2): First calculate the number of electrons required (ER) to give everybody an octet. That would be: ER =2*8= 16 Then calculate how many electrons you actually have. Look in the periodic table. Bromine is in column 17. Ten of that is the d block, so subtract 10. Gives 7, being the ...
What is the Lewis structure of br2? The bromine molecule is a diatomic molecule with a single Br-Br bond. Since the Br atom lies in the 17th group of the periodic table, it has seven valence electrons. Both of the Br atoms use one of their valence electrons to form the single bonds and the remaining six on each of them form three lone pairs on ...
Transcript: This is the Br2 Lewis structure. Steps for Writing Lewis Structures. ; In the Lewis structure for BrCl 3 there are a total of 28 valence electrons. Lewis structures depict the bonds between atoms of a molecule, as well as any unbonded electron pairs. Q1. SBr 2 is similar to the SCl 2 Lewis structure.
A step-by-step explanation of how to draw the Br2 Lewis Dot Structure (Diatomic Bromine).Note that Diatomic Bromine is often called Molecular Bromine or just...
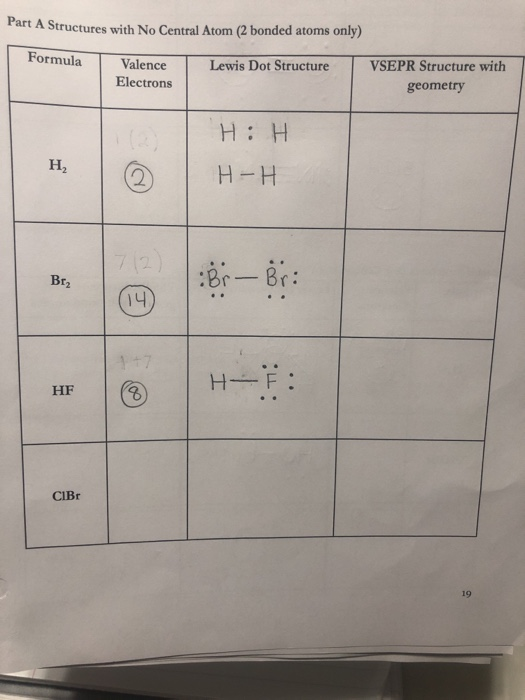
Br2 Sketch the proper Lewis structure for this substance. Be sure to follow octet/duet rules for each atom and use the total number of valence electrons available. Use your drawing to answer the following questions. Count the number of each type of electron domain on each atom. There is not a central atom here, and the two atoms are equivalent.
There are two types of diagrams one is the Lewis diagram the other is the Electron dot diagram. To make the electron dot diagram you put the electron symbol and put a dot on one of the sides for ...
Nov 20, 2021 · Lewis Structure of Br2. A compound’s Lewis Structure depicts the arrangement of its underlying valence shell electrons. The structure uses dots and lines to depict electrons and the bonds between 2 electrons, respectively. The idea behind drawing the Lewis structure of the atom of a compound is to get an understanding of the chemical and physical properties the compound would possess.
How many bonding pairs are in Br2? The one bond and three lone pairs for the bromine atom give it an octet around it. What do Lewis structures show? Lewis structures (also known as Lewis dot structures or electron dot structures) are diagrams that represent the valence electrons of atoms within a molecule.
Br2 is the molecular formula name for bromine gas, dibromine or molecular bromine. Bromine has the Lewis structure of an single bonded bromine (Br) atoms represented by: .. ..:
Answer (1 of 2): H has one peripheric electron, C has 4, N has 5 and O has 6. You wrote the atoms in your molecule in a particular order, which indicates the molecule must have this Lewis structure :
It has 7 valence electrons. We have two of them though. Multiply that by 2, for a total of 14 valence electrons for the Br2 Lewis structure. First, we'll draw two Bromine atoms next to each other. We have 14 valence electrons for Br2. We'll put two between atoms to form a chemical bond. Then we'll go around the atoms: 2, 4, 6, 8 and 10, 12, 14.

Br Bromine Element Information Facts Properties Trends Uses And Comparison Periodic Table Of The Elements Schoolmykids
A step-by-step explanation of how to draw the I2 Lewis Dot Structure (Iodine Gas).For the I2 structure use the periodic table to find the total number of val...
Lewis Structure: The Lewis structure representation of an atom or a molecule is the simplest two-dimensional form of the species. It shows the element with its symbol and the electrons with dots.
Dibromide(.1-) | Br2- | CID 5460533 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety ...
Answer (1 of 2): First calculate the number of electrons required (ER) to give everybody an octet. That would be: ER =2*8= 16 Then calculate how many electrons you actually have. Look in the periodic table. Bromine is in column 17. Ten of that is the d block, so subtract 10. Gives 7, being the ...
To find the number of valence electrons in groups 13-18, you take the series number and subtract 10 from it. Therefore, if Bromine is in series 17, 17-10 is 7, so Bromine has 7 valence electrons. The electron dot diagram to the left depicts the number of valence electrons bromine has. The dots are arranged in a specific pattern to show how many ...
A Lewis electron dot diagram (or electron dot diagram or a Lewis diagram or a Lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. Electrons exist outside of an atom’s nucleus and are found in principal energy levels that contain only up to a specific number of electrons. What is the bond between Br atoms in a Br2 molecule? The bond between Br atoms in an Br2 molecule is concealment and is formed by sharing of two valence ...

Classwork Standard 2e Terms 135 Article 136 Mastering Concept 146 57 64 Practice Problems 139 18 22 141 23 Homework Cornell Notes 5 3 Sec Ppt Video Online Download















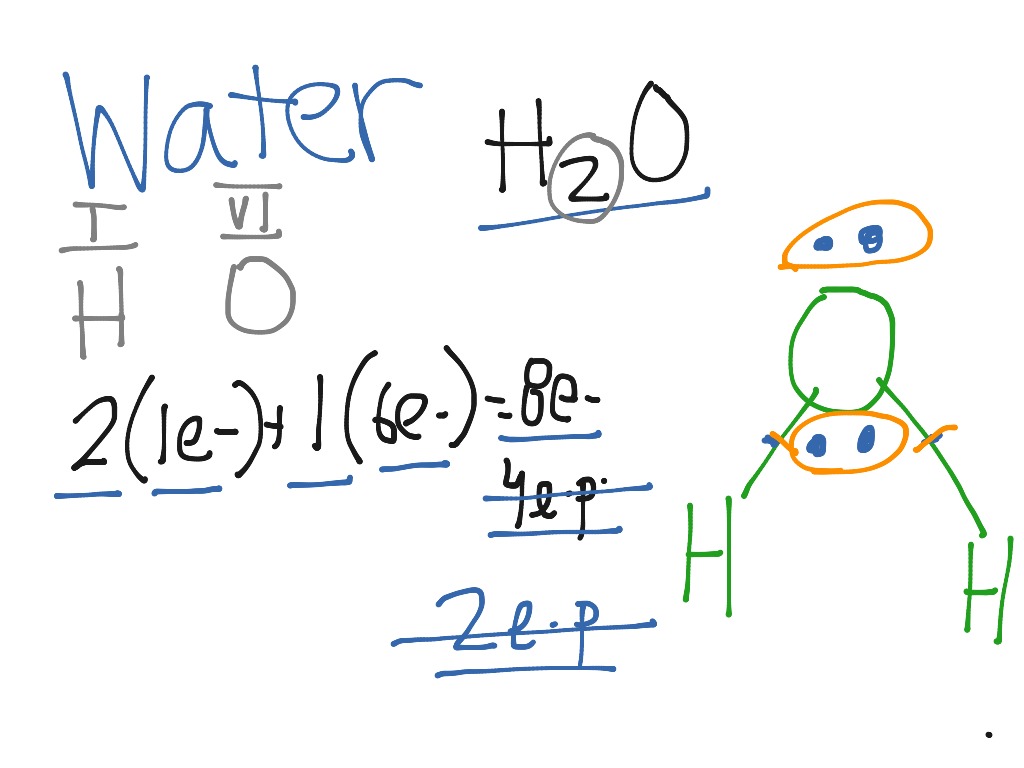










0 Response to "41 electron dot diagram for br2"
Post a Comment