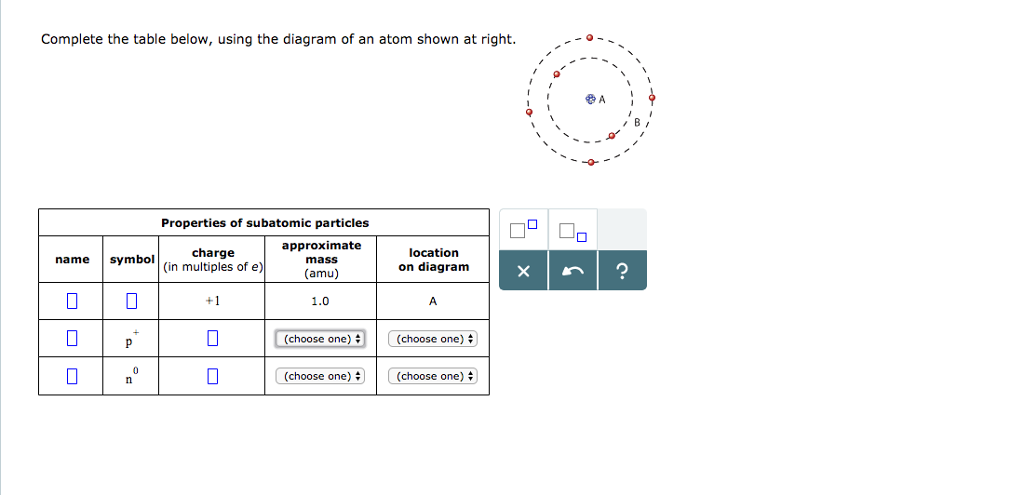
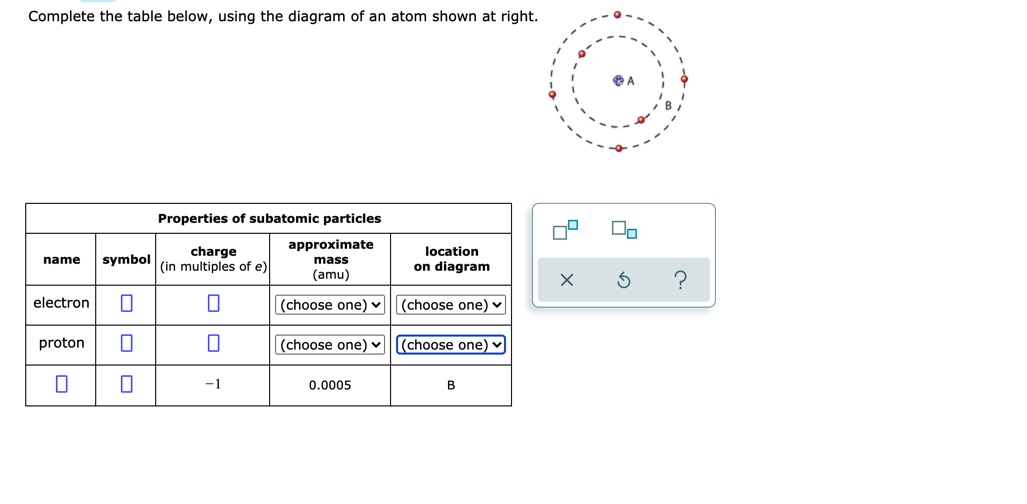
37 complete the table below using the diagram of an atom shown at right
Generalized energy-level diagram for atomic orbitals in an atom with two or more electrons (not to scale). Electrons in successive atoms on the periodic table tend to fill low-energy orbitals first. Thus, many students find it confusing that, for example, the 5 p orbitals fill immediately after the 4 d , and immediately before the 6 s . 4. Atoms tend to react in ways that give each atom a stable outer shell of electrons. true correct 5. Atoms with an outer shell that is almost empty are located on the right side of the Periodic Table while atoms with an outer shell that is full or almost full are located on the left side of the Periodic Table. false correct 6.
Base your answers to questions 16 through 18 on the information below and on your knowledge of chemistry. The Bohr model of the atom was developed in the early part of the twentieth century. A diagram of the Bohr model for one atom, in the ground state, of a specific element, is shown below. The nucleus of this atom contains 4 protons and 5 ...
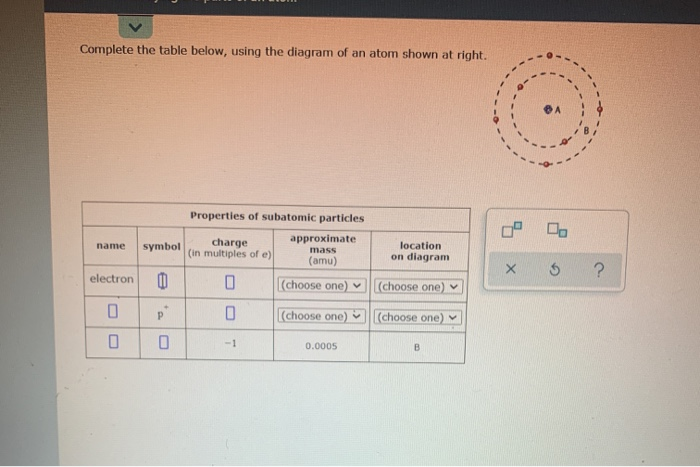
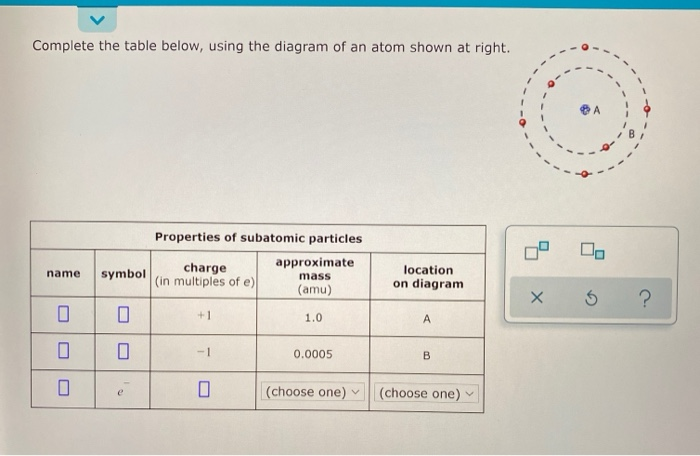
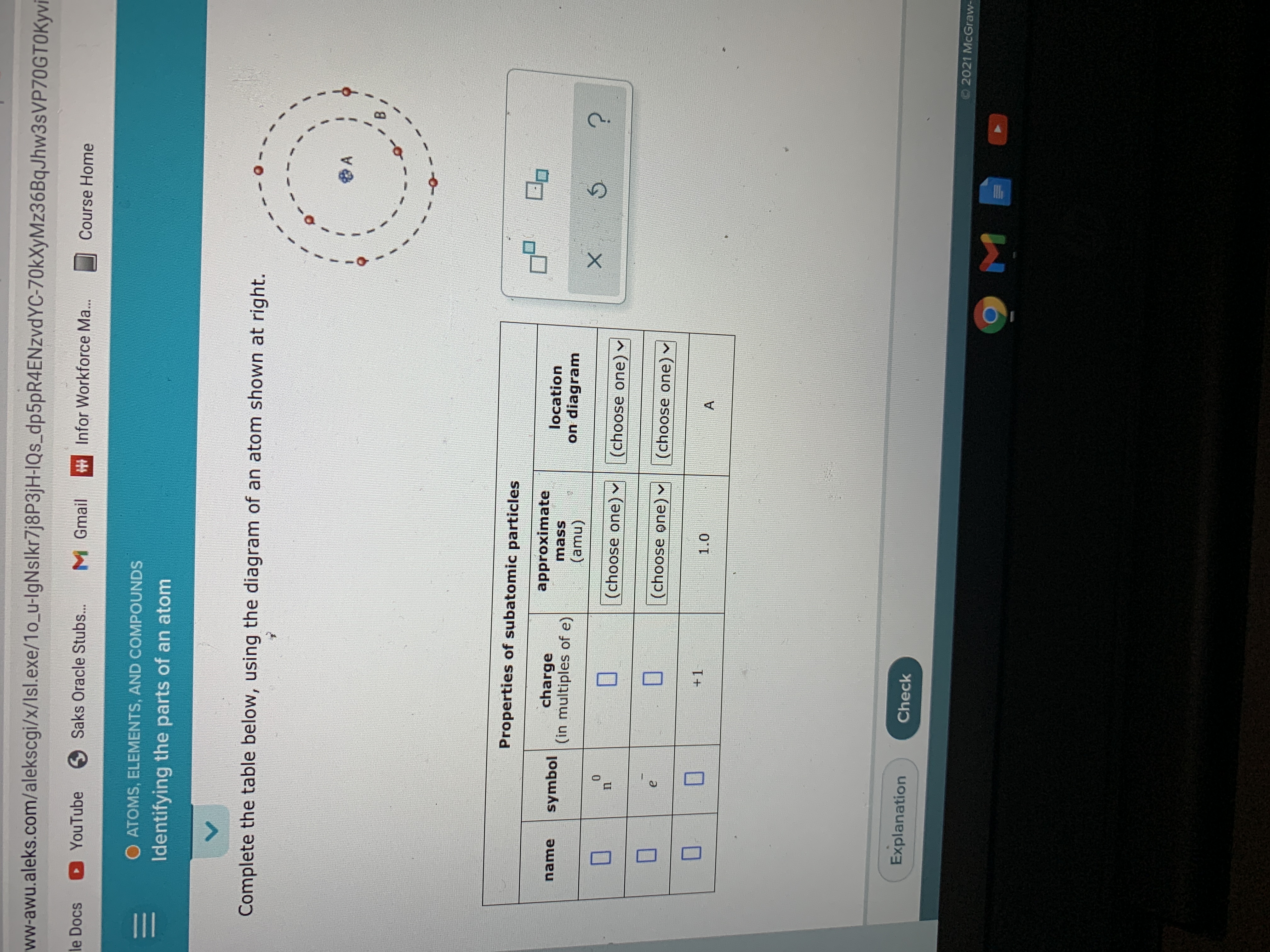
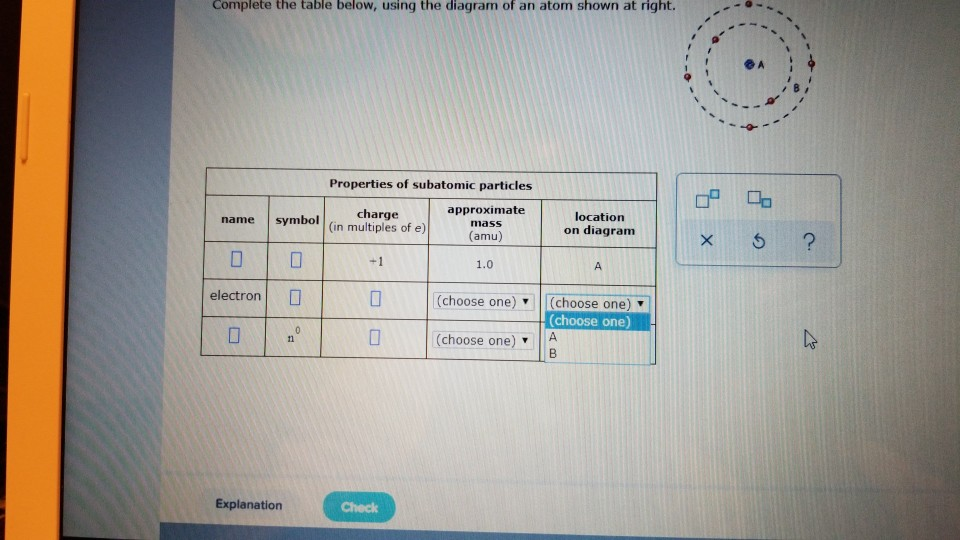
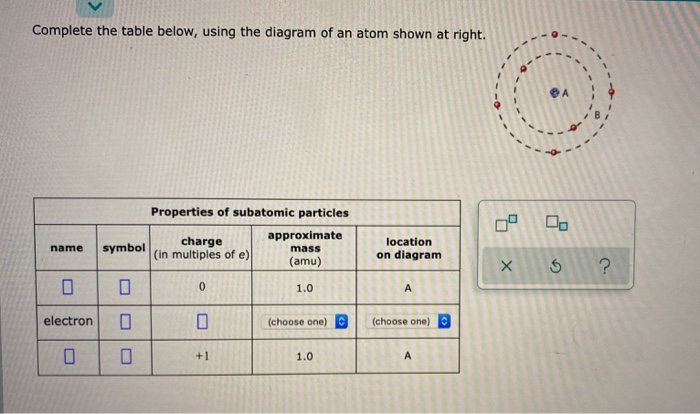
Complete the table below using the diagram of an atom shown at right
We compare the bonding of the atom in the molecule to the valence electron structure If the atom has one more electron in the molecule, it is shown with a "-" charge If the atom has one less electron, it is shown with a "+" charge Neutral molecules with both a "+" and a "-" are dipolar Oct 17, 2021 · Complete the table below, using the diagram of an atom shown at right. A name Properties of subatomic particles Aharge approximate symbol mass (in multiples of e) (amu) name location on diagram X5 ? O 1.0 А 0 1.0 А proton (choose one) (choose one) Complete the table below, using the diagram of an atom shown at right. Question: Complete the table below, using the diagram of an atom shown at right. This problem has been solved! See the answer ...
Complete the table below using the diagram of an atom shown at right. Use your knowledge of atomic structure to label this diagram of a nitrogen atom. The boxes below are labeled with the approximate atomic masses of four different elements. Using the provided atomic masses and the periodic table shown, drag the appropriate subatomic particles into the boxes to create electrically neutral atoms. Transcribed image text: Complete the table below, using the diagram of an atom shown at right. Properties of subatomic particles approximate charge symbol mass in multiples of Camu) Da name location on diagram х р (choose one (choose one 3 +1 1.0 neutron 0 choose one) Choose one cºuld location on diagram name symbol Х 2 (choose one) P Properties of subatomic particles charge approximate ... Complete the table below, using the diagram of an atom shown at right. Properties of subatomic particles name charge symbol (in multiples of e) approximate mass (amu) location diagram 1.0 electron O (choose one) Complete the table below, using the diagram of an atom shown at right. Properties of subatomic... Question: Complete the table below, using the diagram of an atom shown at right. This problem has been solved! See the answer ...
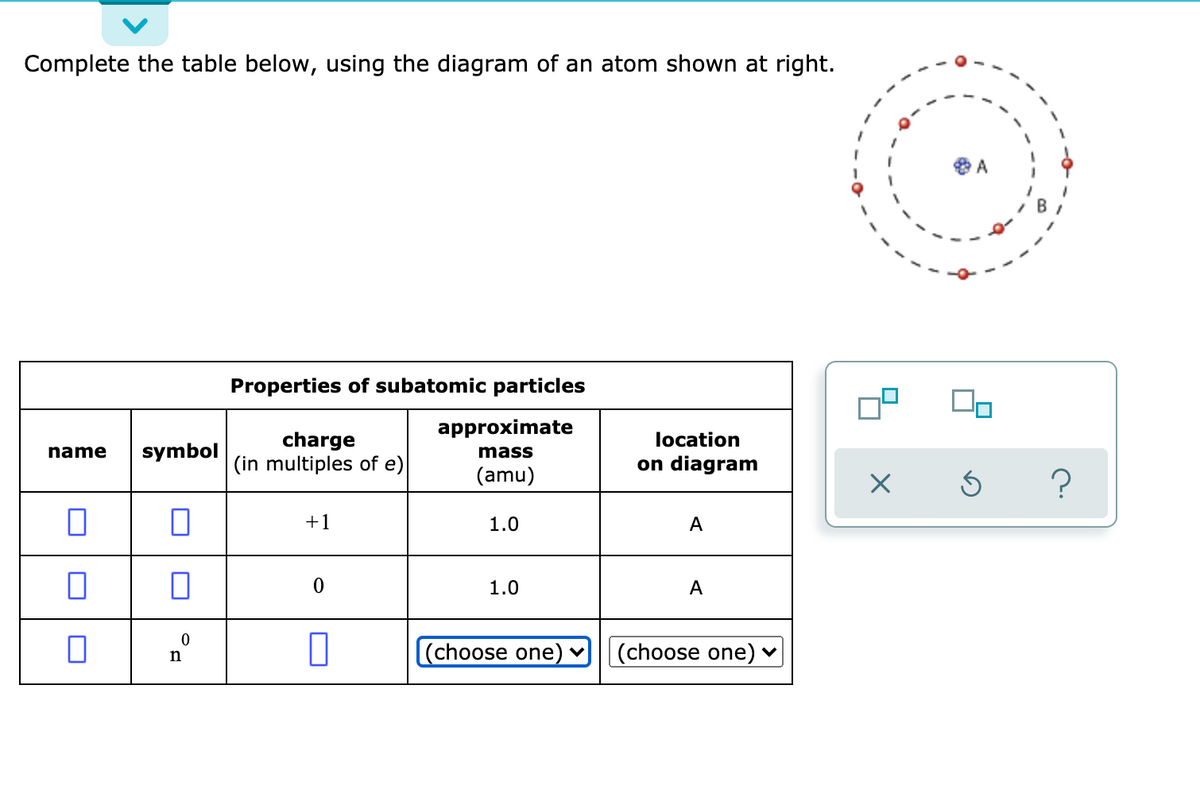
Complete the table below, using the diagram of an atom shown at right. Properties of subatomic particles approximate charge |(in multiples of e) location name symbol mass on diagram (amu) ? +1 1.0 A p (choose one) (choose one) Transcribed image text: Complete the table below, using the diagram of an atom shown at right. Properties of subatomic particles approximate charge symbol ... Basic Diagram of an Atom. Most of an atom is just empty space and consists of a positively charged nucleus of protons and neutrons surrounded by a cloud of negatively charged electrons. The center of an atom is the nucleus and one or more electrons surrounding the nucleus. When one says an atom is electrically neutral, it means that the number ... Solve the given problem. Show complete solution with complete diagrams. 1. Using area-moment method for the overhanging beam shown. a. compute the value of 8 at the right end b. calculate the value of 8 midway between the supports. Use I = 60 in. and...
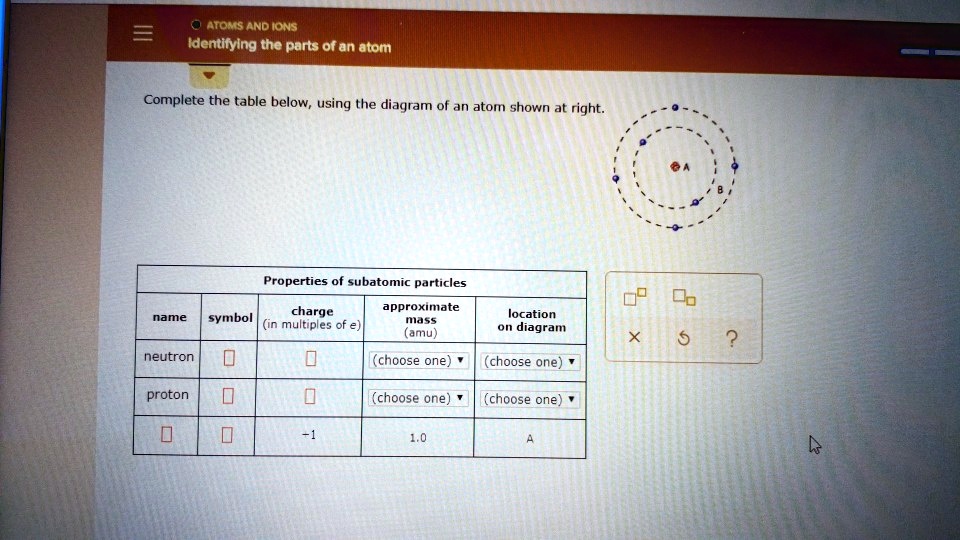
Science Chemistry Q&A Library Complete the table below, using the diagram of an atom shown at right. Properties of subatomic particles approximate charge (in multiples of e) location name symbol mass on diagram (amu) 1.0 A -1 0.0005 B n° (choose one) v (choose one)♥. Complete the table below, using the diagram of an atom shown at right. Transcribed image text: Complete the table below, using the diagram of an atom shown at right. Properties of subatomic particles charge approximate mass (in multiples of e) (amu) name symbol location on diagram X 5 ? +1 1.0 А neutron 0 (choose one) (choose one) 0 1.0 A Complete the table below, using the diagram of an atom shown at right. In the diagram below, this group is designated as an R-group. Within living organisms there are 20 amino acids used as protein building blocks. They differ from one another only at the R-group postion. The basic structure of an amino acid is shown below: Figure 11.2 General Structure of an Alpha Amino Acid Complete the table below, using the diagram of an atom shown at right. Properties of subatomic particles approximate location charge (in multiples of e) symbol mass on diagram name (amu) -1 0.0005 B 1.0, (choose one) (choose one) v :-
Transcribed image text: Complete the table below, using the diagram of an atom shown at right. Properties of subatomic particles approximate mass (amu) ...
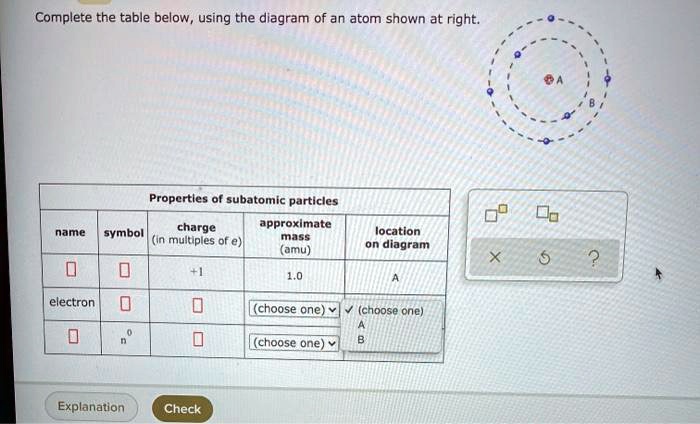
Question: Complete the table below, using the diagram of an atom shown at right. G Properties of subatomic particles name symbol charge (in multiples of e) approximate mass (amu) location on diagram Х 5 ? 0 1.0 A 0 n (choose one) V (choose one) v D -1 0.0005 B.
The atom shown in the diagram above has a total of 5 electron orbitals. ... Complete the sentences below by making use of the periodic table. Bromine has 35 protons. ... Atoms with an outer shell that is almost empty are located on the right side of the Periodic Table while atoms with an outer shell that is full or almost full are located on ...
Complete the table below, using the diagram of an atom shown at right. Properties of subatomic particles approximate symbol charge mass (in multiples of e) (amu) name location on diagram Х х 5 ? +1 1.0 А electron 0 (choose one) (choose one) 0 0.0005 B.
Transcribed image text: Complete the table below, using the diagram of an atom shown at right. Properties of subatomic particles approximate charge symbol ...
(d) The Lewis electron-dot diagram for C2H4 is shown below in the box on the left. In the box on the right, complete the Lewis electron-dot diagram for C2H50H by drawing in all of the electron pairs. H c:: H H ë H H: H c: H H c: H H Diagram should include all bonding pairs plus two nonbonding pairs on the O atom. (A line may be used
Complete the table below, using the diagram of an atom shown at right. Learn this topic by watching Subatomic Particles Concept Videos All Chemistry Practice Problems Subatomic Particles Practice Problems
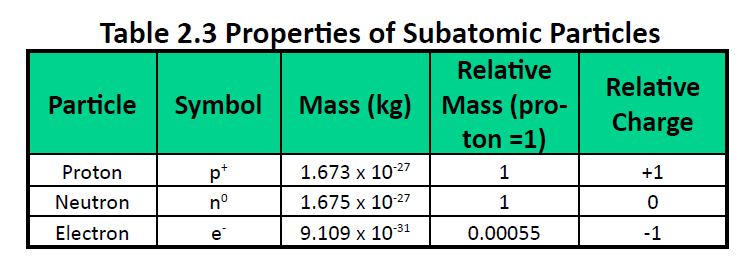
("Subatomic" means "smaller than an atom.") Here are the three types of subatomic particle you need to know for chemistry: name symbol charge (in multiples of e) mass (amu) location proton p + +1 1.0 nucleus neutron n 0 0 1.0 nucleus electron 1 0.0005 or about 1/1800 orbiting the nucleus Using these facts, which you must memorize, you can ...
Chemistry. Chemistry questions and answers. Complete the table below, using the diagram of an atom shown at right. Do name Properties of subatomic particles charge approximate symbol mass (in multiples of e) (amu) 0 0 (choose one) location on diagram Х $ ? (choose one) D 0 +1 1.0 А 0 1.0 А.
-The glucose molecule in this diagram is shown in the (ring) form.-Fructose has (six) carbon atoms. ... An atom consists of a central nucleus surrounded by electrons that are in constant motion. True ... Complete the sentences below by making use of the periodic table.-Bromine has (35) protons.-(Calcium) has an atomic mass of 41. ...
Chemistry questions and answers. Complete the table below, using the diagram of an atom shown at right. name Properties of subatomic particles approximate symbol charge mass location (In multiples of e) on diagram (amu) р (choose one) (choose one) Х $ ? 0 0 0 n (choose one) (choose one) 0 +1 1.0.
Complete the table below, using the diagram of an atom shown at right. Properties of subatomic particles name charge symbol (in multiples of e) approximate mass (amu) location diagram 1.0 electron O (choose one) Complete the table below, using the diagram of an atom shown at right. Properties of subatomic...
Write a brief passage describing a neutral atom of nitrogen-14 (N-14). Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. Use the periodic table to help you.
Complete the table below, using the diagram of an atom shown at right. Properties of subatomic particles approximate charge (in multiples of e) location name symbol mass on diagram (amu) electron (choose one) (choose one) ♥ proton (choose one) v (choose one) ♥ -1 0.0005 B OOO
Question: Complete the table below, using the diagram of an atom shown at right. This problem has been solved! See the answer ...
Oct 17, 2021 · Complete the table below, using the diagram of an atom shown at right. A name Properties of subatomic particles Aharge approximate symbol mass (in multiples of e) (amu) name location on diagram X5 ? O 1.0 А 0 1.0 А proton (choose one) (choose one) Complete the table below, using the diagram of an atom shown at right.
We compare the bonding of the atom in the molecule to the valence electron structure If the atom has one more electron in the molecule, it is shown with a "-" charge If the atom has one less electron, it is shown with a "+" charge Neutral molecules with both a "+" and a "-" are dipolar

:max_bytes(150000):strip_icc()/element-list-names-atomic-numbers-606529_V1_FINAL-f332cfc84a494b7782d84fc986cdaf86.png)

















0 Response to "37 complete the table below using the diagram of an atom shown at right"
Post a Comment