38 molecular orbital diagram for he2
chemical bonding - chemical bonding - Molecular orbitals of H2 and He2: The procedure can be introduced by considering the H2 molecule. The energies of these atomic orbitals are shown on either side of the molecular orbital energy-level diagram in Figure 14. (It may be recalled from the... - MO diagrams for Inorganic complexes. • It is a waste of both the lecturers and students time if the tutorial to ends up being a lecture covering questions. 5. An introduction to Molecular Orbital Theory.
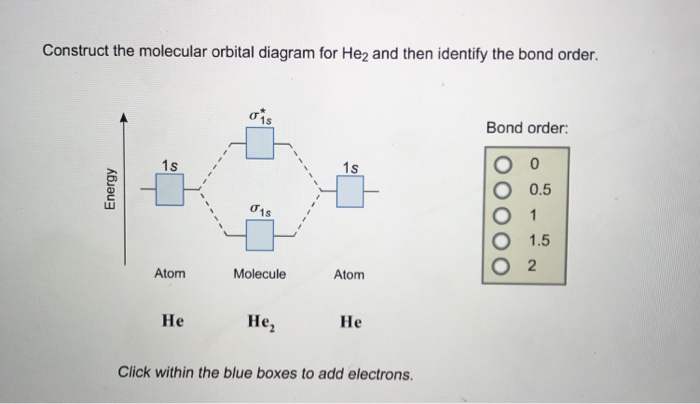
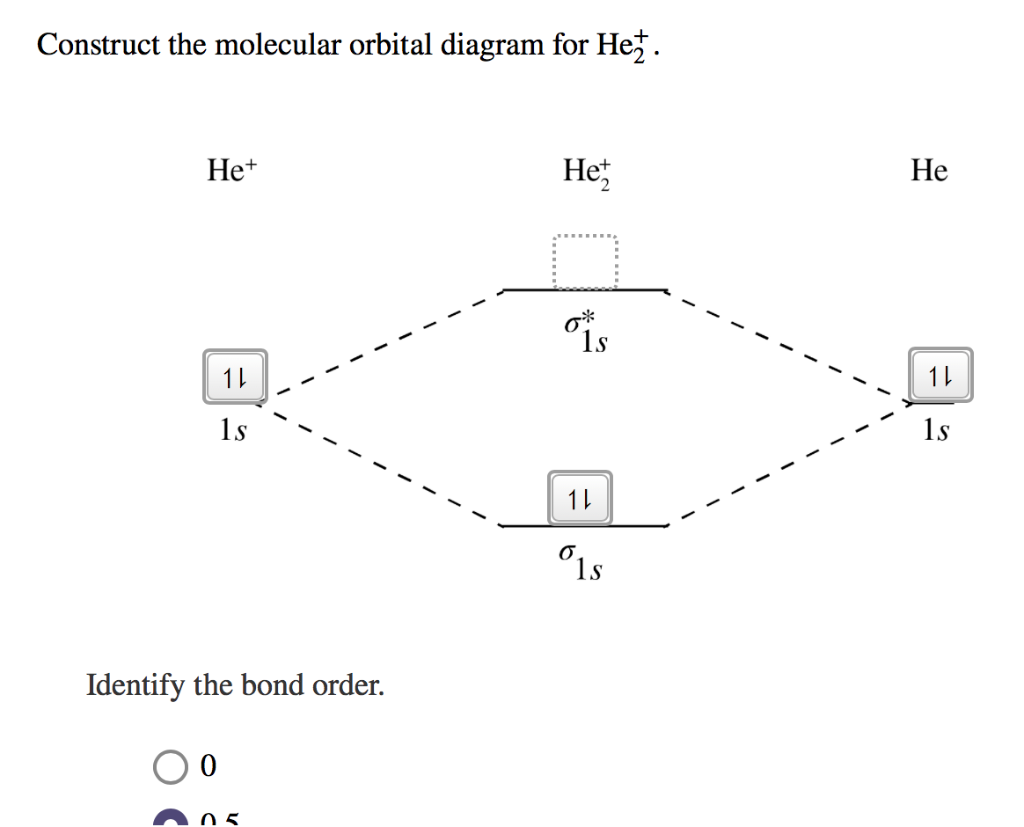
Transcribed image text : Construct the molecular orbital diagram for He2 and then identify the bond order. Bond order: Click within the blue boxes to add electrons.
Molecular orbital diagram for he2
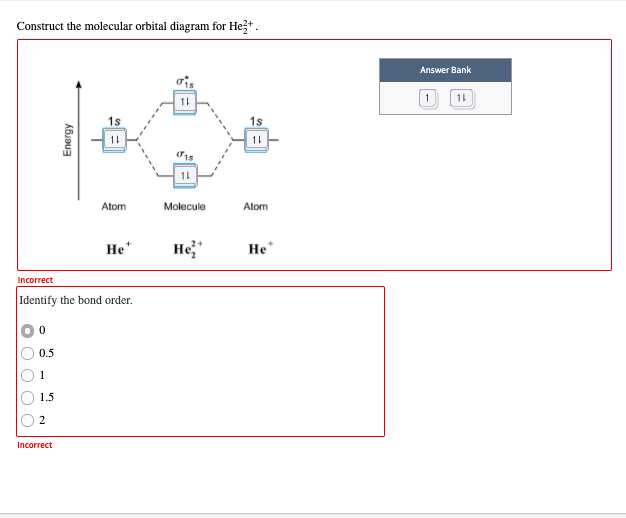
Draw a molecular orbital diagram for He2, calculate the bond order and determine its stability. Show the shapes of the molecular orbitals also. Draw the molecular orbital diagram for each and explain. Molecular orbital: Molecular orbitals are formed by the linear combination of atomic orbitals. There ...1 answer · Top answer: In the molecular di-cation of helium ion, two electrons are less than helium atom. Two electrons are to be filled in the molecular orbitals. These two... A molecular orbital diagram, or MO diagram, is a qualitative descriptive tool explaining chemical bonding in molecules in terms of molecular orbital theory in general and the linear combination of atomic orbitals (LCAO) method in particular.
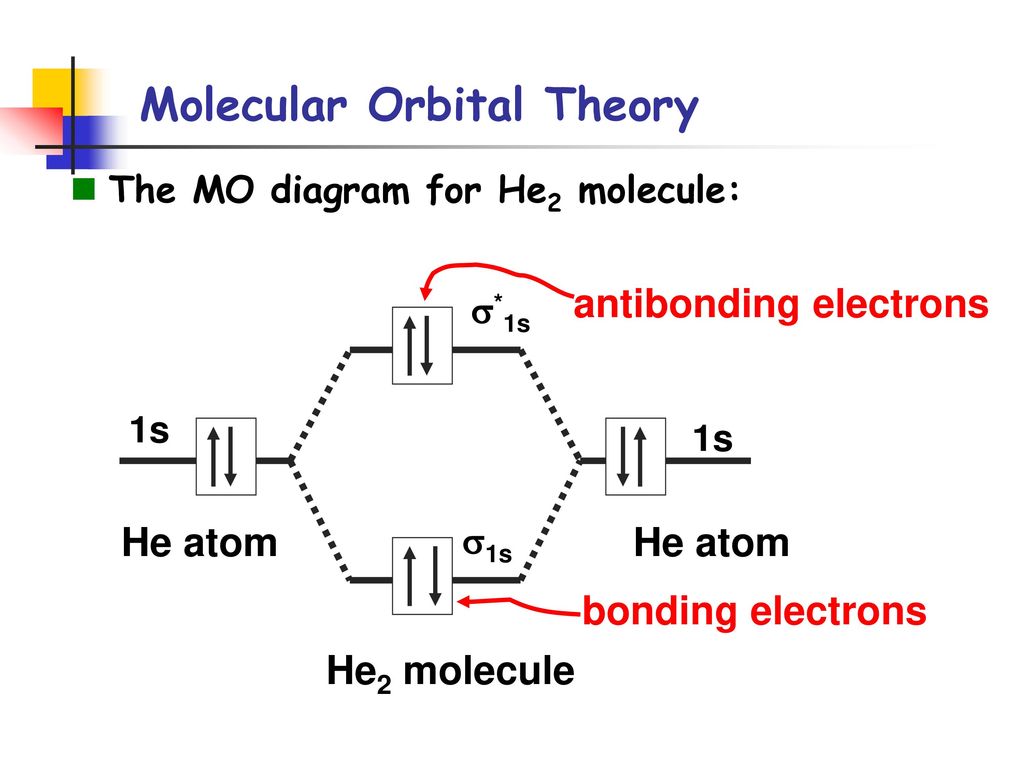
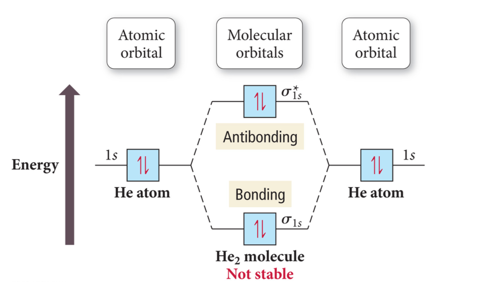
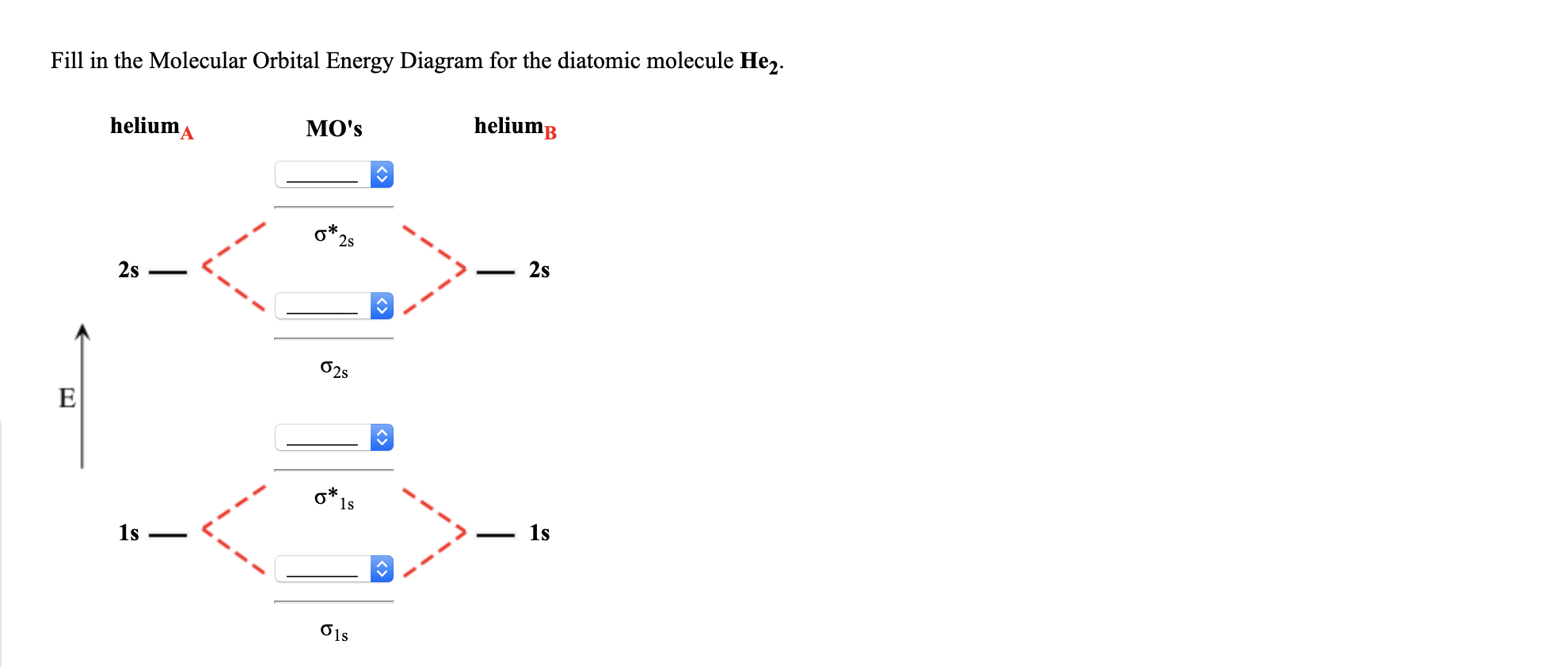
Molecular orbital diagram for he2. Similarly, the molecular orbital diagrams for homonuclear diatomic compounds of the alkaline earth metals (such as Be2), in which each metal atom has an ns2 valence electron configuration, resemble the diagram for the He2 molecule in part (c) in Figure 9.20 "Molecular Orbital Energy-Level... This molecular orbital model can be used to explain why He2 molecules don't exist. Combining a pair of helium atoms with 1s2 electron configurations would The molecular orbital diagram for an O2 molecule would therefore ignore the 1s electrons on both oxygen atoms and concentrate on the... He2. Lewis Structure: Molecular Orbital Energy Diagram. Total # of bonding electrons. The orbital correlation diagram for diboron, however, is not generally applicable for all homonuclear diatomic molecules. It turns out that only when the bond lengths are relatively short (as in B2, C2, and N2) can... The bonding diagram for the hypothetical molecule He2. Molecular orbital diagram for hydrogen: For a diatomic molecule, an MO diagram effectively shows the energetics of the bond between the two atoms, whose AO unbonded energies are shown on the sides.
Before we can draw a molecular orbital diagram for B₂, we must find the in-phase and out-of-phase overlap combinations for boron's atomic orbitals. The video below describes how to generate molecular orbital diagrams for B₂ and other diatomic molecules from Row 2 elements of the... In chemistry, a molecular orbital is a mathematical function describing the location and wave-like behavior of an electron in a molecule. The bond order 12 x number of bonding electrons number of antibonding electrons 12 x 2 20. Nonbonding sigma is occupied and then the sigma... 3) If Nb = Na ,the molecule is again unstable because influence of electrons in the antibonding molecular orbital is greater than the bond influence of electron in the bonding molecular orbitals. No bond is formed between two helium therefore He2 does not exist.
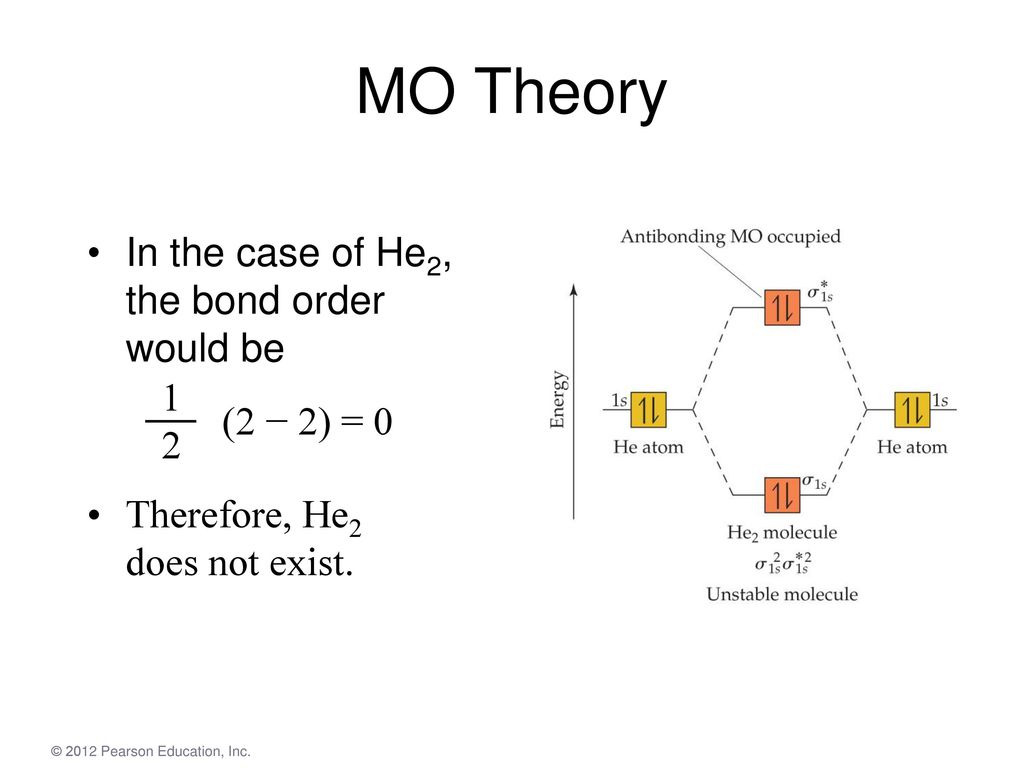
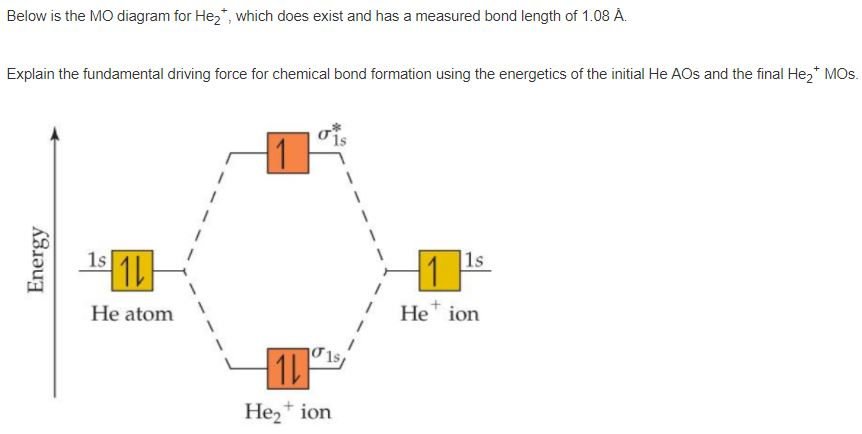
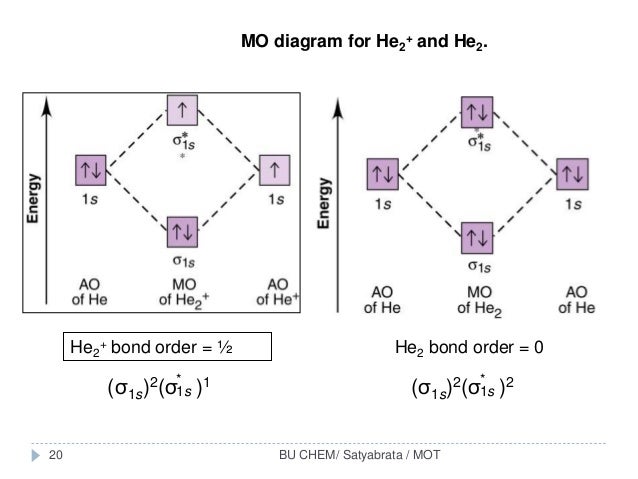
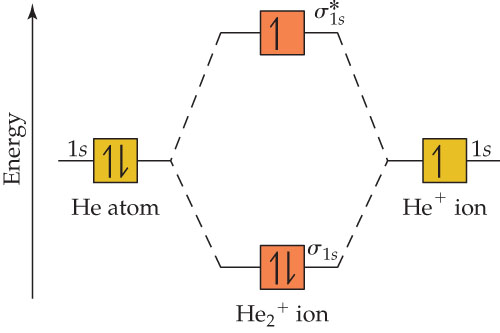
Figure 10. The molecular orbital energy diagram predicts that He2 will not be a stable molecule, since it has equal numbers of bonding and Obtain the molecular orbital diagram for a homonuclear diatomic ion by adding or subtracting electrons from the diagram for the neutral molecule. We now consider a hypothetical molecule He2. This would also have two molecular orbitals formed from the overlap of 1s orbitals on the atoms, giving a molecular orbital diagram of the same appearance as the one above. However, since each He atom has two electrons in its outer shell... : Molecular Orbital Energy-Level Diagram for H 2. The two available electrons (one from each H atom) in this diagram fill the bonding σ 1 s molecular Part (b) in Figure [Math Processing Error]. is the molecular orbital energy-level diagram for He 2 + . This ion has a total of three valence electrons. How to write simple Molecular Orbital Diagrams and determine the Bond order.
Molecular orbital : A molecule in which all the electrons are paired, is called diamagnetic. | Online Chemistry tutorial IIT, CBSE Chemistry, ICSE Chemistry, engineering and medical chemistry entrance exams, Chemistry Viva, Chemistry Job interviews. M.O. diagram of He2 molecule
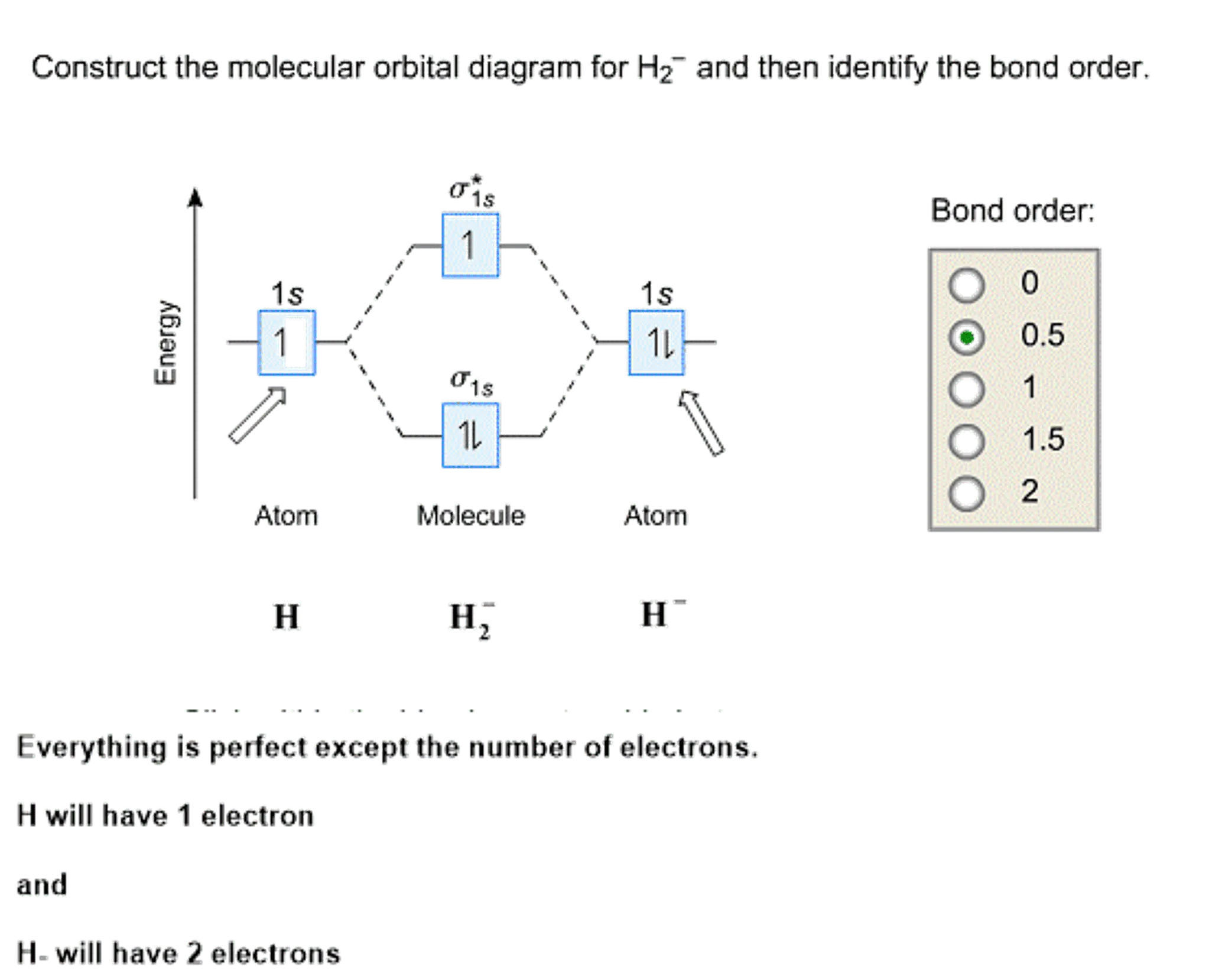
In a bonding molecular orbital, the electron density is high between the two atoms, where it stabilizes the arrangement by exerting a strong attraction for both nuclei. The energy level diagram for He2 is similar to that for H2 except that it has two more electrons.
Figure 8.36 The molecular orbital energy diagram predicts that He2 will not be a stable molecule, since it has equal numbers of bonding and Obtain the molecular orbital diagram for a homonuclear diatomic ion by adding or subtracting electrons from the diagram for the neutral molecule.
Molecular Orbital Diagram for Hydrogen Gas (H2). Fill from the bottom up, with 2 electrons total. Bonding Order is 1, and it is ... MOLECULAR ORBITAL THEORY Lecture 7 Containing below points:- 1. MO Energy Level Diagram for He2 molecule 2. MO ...

Energy-level diagram for the He2+ ion.Which electrons in this diagram contribute to the stability of the He2+ ion?
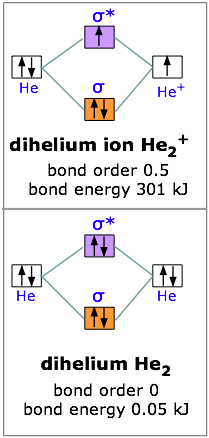
The molecular orbital (MO) theory is a way of looking at the structure of a molecule by using molecular orbitals that belong to the molecule as a whole. In the case of He 2 , on the other hand, the bond order is ½ (2 − 2) = 0. This means that He 2 is not a stable molecule. Multiple Bonds.
A bare molecular orbital diagram is presented and you must drag the correct orbitals and labels onto the diagram. The diagram is then completed by filling the energy levels with the correct number of electrons. The following molecules are currently available: Molecules of the First Row
So for this next problem were given this molecular orbital diagram and were asked to figure out which electrons in this diagram contribute to the stability ...Sep 17, 20201 answer · Top answer: 1/2
Valence bond (VB) theory gave us a qualitative picture of chemical bonding, which was useful for predicting the shapes of molecules, bond strengths, etc. It fails to describe some bonding situations accurately because it ignores the wave nature of the electrons.
1. Sketch the qualitative molecular orbital diagram for XeF2. The molecule is linear and symmetric. Assume the valence 5s-orbitals of Xe are sufficiently Determine the primary MOs that determine the bond order. Compare the general features of your MO diagram to the MO diagram for [F-H-F]...
According to the molecular orbital theory, in a supposed He2 molecule, both the bonding and the antibonding orbitals will have 2 electrons each. In order to predict the bond order, molecular orbital diagram for H2- is to be drawn. According to MOT number of atomic orbitals combined is equal to...
Molecular Orbital Diagram Of H2, He2, Li2 And Be2 Molecules. Cb-31Molecular Orbital Diagramh2He2Explanation In Tamiltn 11Th Stdcbse. 04:59. 570 40.9K. Molecular Orbital (Mo) Diagram For O2(
I have been taught that the MO diagram is different for molecules with 14 or less electrons than the one used for molecules with 15 or more electrons. All this is simply because the primitive molecular orbital theory does not explain the things, rather it rationalizes them (for differences between...
Introduction to Molecular Orbital Theory. Valence Bond Theory fails to answer certain questions like Why He2 molecule does not exist and why O2 is No. 9 Molecular Orbital Diagram for CO. Analysis done by Bond Order. If value of bond order is positive, it indicates a stable molecule and if the value...
Simple molecular orbital diagrams. Dihydrogen and its ion H2+. Dihelium He2. The molecular orbital model is by far the most productive of the various models of chemical bonding, and serves as the basis for most quantiative calculations, including those that lead to many of the...
A molecular orbital diagram, or MO diagram, is a qualitative descriptive tool explaining chemical bonding in molecules in terms of molecular orbital theory in general and the linear combination of atomic orbitals (LCAO) method in particular.
Draw the molecular orbital diagram for each and explain. Molecular orbital: Molecular orbitals are formed by the linear combination of atomic orbitals. There ...1 answer · Top answer: In the molecular di-cation of helium ion, two electrons are less than helium atom. Two electrons are to be filled in the molecular orbitals. These two...
Draw a molecular orbital diagram for He2, calculate the bond order and determine its stability. Show the shapes of the molecular orbitals also.
















0 Response to "38 molecular orbital diagram for he2"
Post a Comment