40 ethylene molecular orbital diagram
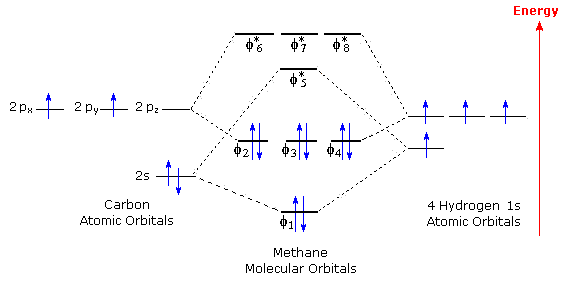
Creating molecular orbital diagrams for molecules with more than two atoms relies on the same basic ideas as the diatomic examples presented here. However, with more atoms, computers are required to calculate how the atomic orbitals combine.
Hello! Just finished a lecture in my ochem class and was going through the notes and had some questions if yall don't mind. I am already watching some youtube videos on molecular diagram and orbitals because I have no clue whats going on there but if you guys could please help out that would be great! * **First starting off with conjugation and energy, here is a pic: https://imgur.com/lmg6PDn** Does higher energy = less stable? So the first one monoene has a higher energy than the two dienes c...
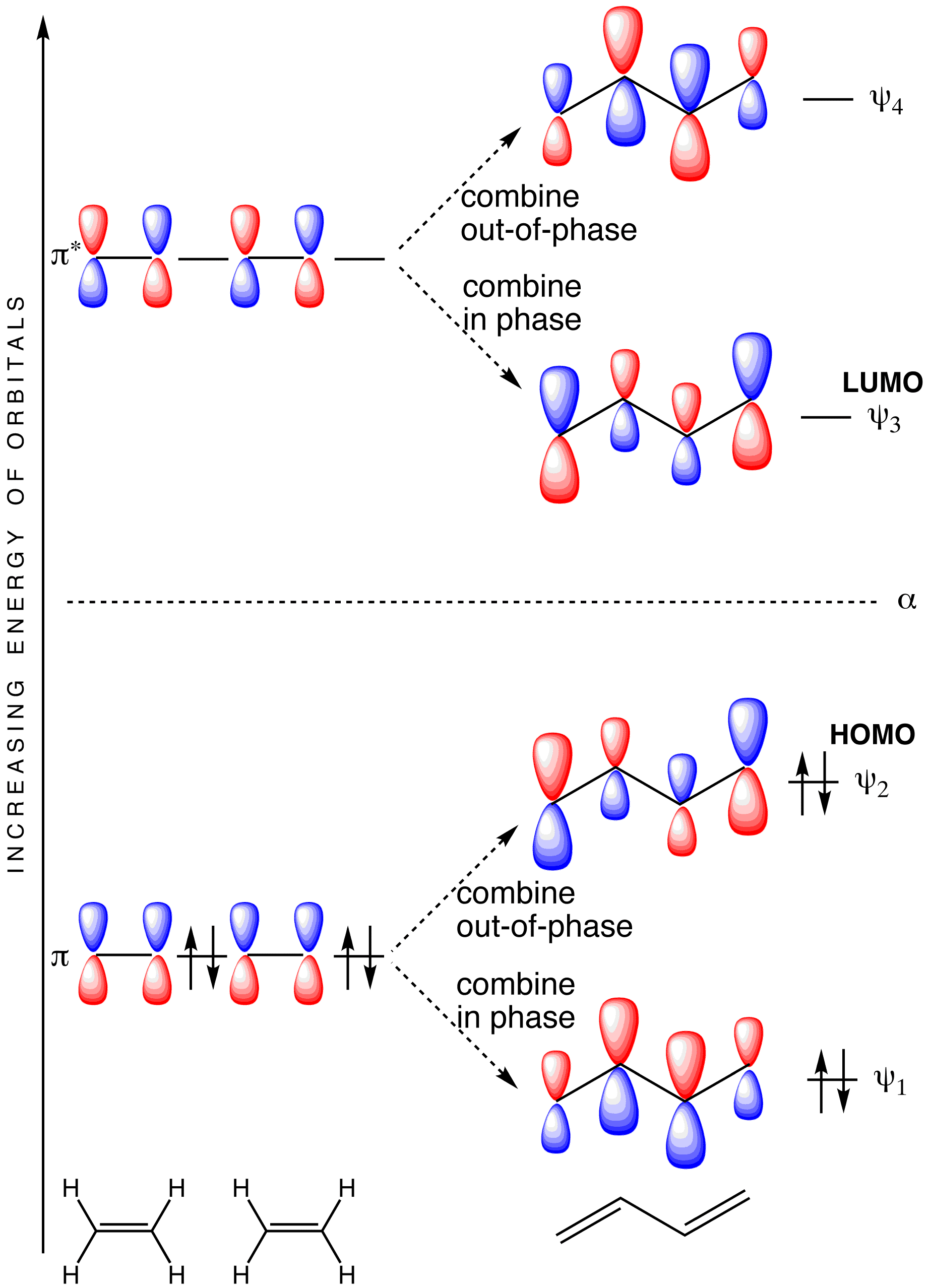
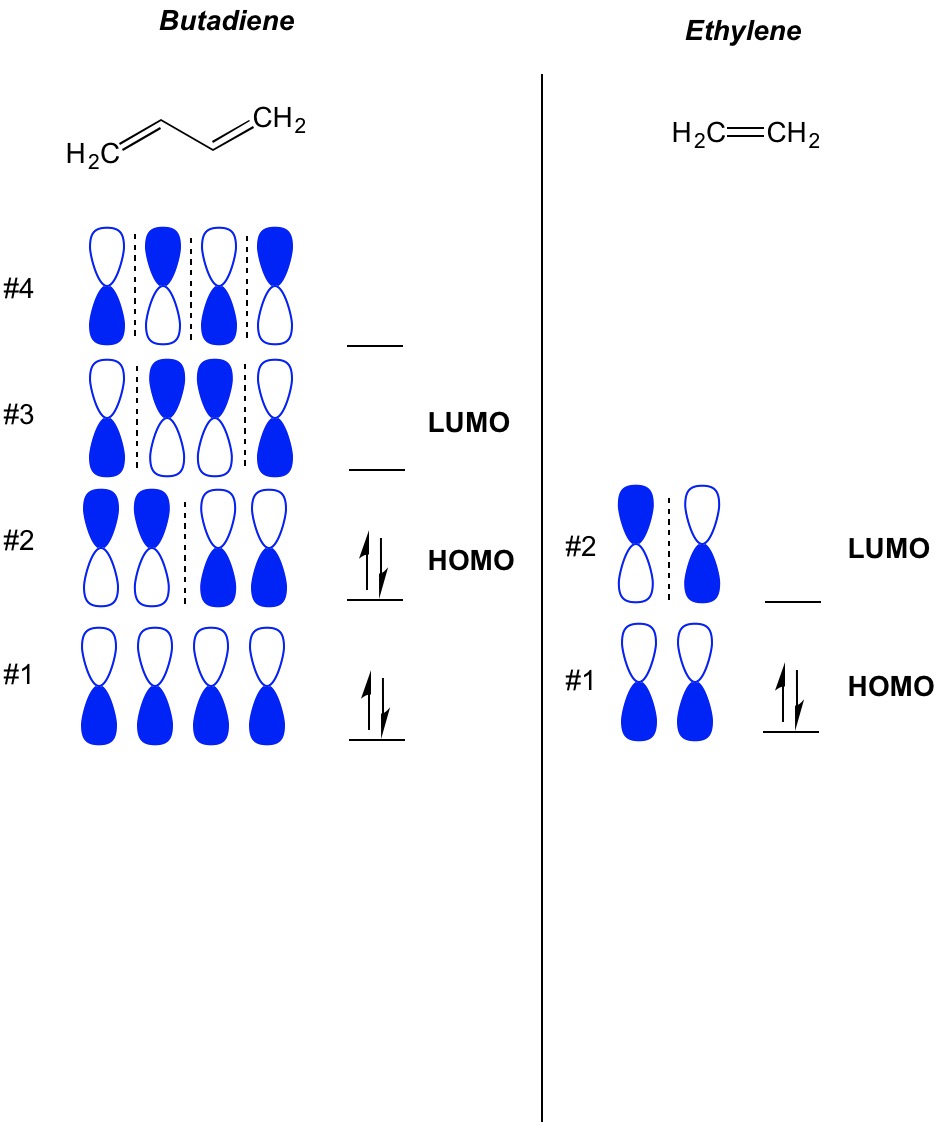
The molecular orbital diagram for the π-molecular orbitals of butadiene as a result of combining the π-molecular orbitals of two ethene molecules. This shows .Bonding orbitals in Ethene (Ethylene) sp 2 Background: Use the buttons to display the sp 2 orbitals that make up the sigma framework and the...
Ethylene molecular orbital diagram
The bonding molecular orbital concentrates electrons in the region directly between the two nuclei. Placing an electron in this orbital therefore stabilizes the H2 molecule. This diagram suggests that the energy of an H2 molecule is lower than that of a pair of isolated atoms.
A molecular orbital diagram, or MO diagram, is a qualitative descriptive tool explaining chemical bonding in molecules in terms of molecular orbital theory in general and the linear combination of atomic orbitals (LCAO) method in particular.
Ethene Molecular Orbital Diagram - Wiring Diagrams. 9 hours ago Bonding orbitals in Ethene (Ethylene) sp 2 Background: Use the buttons to display the sp 2 orbitals that make up the sigma framework and the remaining p orbitals which form the pi-bond. π Molecular Orbitals of Ethene.
Ethylene molecular orbital diagram.
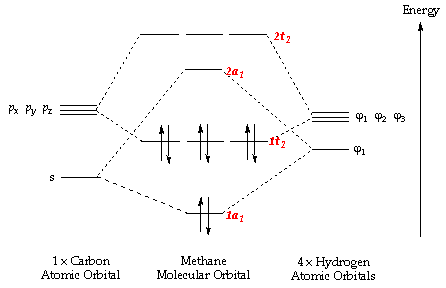
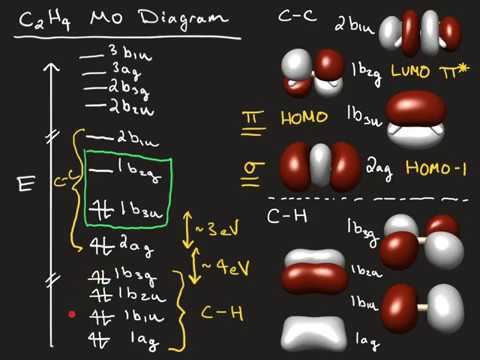
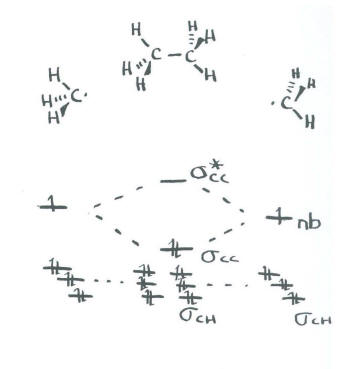
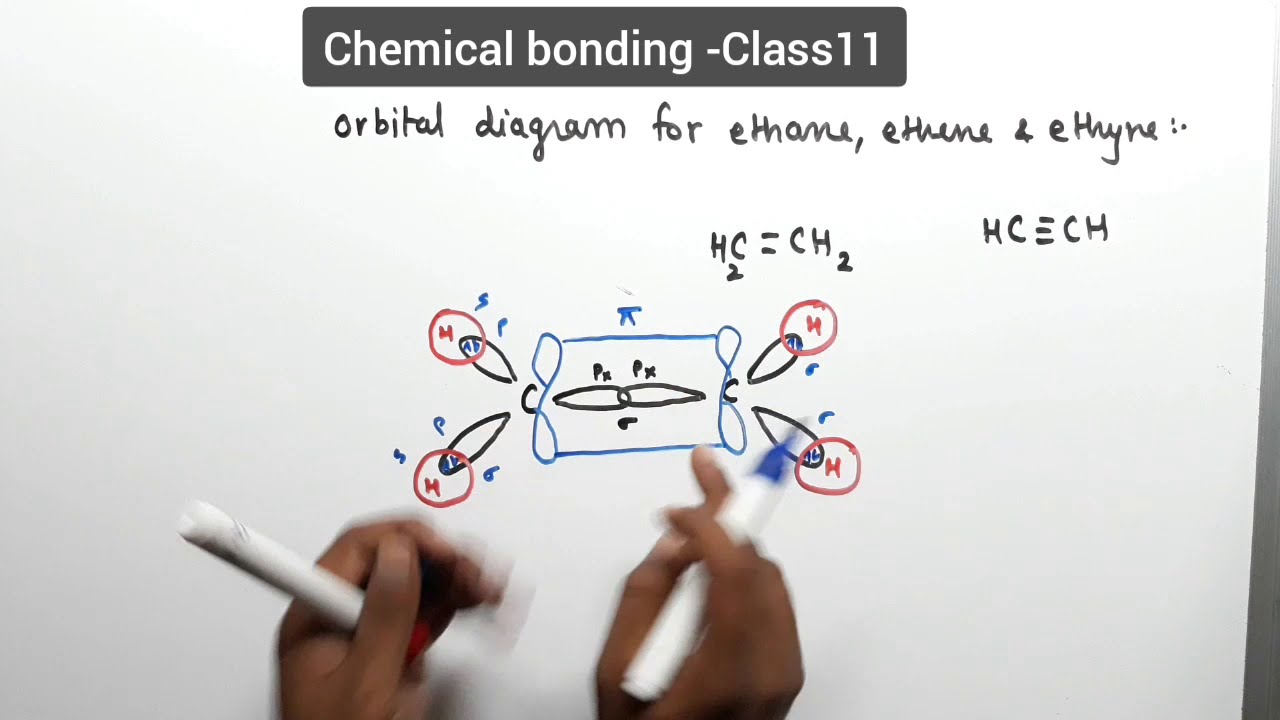
A molecular orbital diagram of ethene is created by combining the twelve atomic orbitals associated with four hydrogen atoms and two sp 2 hybridized carbons Molecular orbitals for ethene (ethylene) In the bonding pi orbital, the two shaded lobes of the p orbitals interact constructively with each other...
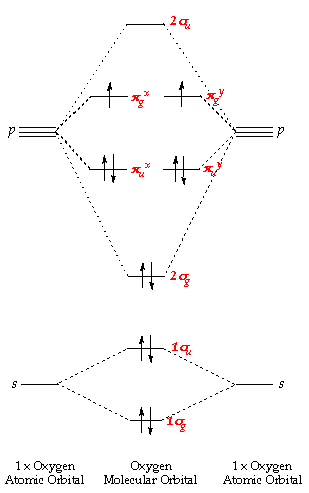
The overall molecular orbital energy level diagram of CO2 is shown in Figure 2-7. The molecular orbital picture of other linear triatomic species, such. As for ethylene and π-allyl, the 2p orbitals of the carbon atoms in the chain may interact in a variety of ways, with the lowest energy π molecular...
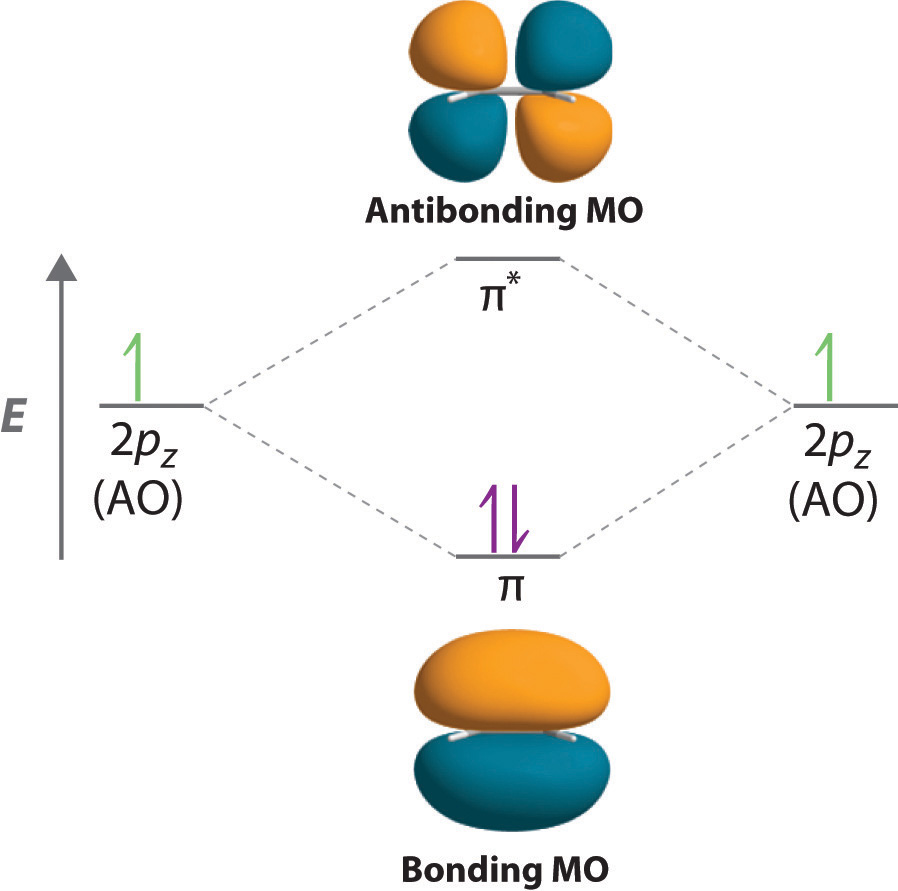
• Molecule orbital theory (Robert Mullikan). • Electrons are delocalised - Different to Lewis and hybridisation (these are not MO). • Energy level diagram represents this interaction. - Two s orbitals interaction to create a low energy bonding and high energy anti-bonding molecular orbital.
(Pi)Molecular orbital diagram of ethylene molecule, Molecular orbital diagrams...
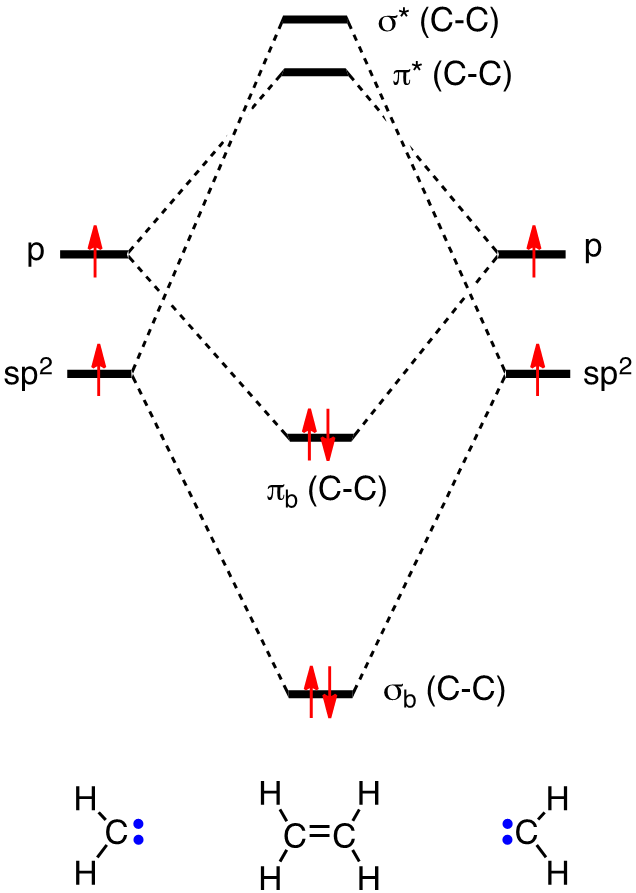
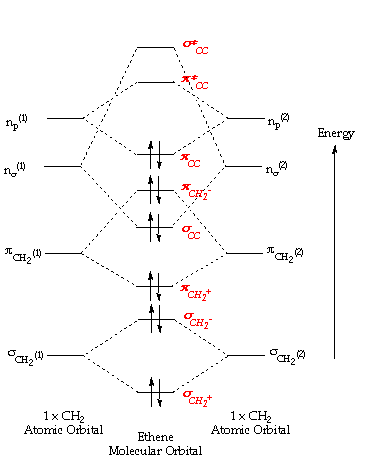
• Standard bonding picture for ethylene is viewed as being made from 2 sp2 hybridized C atoms, and consists of a C-C double bond. •. MOT does not employ hybridization and does not assume bonding arrangements. •. Build ethylene from two CH2 groups without preconceived bonding arrangements. •
The filled molecular orbital diagram shows the number of electrons in both bonding and antibonding molecular orbitals. Figure 10. The molecular orbital energy diagram predicts that He2 will not be a stable molecule, since it has equal numbers of bonding and antibonding electrons.
Molecular orbital theory was put forward by Hund and Mullikan in 1932. This theory is modern and more rational. This theory assume that in molecules, atomic orbitals lose their identity and the electrons in molecules are present in new orbitals called molecular orbitals.
Molecular Orbital Tutorial. Barry Linkletter Department of Chemistry, University of Prince Edward Island Abstract This tutorial examines a method for constructing hybrid orbitals. Page 30 of 35. Fig. 31: Molecular orbital diagram for ethylene.
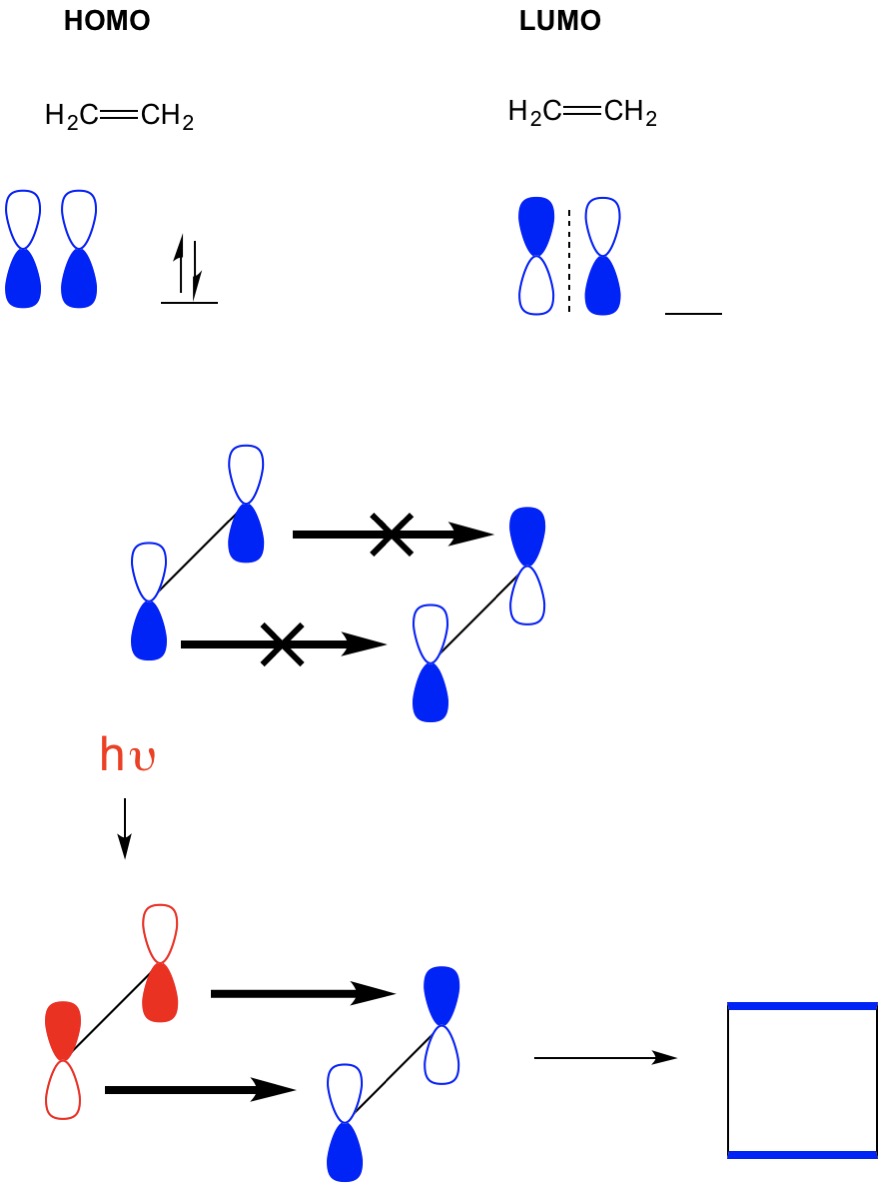
Chad explains how to draw the Bonding and Antibonding Pi Molecular Orbitals for 1,3,5-hexatriene identifying both the HOMO and LUMO. It should also be pointed out that these vertical nodes are always arranged symmetrically in the molecular orbital diagrams.
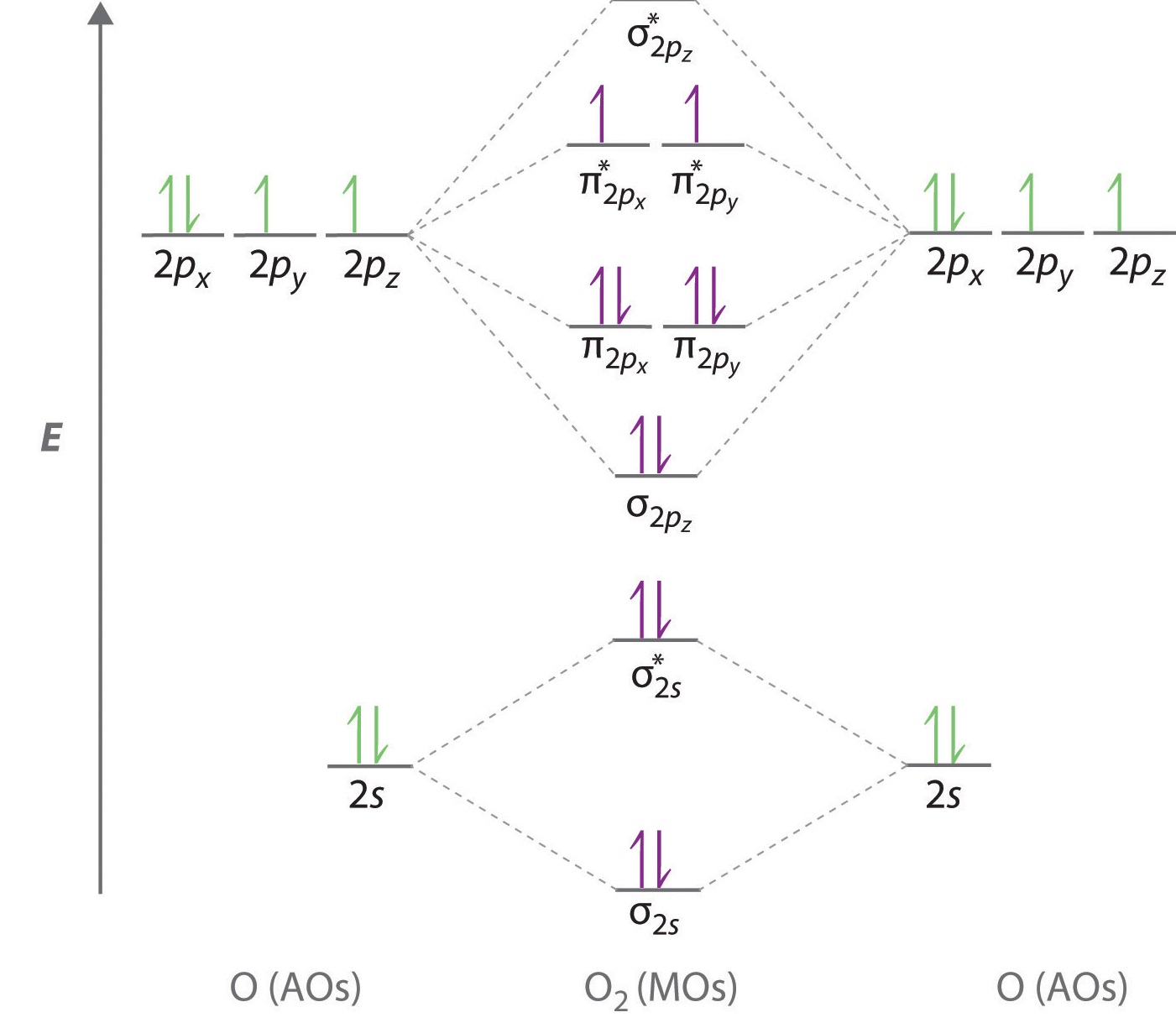
Assuming the molecular orbital diagram in Figure 9.16 applies to BrO, write its electron configuration (where Br uses $4 s$ and $4 p$ orbitals). What is the highest occupied molecular orbital (HOMO) for the molecule? Check back soon!
Hello! Just finished a lecture in my ochem class and was going through the notes and had some questions if yall don't mind. I am already watching some youtube videos on molecular diagram and orbitals because I have no clue whats going on there but if you guys could please help out that would be great! * **First starting off with conjugation and energy, here is a pic: https://imgur.com/lmg6PDn** Does higher energy = less stable? So the first one monoene has a higher energy than the two dienes c...
Chapter 2 - Molecular Orbital Theory Big-picture: Now that we understand aspects of molecular structure, we can look in more detail at bonding Learning goals: • Be able to construct molecular orbital diagrams for homonuclear diatomic, heteronuclear diatomic, homonuclear triatomic, and...
Valence bond (VB) theory gave us a qualitative picture of chemical bonding, which was useful for predicting the shapes of molecules, bond strengths, etc. It fails to describe some bonding situations accurately because it ignores the wave nature of the electrons.
A bare molecular orbital diagram is presented and you must drag the correct orbitals and labels onto the diagram. The diagram is then completed by filling the energy levels with the correct number of electrons. The following molecules are currently available: Molecules of the First Row
Molecular orbital (MO) theory describes the behavior of electrons in a molecule in terms of combinations of the atomic wavefunctions. Application: Computational Chemistry in Drug Design. Molecular Orbital Energy Diagrams. Bond Order. Bonding in Diatomic Molecules.
The molecular orbital model is by far the most productive of the various models of chemical bonding, and serves as the basis for most quantiative Construct a "molecular orbital diagram" of the kind shown in this lesson for a simple diatomic molecule, and indicate whether the molecule or its positive...
Hello! Just finished a lecture in my ochem class and was going through the notes and had some questions if yall don't mind. I am already watching some youtube videos on molecular diagram and orbitals because I have no clue whats going on there but if you guys could please help out that would be great! * **First starting off with conjugation and energy, here is a pic: https://imgur.com/lmg6PDn** Does higher energy = less stable? So the first one monoene has a higher energy than the two dienes c...
Molecular Orbital Theory: Detailed information on the molecular orbital theory class 11 and more in this article above. Postulates Of Molecular Orbital theory. Just as electrons of an atom are present in various atomic orbitals, electrons of a molecule In terms of energy level diagram, it represents as.
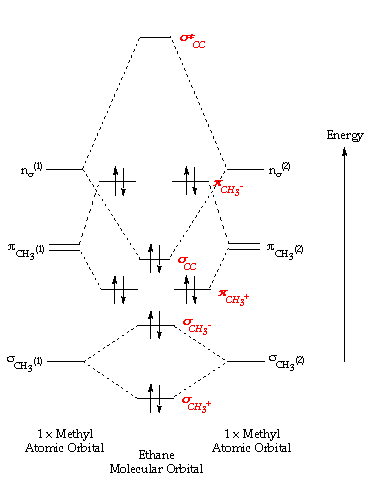
Notes on molecular orbital calculations. First printing, 1961 Second printing, with stretching vibrations of ethylene in the infrared a r e different f r o m those of acetylene and ethane. IN THE APPLICATION of molecular orbital theory to calculations of chemical binding energies, we...
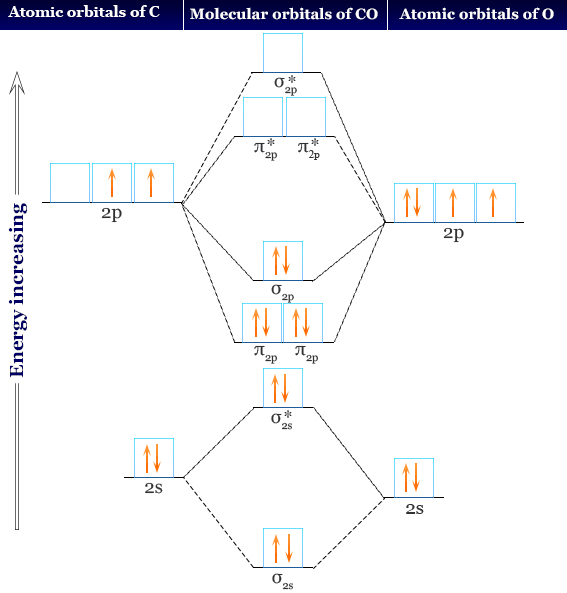
The molecular orbital diagram representing this order of energy levels is shown in fig. No. 9 Molecular Orbital Diagram for CO. Analysis done by Bond Order. If value of bond order is positive, it indicates a stable molecule and if the value is negative or zero, it means that the molecule is unstable.
A molecular orbital diagram, or MO diagram, is a qualitative descriptive tool explaining chemical bonding in molecules in terms of molecular orbital theory in general and the linear combination of atomic orbitals (LCAO) method in particular. A fundamental principle of these theories is that as atoms.
The molecular orbital theory is a concept of quantum mechanics where atomic linearly combines to form molecular orbitals and we describe the wave nature of atomic particles. The above diagram shows the Molecular Orbital(MO) diagram of ethene/ethylene.
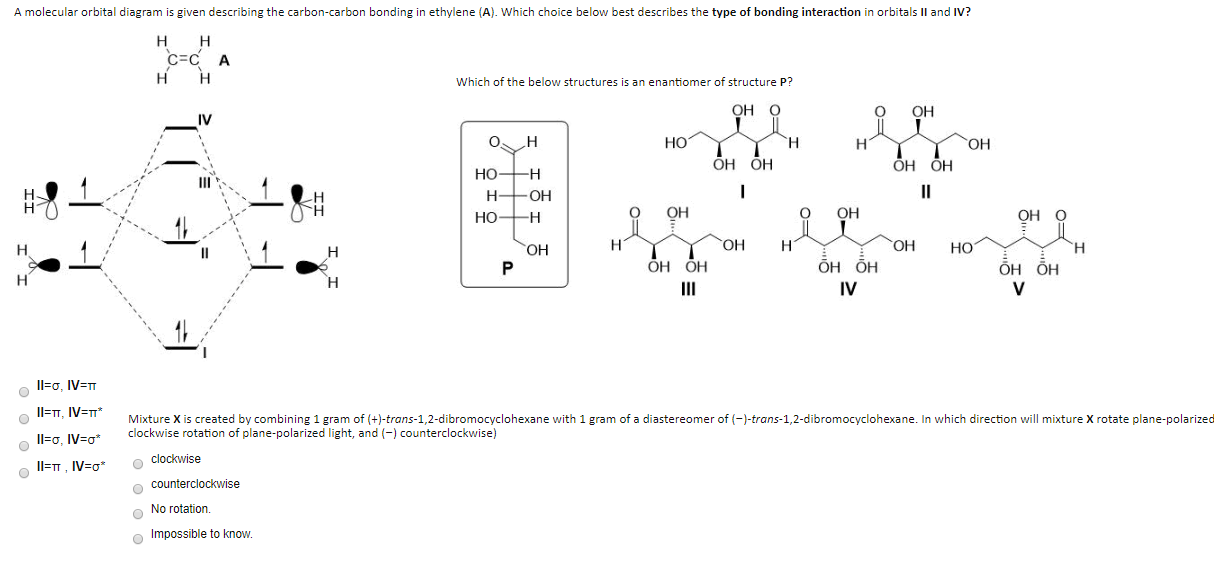
Which molecular orbital of ethylene (tt or tt ) is the most impor 1 tant one to look at in a reaction in which ethylene is attacked by an electrophile J Refer to the molecular orbital diagrams of allyl cation (Figure 10 13) and those presented earlier in this chapter for ethylene and 1 3 butadiene...
Molecular Orbital Theory. Bonding and Antibonding Molecular Orbitals. In molecular orbital theory, bond order is also defined as the difference, divided by two, between the number of bonding and antibonding electrons; this often, but not always, yields the same result.
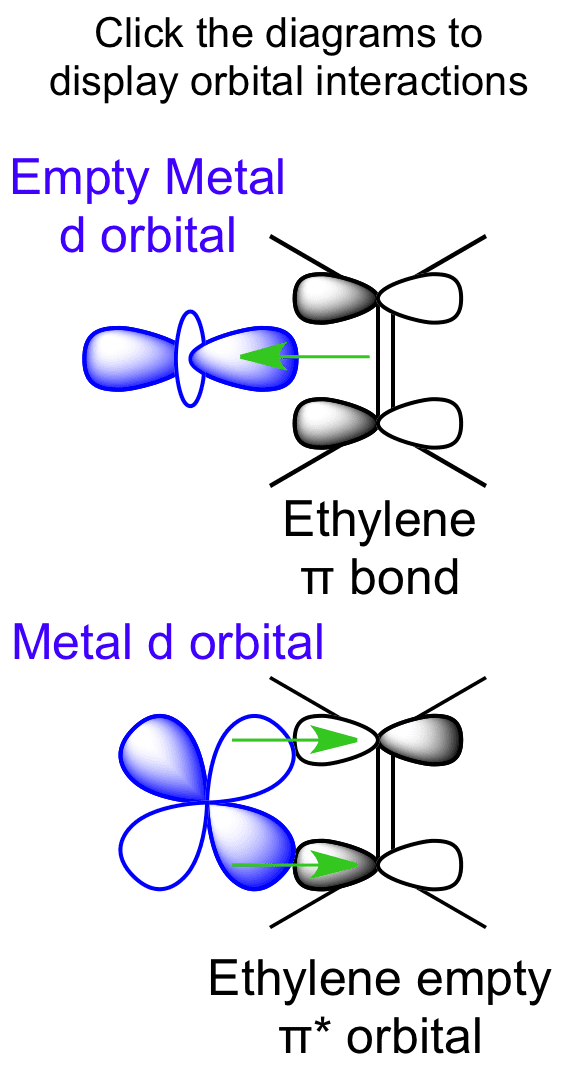
Ethylene is capable of acting as a ligand as the C=C π bond can donate electron density to an empty metal d orbital, forming a σ bond. View Ethylene Molecular Orbitals here. Explore Metal-Ligand bonding with other molecules.
element. Accordingly, a molecular orbital diagram such as Figure 9-5 is inappropriate for heteronuclear diatomic molecules. If the two elements are similar (as in NO or CN mole-cules, for example), we can modify the diagram of Figure 9-5 by skewing it slightly.














0 Response to "40 ethylene molecular orbital diagram"
Post a Comment