35 lewis dot diagram for nitrogen
Lewis Dot Diagram Worksheet Use the Bohr models to determine the number of valance electrons. Once you have found the number of valance electrons, place them around the elements symbol. Element Atomic # Atomic Mass Protons Neutrons Electrons Lewis Dot Carbon 6 12 6 6 6 l Hydrogen 1 1 1 0 1 H Lithium 3 7 3 4 3 Li Magnesium 12 24 12 12 12 Mg: Boron B5 …
20/12/2021 · The molar mass of nitrogen tetroxide is 92.011 g/mol. It’s melting and boiling points are -11.2°C and 21.69°C respectively. Its density is 1.44 g/cm3. It easily reacts with water to form nitric and nitrous acids. Now, let us discuss other properties of N2O4 such as lewis structure, geometry, hybridization, and MO diagram. N2O4 Lewis Structure
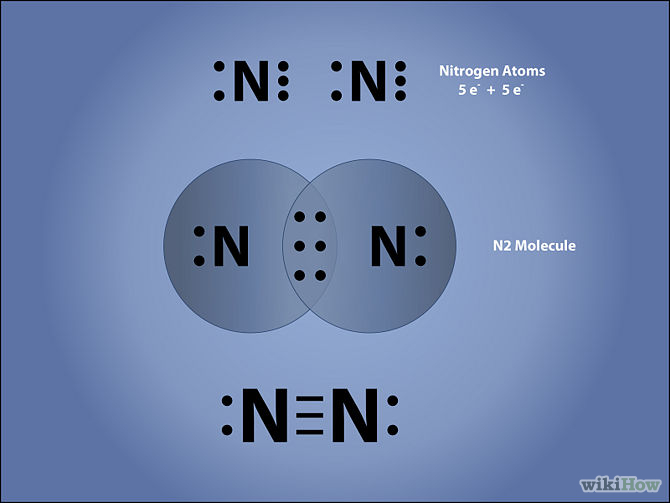
The N2 Lewis structure has a triple bond between two nitrogen atoms. According to the octet rule, nitrogen atoms need to bond three times.

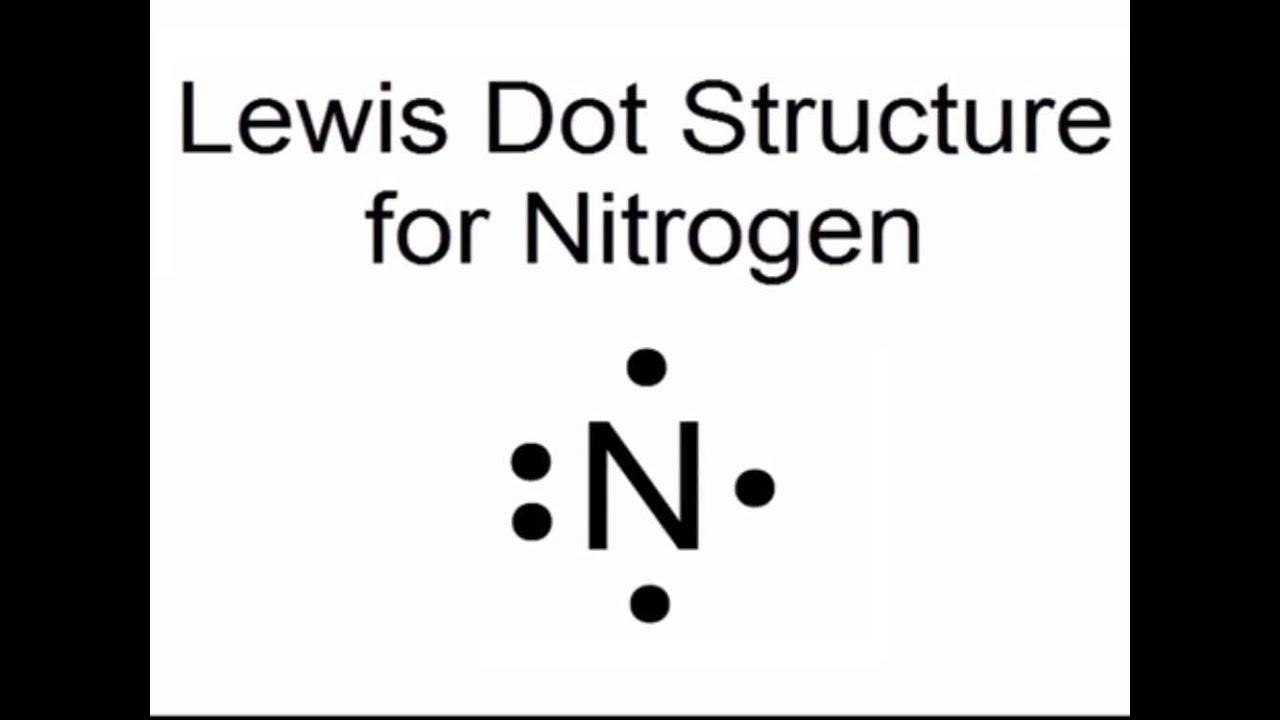
Lewis dot diagram for nitrogen
08/12/2021 · Let us sketch a probable Lewis Structure for Nitrogen Trifluoride. We have put the valence dot structures and made a sketch of the NF3 molecule. Let us now understand the next important concept of Lewis Structure: Octet Rule. The main group (group 1- 17) elements tend to attain the electronic configuration of the outermost shell of noble gas elements. Noble gases …
Lewis structures, also known as Lewis dot formulas, Lewis dot structures, electron dot structures, or Lewis electron dot structures (LEDS), are diagrams that show the bonding between atoms of a molecule, as well as the lone pairs of electrons that may exist in the molecule. A Lewis structure can be drawn for any covalently bonded molecule, as well as coordination compounds.
Steps to draw the lewis dot structure for CN¯ (Cyanide) 1. Count Valence electrons in CN¯ In the first step, count all valence electrons present in the CN molecule. As carbon belongs to the 14th group in the periodic table, so, it has 4 valence electrons and Nitrogen belongs to the 15th, so, it has 5 valence electrons.
Lewis dot diagram for nitrogen.
Ammonia is NH3. Nitrogen has five valence electrons and each hydrogen brings one to the molecule. It's easy to see that those three electrons from the hydrogens ...
A Lewis dot structure is also called a Lewis structure, a Lewis dot diagram, an electron dot structure, or a dot diagram. When people mention Lewis or dots, they are talking about drawing a molecule to show how the atoms bond. Download the Lewis Dot Structure Worksheet . Download and print the black and white pdf. It’s 5 printer-friendly pages. There’s an answer key …
Lewis Structure (electron dot diagram) for the oxygen molecule, O 2, OR ... Lewis Structure (electron dot diagram) for the nitrogen molecule, N 2, :N..... N: There are 3 bonding pairs of electrons shared between the 2 nitrogen atoms, and each nitrogen atom also has 1 lone pair (non-bonding) pair of electrons. In the Valence structure for the nitrogen molecule, each …
11 Apr 2016 — The Lewis structure of Nitrogen atom can be drawn if one knows the number of valence electrons of Nitrogen. The electronic configuration of ...1 answer · Explanation: The Lewis structure of Nitrogen atom can be drawn if one knows the number of valence electrons of Nitrogen. The electronic configuration ...
Lewis Dot Structures Objectives: 1. Draw Lewis structures for atoms, ions and simple molecules. 2. Use Lewis structures as a guide to construct three-dimensional models of small molecules. 3. Determine the electron and molecular geometry of the produced molecules. Background: Scientists often create models to represent either a physical or abstract system or …
In N2 Lewis structure,two nitrogen atoms has shared six valence electrons and every nitrogen atom has one lone pairs .In N2 Lewis structure,there are ten ...3 answers · 4 votes: As nitrogen is in fifth group in periodic table therefore it will have five electrons in the ...
14/02/2019 · NO2 (Nitrogen Dioxide) Lewis Dot Structure. Alex Bolano on February 14, 2019 1 Comment 🤔. Nitrogen Dioxide (NO 2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom. At room temperatures, nitrogen dioxide is a reddish-brown gas that has a density of 1.8 g/dm 3. It …
The NO2 Lewis structure has a total of 17 valence electrons. It's not common to have an odd number of valence electrons in a Lewis structure. Because of this we ...25 Oct 2016 · Uploaded by Wayne Breslyn
As per the NCl3 lewis dot structure, nitrogen is the central atom that has 3 bonded pairs of electrons and one lone present on it. Hence the formula of NCl3 becomes AX 3 N 1 So, according to the VSEPR chart , if the molecule has the formula of AX 3 N 1 then the molecule shape of that molecule is trigonal pyramidal, and electron geometry is tetrahedral.
Doc Brown's Chemistry: Chemical Bonding and structure GCSE level, IGCSE, O, IB, AS, ... The full Lewis dot and cross diagram for the nitrogen molecule ...
Describe the electron dot diagram system of representing structure. Draw electron dot diagrams for ... nitrogen, 1 s 2 2 s 2 2 p 3, 5 valence electrons.Nitrogen: 1 s 2 2 s 2 2 p 3Neon: 1 s 2 2 s 2 2 p 6Lithium: 1 s 2 2 s 1Beryllium: 1 s 2 2 s 2







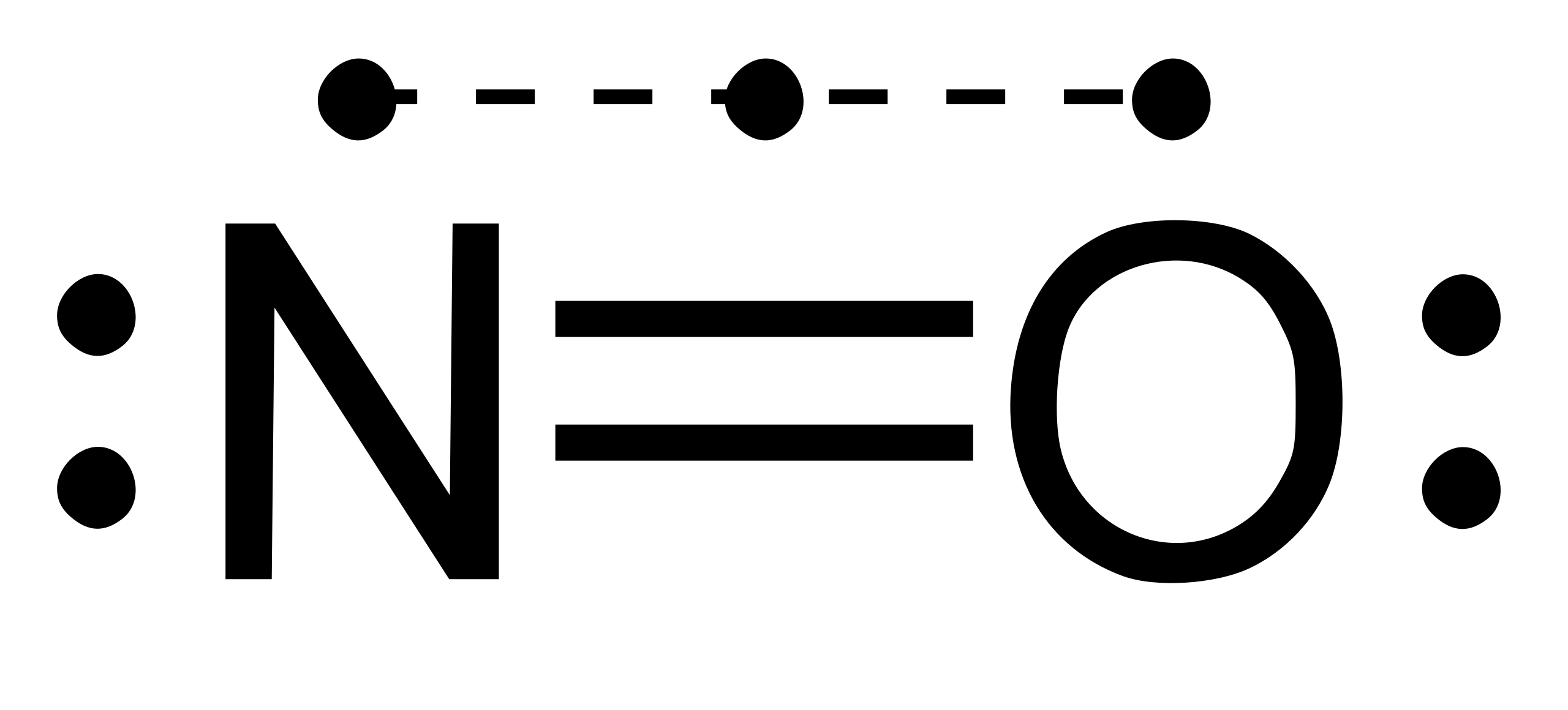







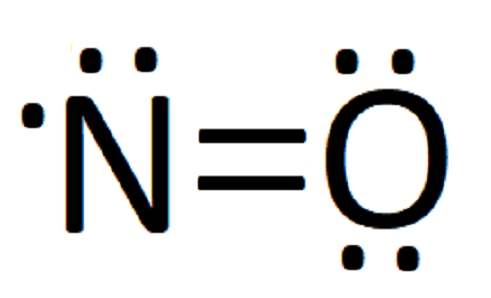
![Draw the electron dot structure of Nitrogen molecule [N = 7]](https://haygot.s3.amazonaws.com/questions/1890007_1909574_ans_16e2a124f2974b5694de1a9f3c97eebd.png)





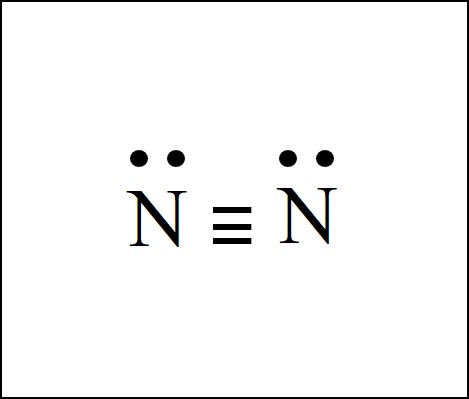

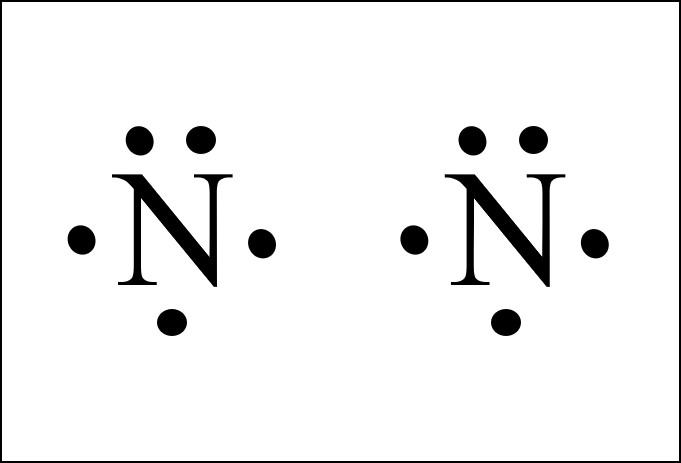


0 Response to "35 lewis dot diagram for nitrogen"
Post a Comment