41 hcn molecular orbital diagram
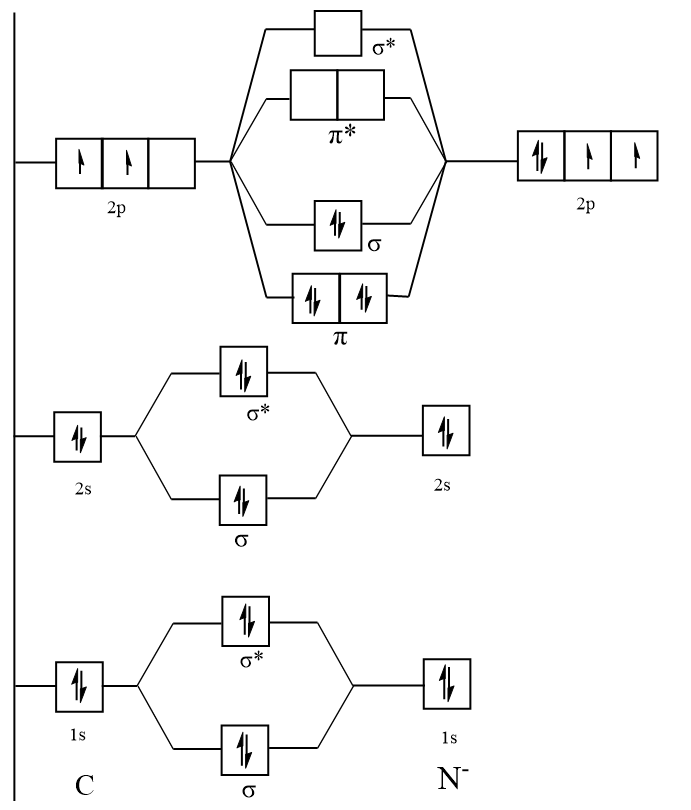
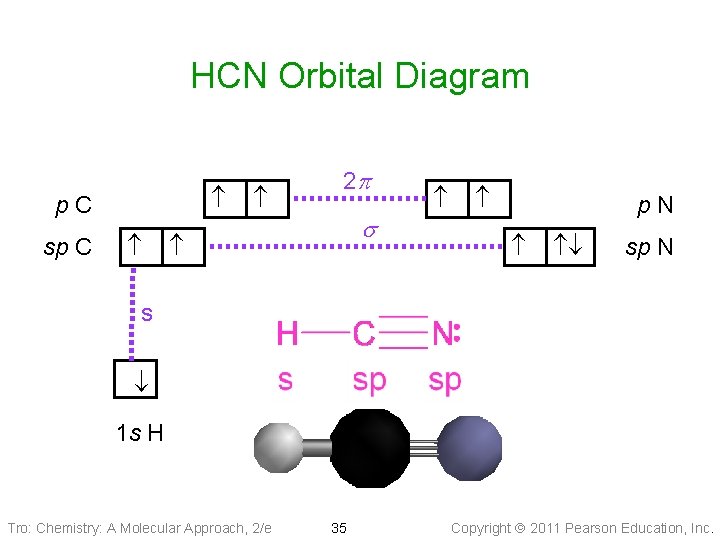
HCN MO (Molecular Orbital) Diagram What is an MO Diagram? MO diagram is nothing but a description of how the chemical bonds are formed in any compound. The diagram is a representation of different energy levels and why a compound exists in nature or why some compounds don’t exist at all.
HCN Molecular Geometry The molecular Geometry of any given molecule helps understand its three-dimensional structure and the arrangement of atoms in a molecule, and its shape. Hydrogen Cyanide has geometry like AX2 molecule , where A is the central atom and X is the number of atoms bonded with the central atom.
For the molecule hydrogen cyanide (HCN):a) Draw the filled atomic orbitals of the nitrogen atom.b) Which atomic orbitals are used for hybridization and event...
Hcn molecular orbital diagram
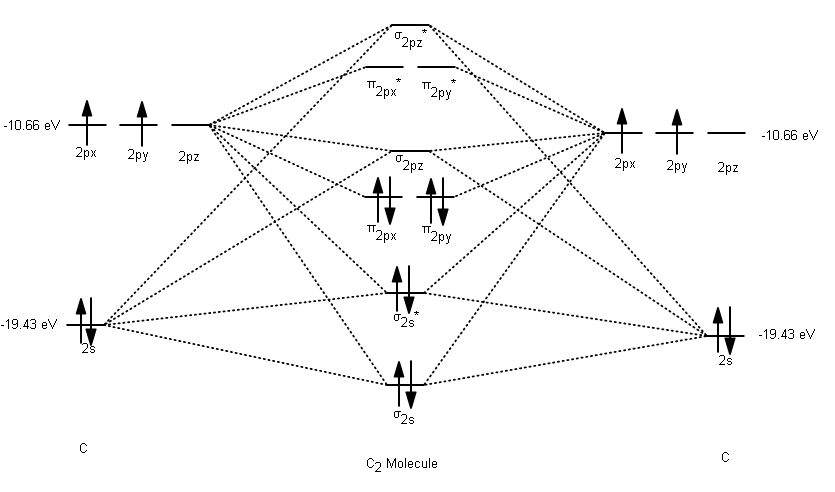
by DC Pan · 1967 · Cited by 50 — Walsh's orbital-energy diagrams are found to be almost independent of ... work on HCN consists of: (a) a molecule-minus-atom.
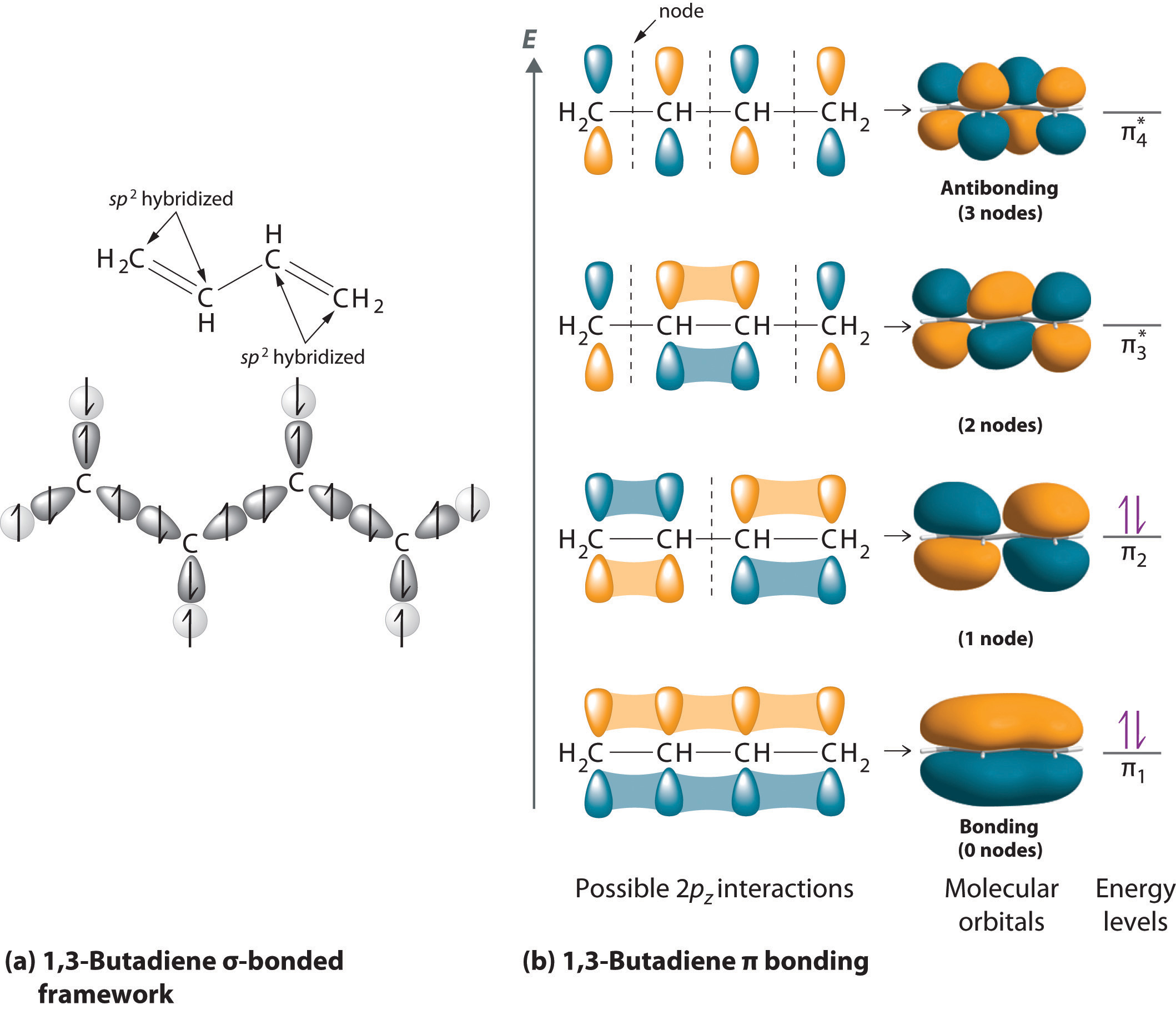
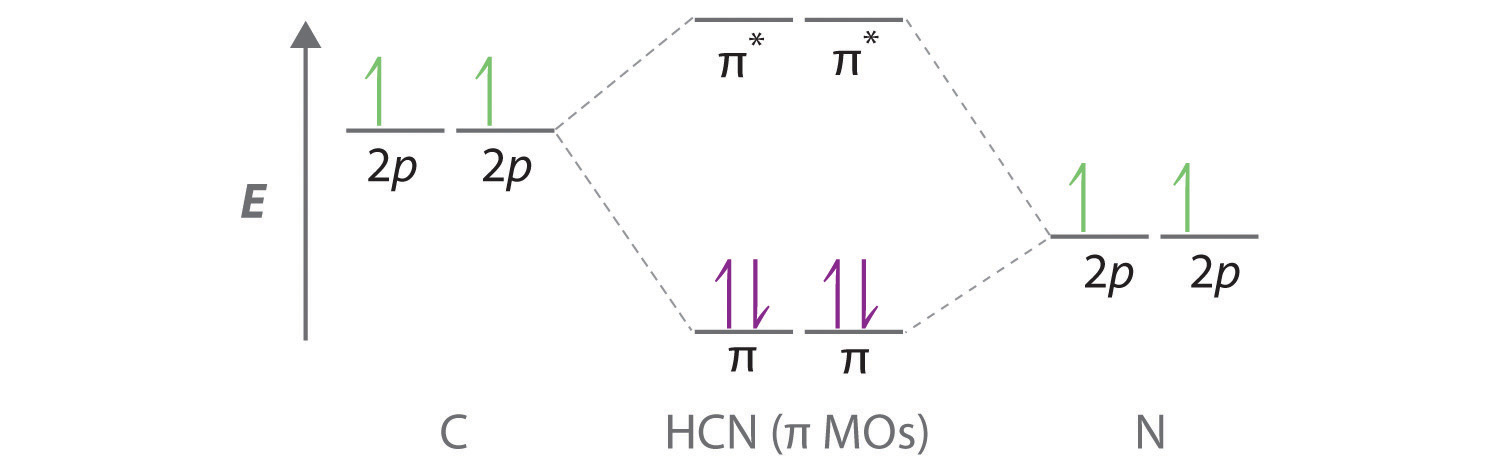
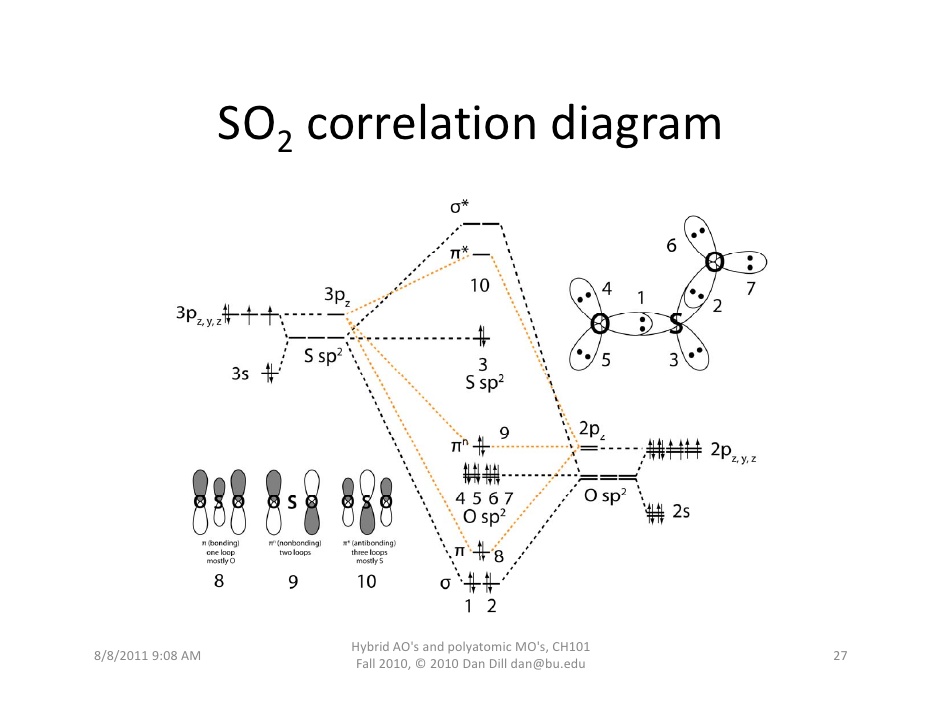
8 May 2021 — Figure 4.11.1: Molecular Orbital Energy-Level Diagram for π ... A Because HCN is a linear molecule, it is likely that the bonding can be ...
Hydrogen cyanide | HCN or CHN | CID 768 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities ...
Hcn molecular orbital diagram.
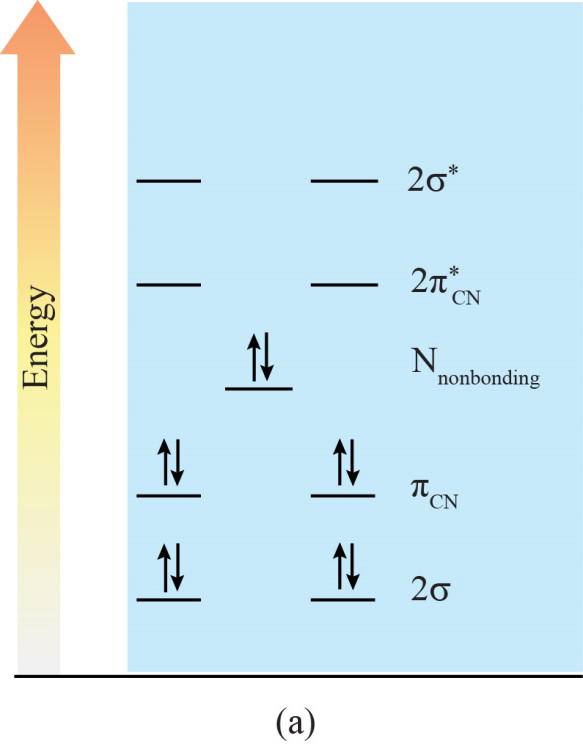
Energy level diagram of HCN (a) and the HCN-water complex (b). Molecular orbitals (MOs) marked in red and black are included in the active spaces for RIXS ...
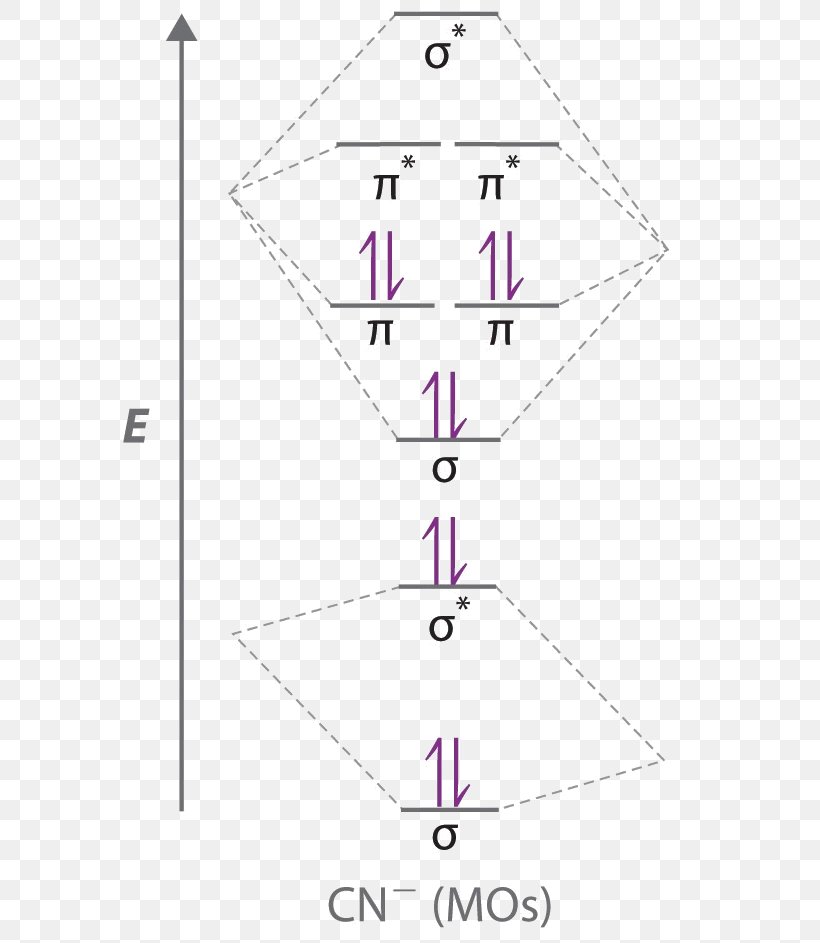
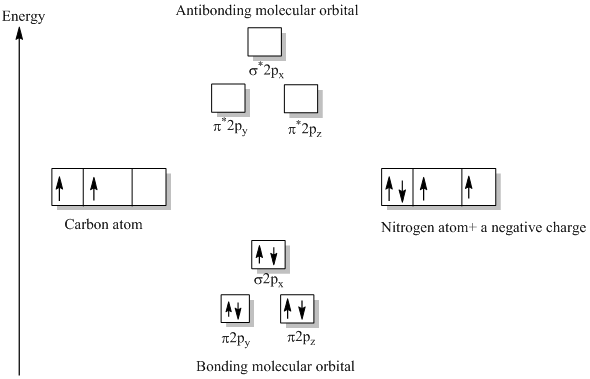
Problem 66QP: Draw the molecular orbital diagram for the cyanide ion (CN−). (Assume the ordering of moleculer orbitals to be like that in N2.) Write the electron configuration of the cyanide ion (CN−). Oct 10, · We see the existence of the paired HOMO's and LUMO's in the molecular orbital energy diagram for HCN, shown below: Next, we move ...
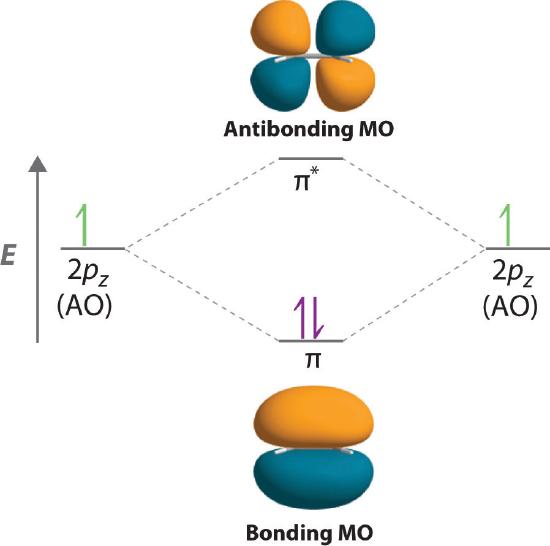
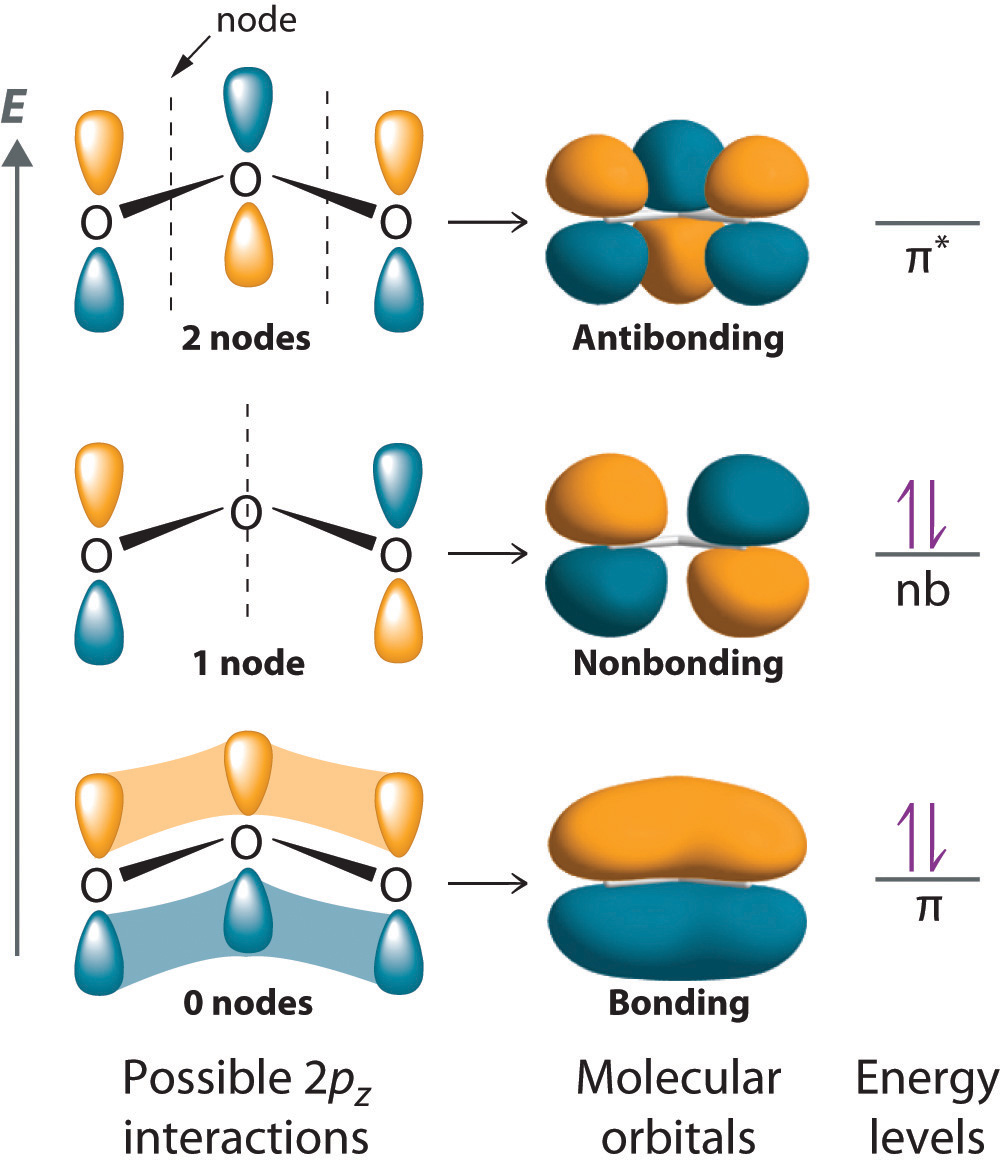
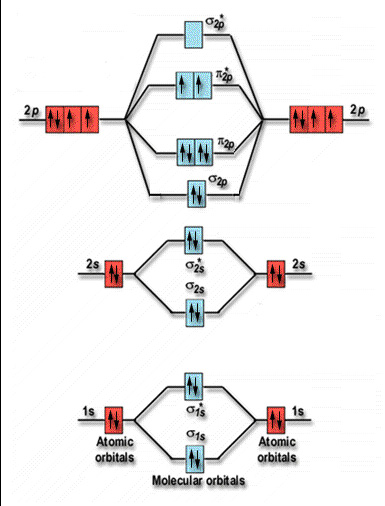
Simple Molecular Orbitals - Sigma and Pi Bonds in Molecules An atomic orbital is located on a single atom. When two (or more) atomic orbitals overlap to make a bond we can change our perspective to include all of the bonded atoms and their overlapping orbitals. Since more than one atom is involved, we refer to these orbitals as molecular orbitals.
In HCN, there are two sigma bonds, C-H and C-N. The number of sigma bonds is equal to the number of hybrid orbitals formed. So the hybridization for HCN molecule is sp hybridization. Concluding Remarks. We can say that HCN, Hydrogen Cyanide, is a linear molecule with sp hybridization to conclude this blog post.
In HCN molecule, the C atom includes sp -hybridized orbital, since it will combine with only two other atoms to form HCN. One of the sp -hybrid orbitals of carbon atom overlaps with the 1 s orbital of H atom, while the other sp-hybrid orabital mixes with one of the nitrogen’s atom’s three atomic p orbitals which were unhybridized.
Molecular Geometry. Hydrogen cyanide has linear molecular geometry with bond angles of 180 degrees. As hydrogen and nitrogen tend to be far from each other, HCN forms a linear shape. It is slightly polar as nitrogen tries to pull the electrons to itself due to its electronegative value.
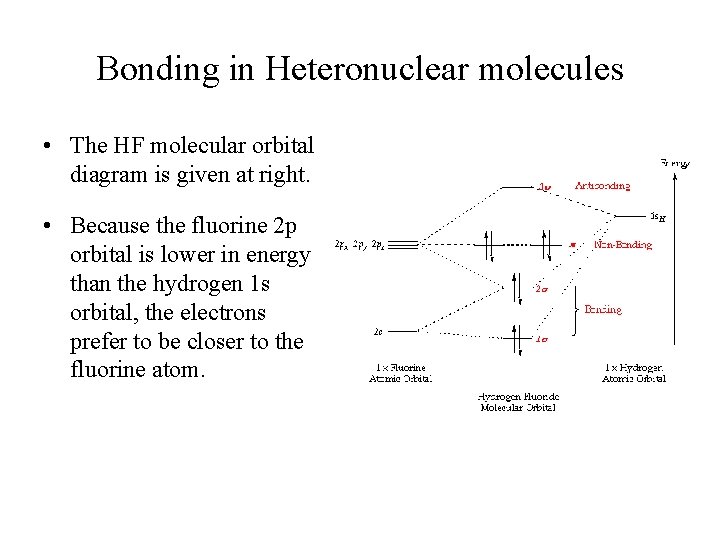
an infamous small molecule known as hydrogen cyanide is shown draw a molecular orbital diagram. then draw MOs of the HCN molecule in the middle. indicate which MO is LUMO AND HOMO The molecular orbital diagram of NO shown in Figure 10.47 also applies to OF .
Sigma (σ) bonding molecular orbital - Shared electron density is directly ... (H-CC-H) as a model to draw an MO diagram for hydrogen cyanide (H-CN) and.13 pages



















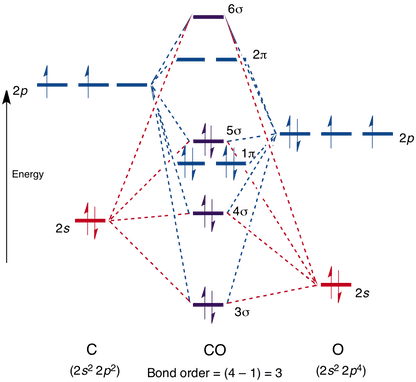
0 Response to "41 hcn molecular orbital diagram"
Post a Comment