40 orbital diagram for arsenic
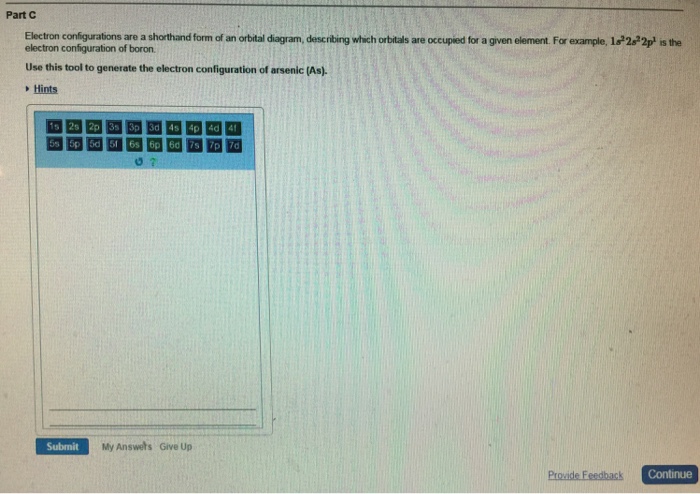
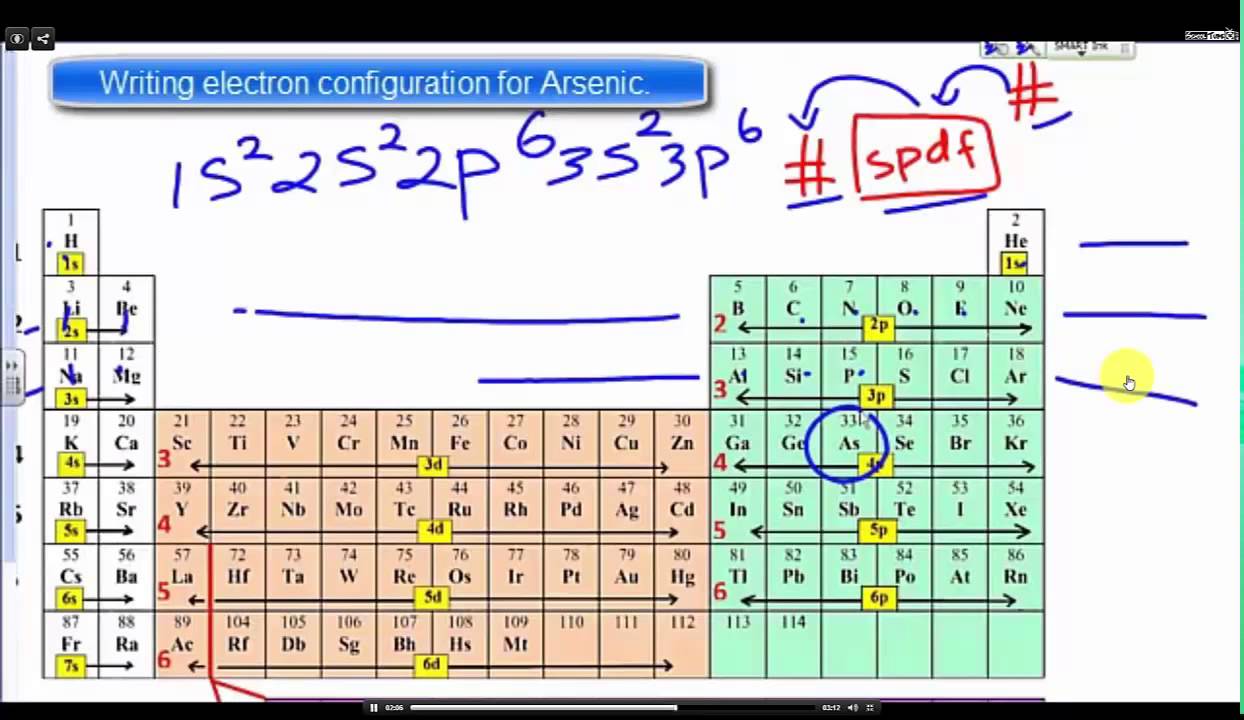
Electron configurations are a shorthand form of an orbital diagram, describing which orbitals are occupied for a given element. For example, 1s2 2s2 2p1 is the electron configuration of boron. Use this tool to generate the electron configuration of arsenic (As). About Press Copyright Contact us Creators Advertise Developers Terms Privacy Policy & Safety How YouTube works Test new features Press Copyright Contact us Creators ...
7. Compare the orbital diagrams for nitrogen (given above), phosphorus and arsenic. What is similar about the electrons in the last sublevel for each of them? They all have 3 electrons in a p sublevel. Information: Electron Configurations vs. Orbital Diagrams The electron orbital diagram of an atom can be abbreviated by using what is called ...
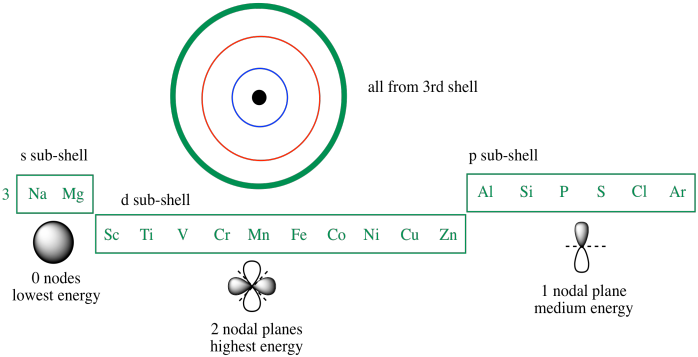
Orbital diagram for arsenic
The orbital notation of arsenic (As) is 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p3See the Related Questions for the electron configuration of all the elements. What is the orbital diagram for bromine? The... Orbital diagram of Arsenic (As) 34: Orbital diagram of Selenium (Se) 35: Orbital diagram of Bromine (Br) 36: Orbital diagram of Krypton (Kr) 37: Orbital diagram of Rubidium (Rb) 38: Orbital diagram of Strontium (Sr) 39: Orbital diagram of Yttrium (Y) 40: Orbital diagram of Zirconium (Zr) 41: What is the orbital diagram of arsenic? The orbital diagram of arsenic can be written as 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p3. Arsenic has 33 electrons, including 3 in its outermost shell. What is the...
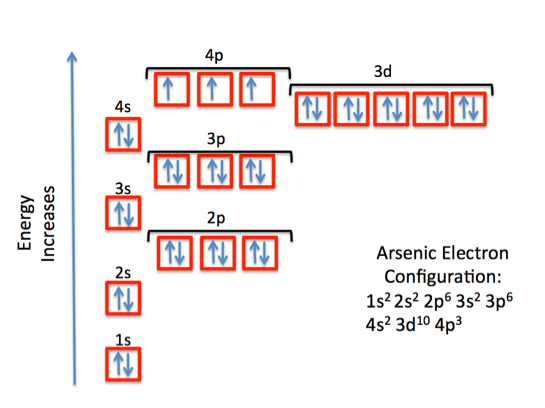
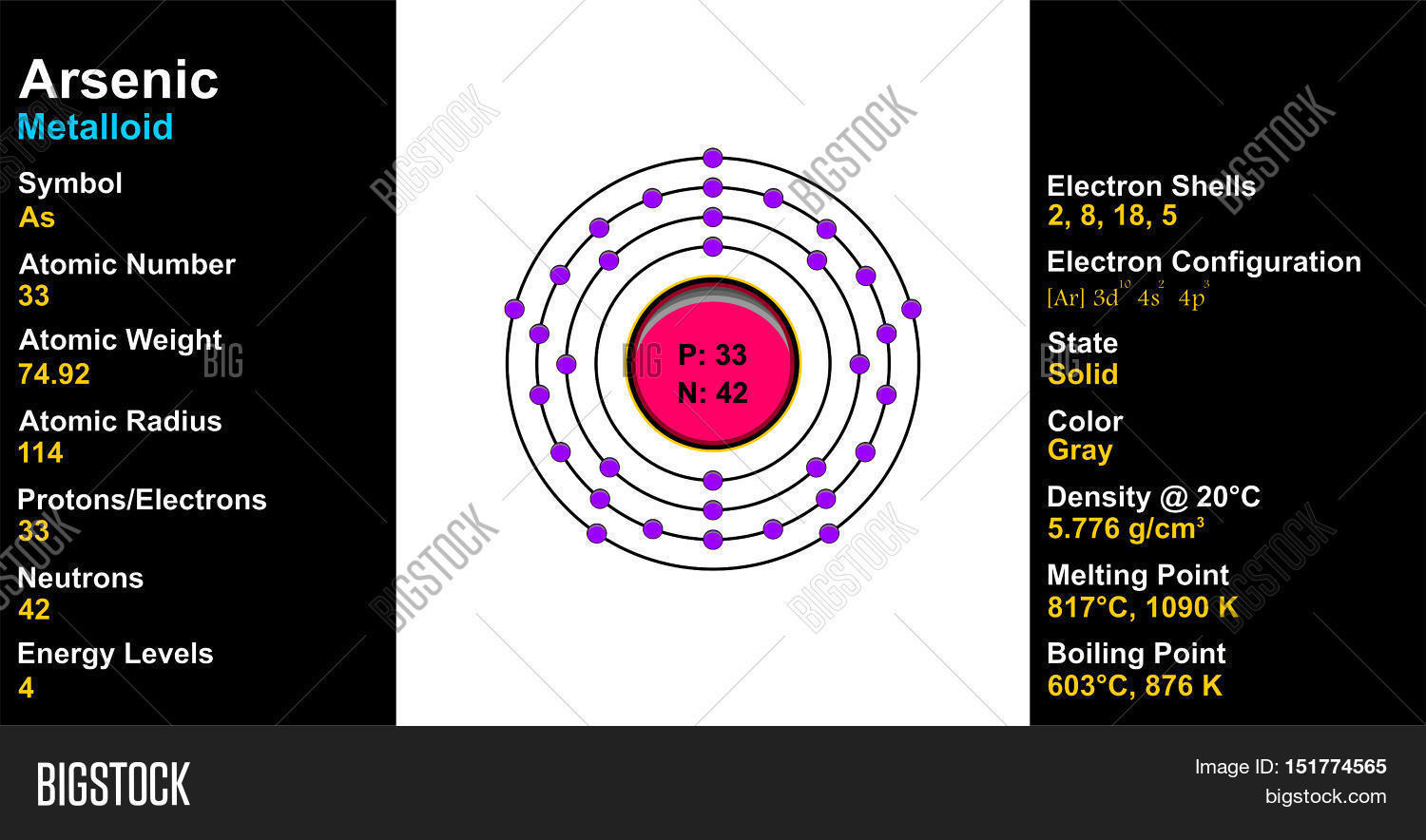
Orbital diagram for arsenic. What is the orbital filling diagram for arsenic? Electrons & Oxidation. Oxidation States: Electrons Per Shell: 2 8 18 5: Electron Configuration [Ar] 3d10 4s2 4p3: 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p3: Orbital Diagram 1s ↿⇂ 2s ↿⇂ 2p ↿⇂ ↿⇂ ↿⇂ 3s ↿⇂ 3p ↿⇂ ↿⇂ ↿⇂ 3d ↿⇂ ↿⇂ ↿⇂ ↿⇂ ↿⇂ 4s ↿⇂ 4p ... How many energy levels does arsenic have? What is the orbital diagram for phosphorus? The p orbital can hold up to six electrons. We'll put six in the 2p orbital and then put the next two electrons in the 3s. Since the 3s if now full we'll move to the 3p where we'll place the remaining three electrons. Sep 26, 2019 · Orbital Diagram For Arsenic A Arsenic Atom Diagram. Orbital Diagram For Arsenic Electron Orbitals And Orbital Filling Diagrams Ppt Download. Orbital Diagram For Arsenic 64 Electronic Structure Of Atoms Electron Configurations Chemistry. Orbital Diagram For Arsenic Ecotoxicology Of Arsenic In The Hydrosphere Implications For Public. Arsenic is a carcinogen, associated with lung cancer when inhaled. It is mainly produced as a by-product of refining certain sulphide ores. It is naturally occurring in many household products. The electronic configuration of arsenic is 1s2,2s2,2p6,3s2,3p6,4s2,3d10,4p3. Electrons per Energy Level : 2,8,18,5
Electronic configuration of the Arsenic atom in ascending order of orbital energies: 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 3. Electronic configuration of the Arsenic atom in ascending order of the levels: 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 3. Reduced electronic configuration As: [Ar] 3d 10 4s 2 4p 3. Below is the electronic diagram of the Arsenic atom Distribution of electrons over energy levels in the As atom. Arsenic. Full electron configuration of arsenic: 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p3. germanium ← arsenic → selenium. Arsenic Electron Configuration (As) with Orbital Diagram. Arsenic Electron Configuration: Chemical element, Arsenic has atomic number 33 and has the symbol “As”. Arsenic chemical occurs in many minerals like elements with a combination of Sulphur and metals. This is the pure elemental crystal. Arsenic comes in a metalloid category. The orbital diagram for a ground-state nitrogen atom is. 13. The orbital diagram for a ground-state oxygen atom is. 14. The orbital diagram for a ground state carbon atom is ... arsenic D) vanadium E) none of these. 18. How many unpaired electrons does a ground-state atom of sulfur have? A) 0 B) 1 C) 2 D) 3 E) 4. 19. A ground-state atom of ...
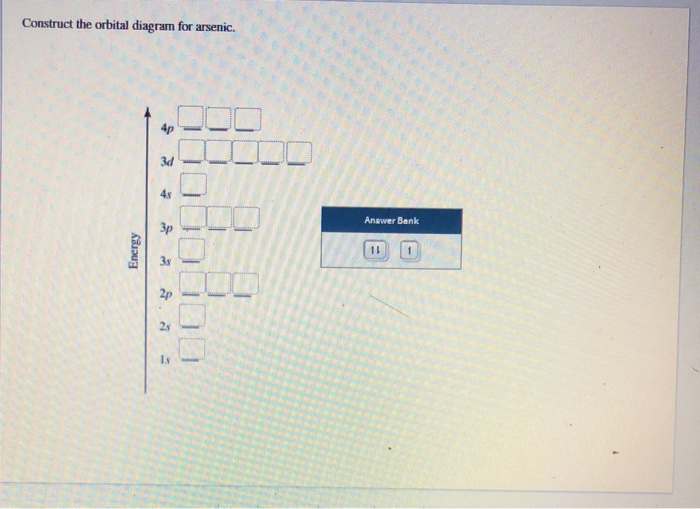
Electron orbital diagrams and written configurations tell you which orbitals are filled and which are partially filled for any atom. The number of valence electrons impacts on their chemical properties, and the specific ordering and properties of the orbitals are important in physics, so many students have to get to grips with the basics. Experts are tested by Chegg as specialists in their subject area. We review their content and use your feedback to keep the quality high. 90% (29 ratings) Transcribed image text: Construct the orbital diagram for arsenic. Answer Bank Energy. The orbital diagram of arsenic can be written as 1s2 2s2 2p6 3s23p6 4s2 3d10 4p3. Arsenic has 33 electrons, including 3 in itsoutermost shell. schematron.org! Arsenic atomic orbital and chemical bonding information. There are also tutorials on the first thirty-six elements of the periodic table. So the electron configuration of potassium will involve 19 electrons. The full electron configuration of potassium is "1s"^2"2s"^2"2p"^6"3s"^2"3p"^6"4s"^1". The noble gas notation is "[Ar]4s"^1". The following orbital diagram shows the increase in energy from one energy sublevel to the next, but you can write them on the same level horizontally,
Feb 08, 2018 · What is the correct orbital diagram for arsenic? Electrons & Oxidation. Oxidation States. ±3,+5. Electrons Per Shell. 2 8 18 5. Electron Configuration. [Ar] 3d10 4s2 4p3.
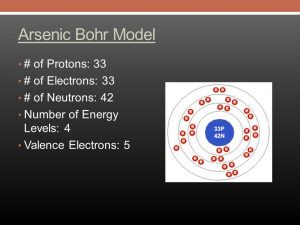
Arsenic, As, has atomic number 33, which is the number of protons in the nuclei of its atoms. A neutral As atom would also have 33 electrons. The electron configuration of a neutral arsenic atom is [Ar]3d104s24p3 . What is the correct orbital diagram for arsenic? Electrons & Oxidation
Draw the orbital diagram for arsenic. Arsenic Poisoning: Arsenic has been used throughout history as a poison to kill people, often by stirring it into food. It causes a slow, painful death by ...
Electron configurations are a shorthand form of an orbital diagram, describing which orbitals are occupied for a given element. For example, 1s22s22p1 is the electron configuration of boron. Use this tool to generate the electron configuration of arsenic (As).Draw an orbital diagram for scandium ScDraw an orbital diagram for scandium (Sc ...
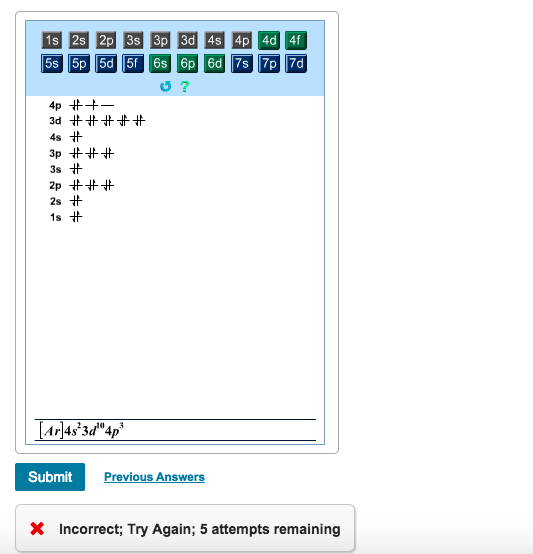
Know the answer? Add Answer to: Construct the orbital diagram for arsenic. பட்டம் Answer Bank Energy. Your Answer: Post as a guest.1 answer · 0 votes: AS CABeni ato ric Unber: 33 ২ electn Carbiguration: up Cmstruct ovsita Diaran: us 3P 35 IS
The orbital diagram of arsenic can be written as 1s2 2s2 2p6 3s23p6 4s2 3d10 4p3. There are also tutorials on the first thirty-six elements of the periodic table. Arsenic 1s2 2s2 2p6 3s2 3p6 … What four features of bacteria that enable them to survive in a wide variety of habitats? Why don't libraries smell like bookstores?
90.96043 (97)#. 92 As. 92. 91.96680 (97)#. Mass Number. The sum of the number of protons and neutrons of an atomic nucleus. In other words, it's the sum of the number of nucleons in an atom. Relative Atomic Mass. The ratio of the average mass per atom of an isotope to 1/12 the mass of a carbon-12 atom.
By using the Aufbau diagram (shown below) we can determine the full electron configuration for a neutral arsenic atom. 1s22s22p63s23p63d104s24p3 The shorthand electron configuration, which uses the symbol for the noble gas in the previous period, in this case argon. [Ar]3d104s24p3 Aufbau Diagram Answer link
Orbital Diagram and Symbol # e- Longhand Electron Configuration 1. 4. Mg 35 2 ... E xcited state electron configuration shows an electron becoming excited and jumping into a higher orbital. 10. ... Complete the orbital diagram for arsenic.. AsF5 Molecular Geometry and Bond Angles (Arsenic . Nov 05, 2020 · sp ...
Is this the correct atomic orbital diagram for a neutral atom of arsenic? Please indicate "true" or "false" accordingly on your Google quiz form.
B) f orbital Basically, when l = 0 the is one orbital and it is called an s-orbital. It can hold 2 electrons total. When l = 1, the orbitals are called p-orbitals and there are 3 of them. Each of the individual p-orbitals can hold 2 electrons each. This gives us a total of 6 electrons that can go into the 3 p-orbitals.
The orbital diagram of arsenic can be written as 1s2 2s2 2p6 3s23p6 4s2 3d10 4p3. Arsenic has 33 electrons, including 3 in itsoutermost shell. schematron.org! Arsenic atomic orbital and chemical bonding information. There are also tutorials on the first thirty-six elements of the periodic table.
What is the orbital diagram of arsenic? The orbital diagram of arsenic can be written as 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p3. Arsenic has 33 electrons, including 3 in its outermost shell. What is the...
Orbital diagram of Arsenic (As) 34: Orbital diagram of Selenium (Se) 35: Orbital diagram of Bromine (Br) 36: Orbital diagram of Krypton (Kr) 37: Orbital diagram of Rubidium (Rb) 38: Orbital diagram of Strontium (Sr) 39: Orbital diagram of Yttrium (Y) 40: Orbital diagram of Zirconium (Zr) 41:
The orbital notation of arsenic (As) is 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p3See the Related Questions for the electron configuration of all the elements. What is the orbital diagram for bromine? The...
















0 Response to "40 orbital diagram for arsenic"
Post a Comment