39 lewis dot diagram for arsenic
What would be the correct Lewis dot diagram for arsenic trifluoride AsF3? What is the correct Lewis dot diagram for CH4? What is wrong with the Lewis dot structure for IO-The bond between I and O should be represented with a single line. What does the charge on a polyatomic ion mean?
A Lewis structure is a model that uses electron-dot structures to show how electrons are arranged in molecules. Pairs of dots or lines represent bonding pairs. Drawing the Lewis Structure for AsH3 (Arsenic Trihydride). Viewing Notes: The Lewis structure for AsH3 is similar to AsF3 structure. The Arsenic atom goes in.
The amount of dots in total in the Lewis Dot Diagram of AsF4+ (Arsenic tetrafloride) is twenty six. Each "F" (fluorine atom) has six dots in total, two on three "sides". The As (arsenic atom) in...
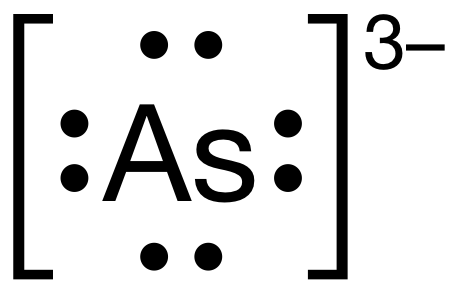
Lewis dot diagram for arsenic
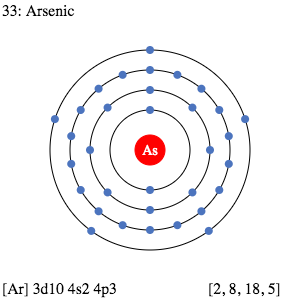
Electron Distributions Into Shells for the First Three Periods. A chemical element is identified by the number of protons in its nucleus, and it must collect an equal number of electrons if it is to be electrically neutral.
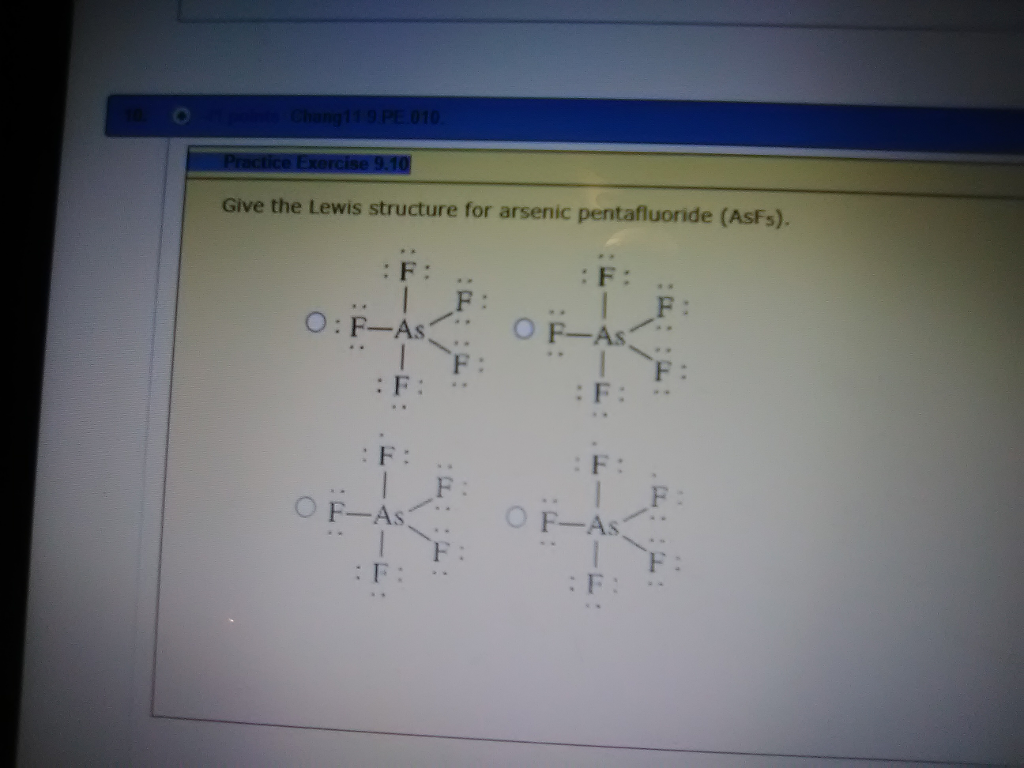
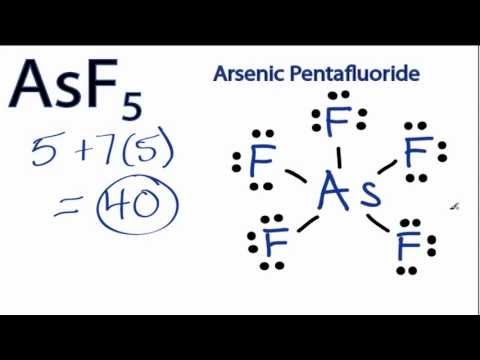
AsF5 lewis structure is made up of one Arsenic atom situated in a central position and five fluorine atoms that spaced evenly around the central atom. There is a total of 10 bonding electrons and 30 nonbonding electrons present in the lewis structure of AsF5.
Lewis Dot Diagram For Arsenic; Cub Cadet Lt1040 Drive Belt Diagram; Seymour Duncan Hsh Wiring Diagram; Sunrise Cicada F4 Aio No Osd Wiring Diagram; T9.560 Wiring Diagram; 2002 Jeep Cherokee 4.7 High Output O2 Sensor Wiring Diagram; 68 Camaro 700r4 Wiring Diagram; Kohler Ch20s Wiring Diagram; Duo Therm By Dometic Thermostat Wiring Diagram
Lewis dot diagram for arsenic.
Electron Dot Diagram Arsenic Electron dot diagrams, sometimes called Lewis dot diagrams, were first used by Gilbert N. Lewis in These diagrams are used as a. Comprehensive information for the element Arsenic - As is provided by this page including scores of Atomic Structure of Arsenic Electron Dot Model.
The answer is B) trigonal pyramidal. To determine the molecular geometry of arsenic trichloride, AsCl_3, you must take a look at its Lewis structure. One arsenic trichloride molecule will have a total of 26 valence electrons - 5 from the arsenic atom and 7 from each of the three chlorine atoms. The arsenic atom will be bonded to the three chlorine atoms through single bonds that account for 6 ...
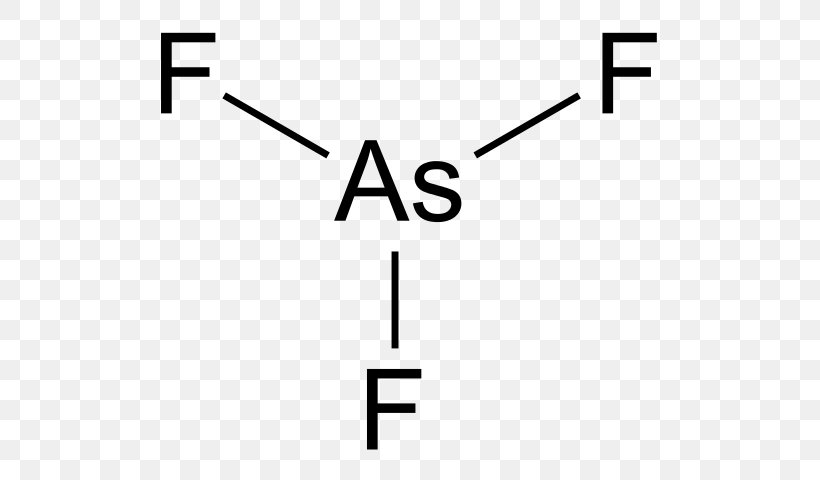
Nov 10, 2018 · Lewis Dot Diagram For Arsenic. The left diagram shows a Lewis dot structure of sodium with. Likewise, they .. A Lewis structure shows an arsenic atom single bonded to three fluorine atoms. These diagrams show the symbol of the element with as many dots around it as there are electrons in the outermost energy level.
A step-by-step explanation of how to draw the Arsenic (As) Lewis Dot Structure.For the ArsenicLewis structure use the periodic table to find the total number...
Draw the correct Lewis dot structure for SCl. 2. Draw the correct Lewis dot structure for SCl. 2. Draw the correct Lewis Dot Structure for O. 2. Draw the correct Lewis Dot Structure for O. 2. Electron Configuration. ... Arsenic, As. Write the orbital notation for the element carbon ↑↓↑↓↑ ↑ .
Paul Books Chapter. Give The Lewis Structure For Arsenic Pentafluoride Asf5 31+ Pages Solution [3mb] - Updated 2021
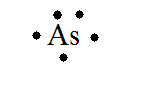
Answer : The correct representation of Lewis-dot structure for 5A element, Arsenic is shown below. Lewis-dot structure : It shows the bonding between the atoms of a molecule and it also shows the unpaired electrons present in the molecule. The number of valence electrons shown by 'dot'.
Lewis Dot Diagrams (Structures) for Atoms and Ions Predicting Oxidation Numbers. Pre AP Chemistry Unit 6 HW Packet Name _____ WKS 6.1- Classifying Ionic versus Covalent/ Lewis Dot Structures of Atoms. Classify the following compounds as ionic ([metal or ammonium ion] + [non-metal or polyatomic ion]), covalent (nonmetal+ nonmetal). CaCl2 / CO2 / H2O
Arsenic pentoxide is non-combustible; the substance itself does not burn, but it may decompose upon heating to produce corrosive and/or toxic fumes. Fire may produce irritating, corrosive, and/or toxic gases. For small fires use dry chemical, CO2, or water spray. For large fires use water spray, fog, or regular foam.
In the Lewis dot structure for AsH3, how many dots are on the arsenic? 2 4 6 0
May 29, 2021 · What is the electron dot notation for arsenic? After the 3d sublevel is filled, the remaining three electrons will be in orbitals in the 4p sublevel. Thus, the electron configuration for arsenic is [Ar]4s 2 3d 10 4p 3. Before writing the electron – dot structure for arsenic, note that arsenic’s ten 3d electrons are not in the highest ...
The amount of dots in total in the Lewis Dot Diagram of AsF4+ (Arsenic tetrafloride) is twenty six. Each "F" (fluorine atom) has six dots in total, two on three "sides".
The Lewis structure for AsF 3 is similar to AsCl 3 structure. Since they are in the same Group on the periodic table they each have the same number of electrons their structures are similar. The Arsenic atom goes in the center of the Lewis structure since it is the least electronegative atom. For the AsF 3 Lewis structure there are a total of ...
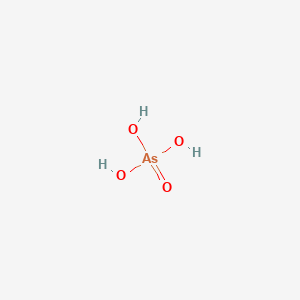
Arsenic acid is an arsenic oxoacid comprising one oxo group and three hydroxy groups attached to a central arsenic atom. It has a role as an Escherichia coli metabolite. It is a conjugate acid of an arsenate (1-) and an arsenate ion. Arsenic acid, liquid appears as a clear colorless aqueous solution. Noncombustible.
Arsenic is isoelectronic with nitrogen (they are both Group V elements), so there are 5 valence electrons. Look at the position of arsenic in the Periodic Table. It is under phosphorus, which is under nitrogen. These elements are thus isoelectronic, and their chemistry should be similar to a first approximation. Consider ammonia versus phosphine versus arsine, i.e. NH_3 versus PH_3 versus AsH_3.
A step-by-step explanation of how to draw the AsBr3 Lewis Dot Structure.For the AsBr3 structure use the periodic table to find the total number of valence el...
DRAWING LEWIS DOT DIAGRAMS 1) Given a Bohr model, count the 2 Mg DRAWING ELECTRON DOT DIAGRAMS Draw the Dot Diagram for arsenic (As). Comprehensive information for the element Arsenic - As is provided by this page including scores of Atomic Structure of Arsenic Electron Dot Model. The left diagram shows a Lewis dot structure of sodium with.
The lewis dot structure is a diagram to show the bonding between the atoms of a molecule and pairs of electrons that may exist. This eight electrons are found in four pairs. I show you where magnesium is on the periodic table and how to determine how many valence electrons magnesium has. Ionic bonding chemistry for non majors.
2. Draw the Lewis dot structures for each of the following molecules: a. H 2 S c. SO 3 b. CH 2 Br 2 d. HCN 3. Draw the Lewis dot structure for each of the following polyatomic ions: a. NH 4 + c. PO 4 -3 b. NO 3 - d. CO 3 2- 4. For the following molecules or ions (where the central atom is underlined): i. Draw the Electron dot structure. ii.
Drawing the Lewis Structure for AsH 3 (Arsenic Trihydride) Viewing Notes: The Lewis structure for AsH 3 is similar to AsF 3 structure. The Arsenic atom goes in the center of the Lewis structure since it is the least electronegative atom. Remember that Hydrogen (H) atoms always go on the outside of a Lewis structure.
The Lewis-dot structure of arsenic is shown below. Send VLD 11 months ago 6 0 Because Arsenic is in group 15 it will receive 5 valence electrons Start out by adding one dot around the symbol for Arsenic. You"ll end up with 4 around it and all you have to do is add another one.
9. Draw a Lewis dot diagram for the barium atom. 10. Draw the Lewis dot diagram for the silicon atom. I I, Draw the Lewis dot diagram for the iodine atom. 12. Draw the Lewis dot diagram for the xenon atom. 13. Hypothesize: Why are noble gases considered to be non-reactive? Your group will check your answers with the instructor before moving on.
What is the Lewis dot structure for arsenic? Chemical Bonding: AsH3 Lewis Structure The Arsenic atom goes in the center of the Lewis structure since it is the least electronegative atom. Remember that Hydrogen (H) atoms always go on the outside of a Lewis structure. For the AsH3 Lewis structure there are a total of 8 valence electrons available.
What is the Lewis dot structure for arsenic? The Arsenic atom goes in the center of the Lewis structure since it is the least electronegative atom. Remember that Hydrogen (H) atoms always go on the outside of a Lewis structure. For the AsH3 Lewis structure there are a total of 8 valence electrons available.

Draw an electron dot structure for an atom of each element a carbon b arsenic c polonium d potassium


























0 Response to "39 lewis dot diagram for arsenic"
Post a Comment