40 energy level diagram hydrogen
The energy level of the bonding orbitals is lower, and the energy level of the antibonding orbitals is higher. For the bond in the molecule to be stable, the covalent bonding electrons occupy the lower energy bonding orbital, which may be signified by such symbols as … The following diagram shows the energy level diagram of the Hydrogen atom. A Hydrogen atom has only one single electron revolving around the nucleus at the lowest energy level. When we heat a hydrogen atom, the electron gains energy and moves up to higher energy levels.
Energy storage is the capture of energy produced at one time for use at a later time to reduce imbalances between energy demand and energy production. A device that stores energy is generally called an accumulator or battery.Energy comes in multiple forms including radiation, chemical, gravitational potential, electrical potential, electricity, elevated temperature, latent …
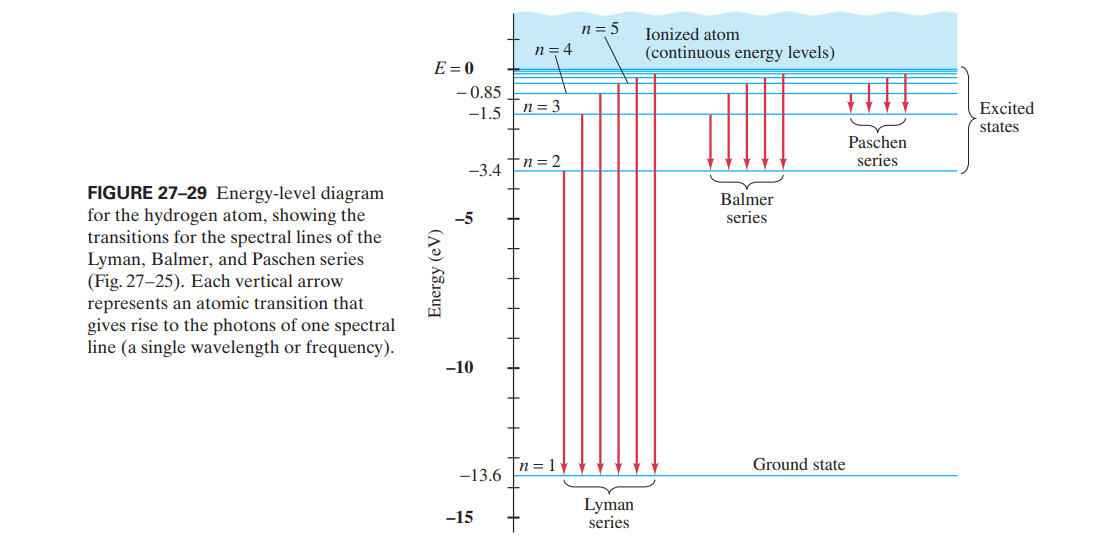
Energy level diagram hydrogen
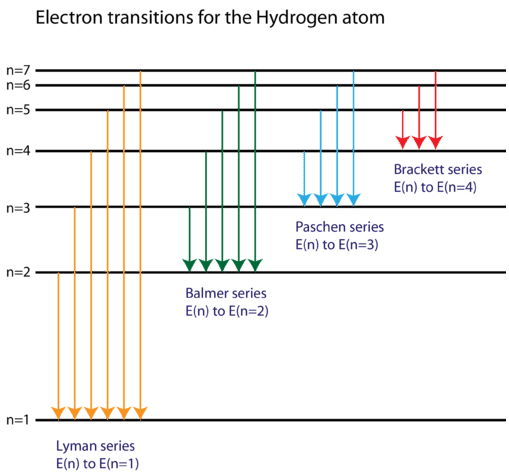
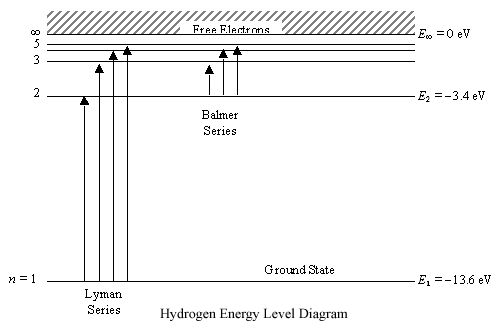
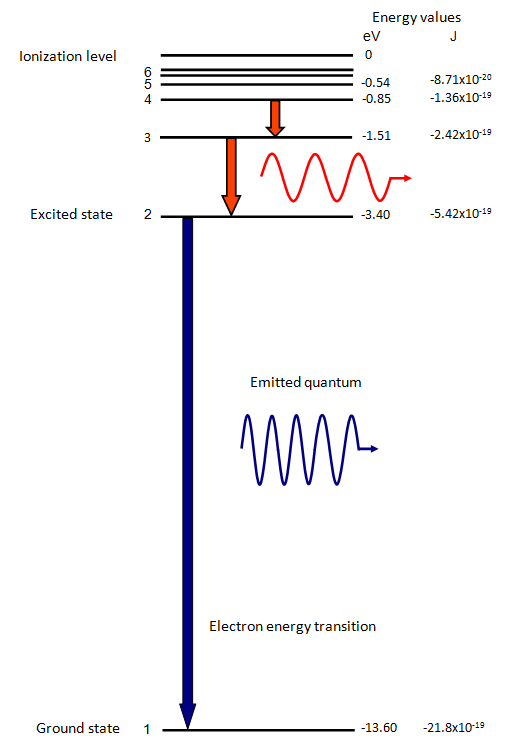
The energy levels agree with the earlier Bohr model, and agree with experiment within a small fraction of an electron volt. If you look at the hydrogen energy levels at extremely high resolution, you do find evidence of some other small effects on the energy. The 2p level is split into a pair of lines by the spin-orbit effect. Energy level diagram . The energy of the electron in the n th orbit of the hydrogen atom is given by, En = -13.6 /n 2 eV. Energy associated with the first orbit of the hydrogen atom is, E 1 = -13.6 /1 2 = -13.6 eV. It is called ground state energy of the hydrogen atom. Energy associated with the second orbit is given by, E 2 = -13.6 /2 2 = -3.4 eV The electrolyzer, being the conversion-in component, converts electrical energy into oxygen, low level heat, and hydrogen, which acts as the energy storage medium. Depending on the volume of gas to be stored, and the local conditions, gas storage is either on the surface, such as tube storage, or underground, preferably in salt caverns.
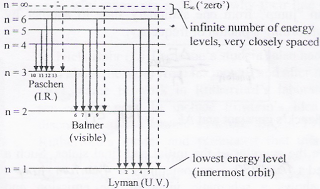
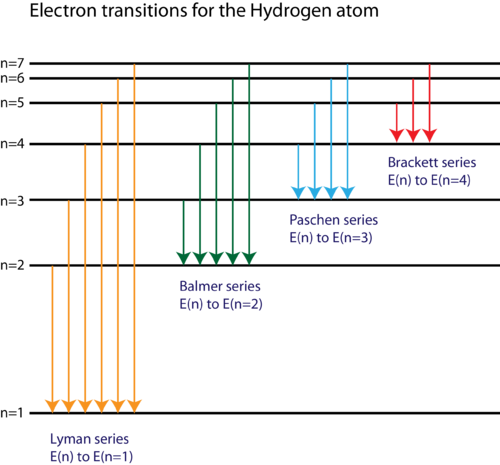
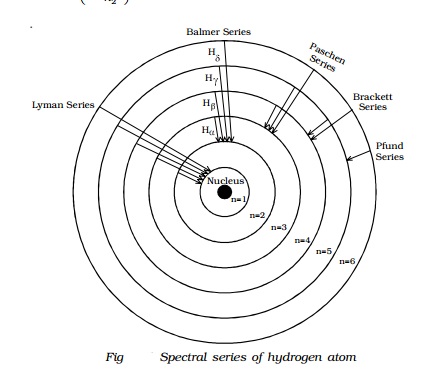
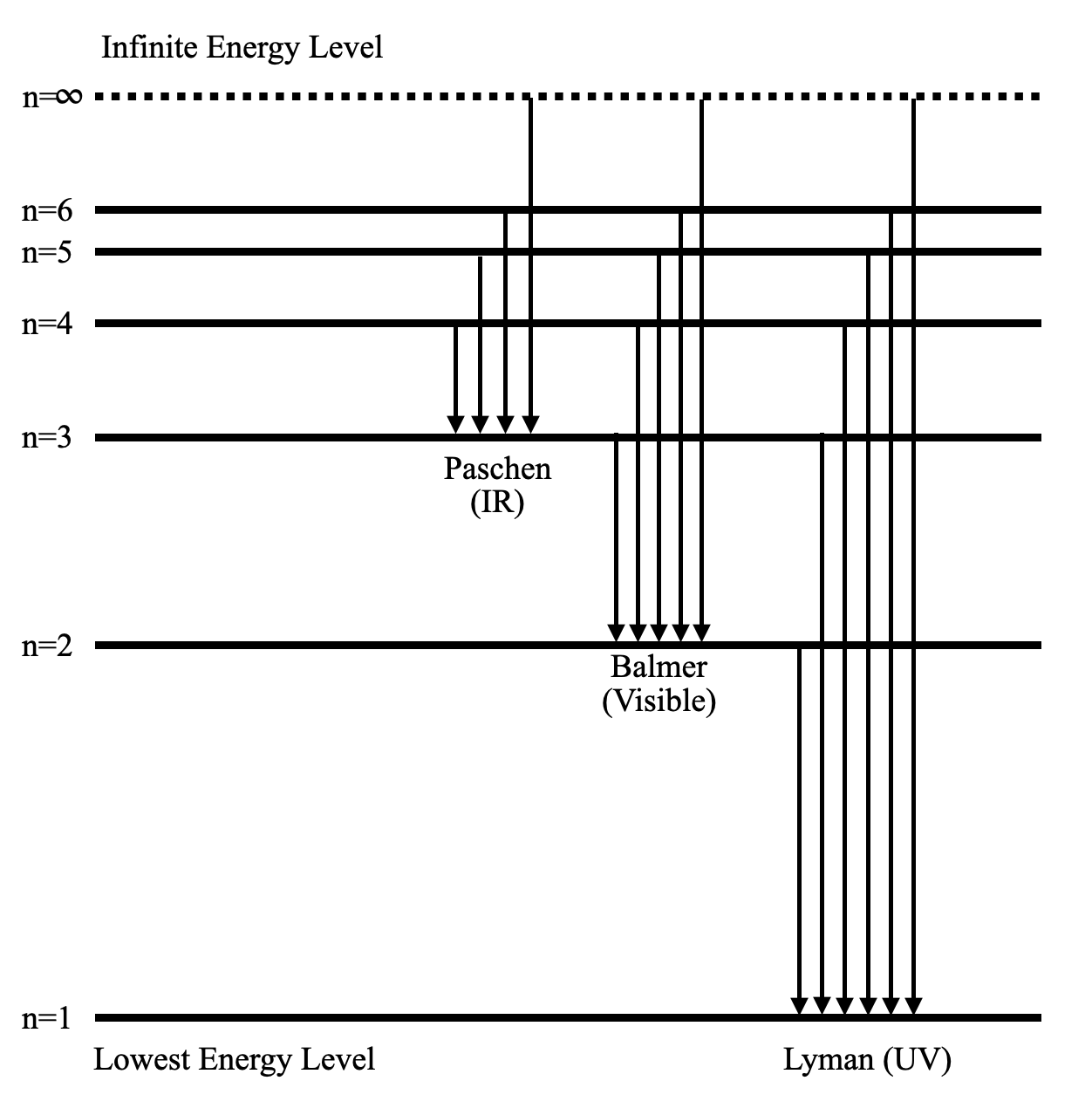
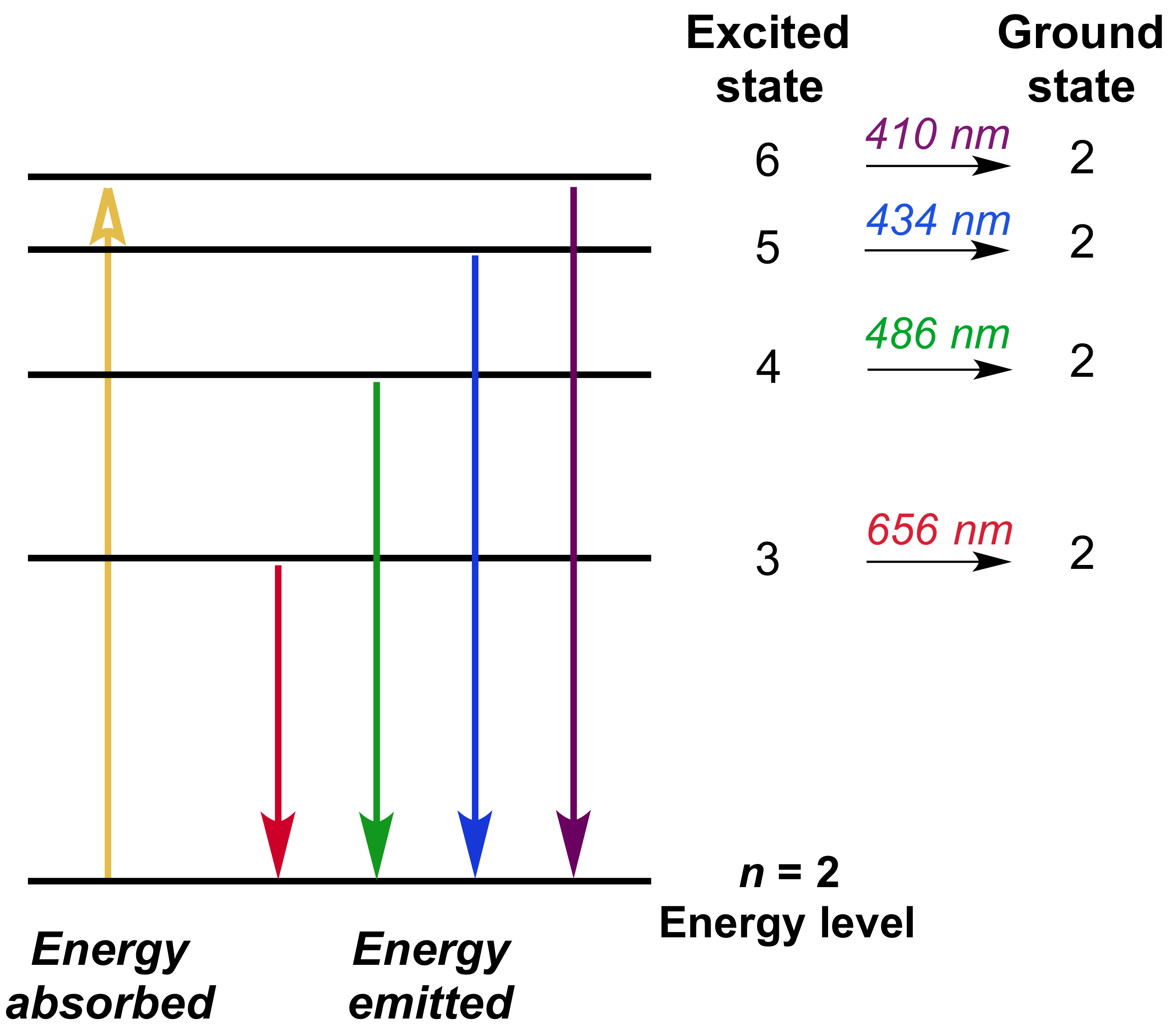
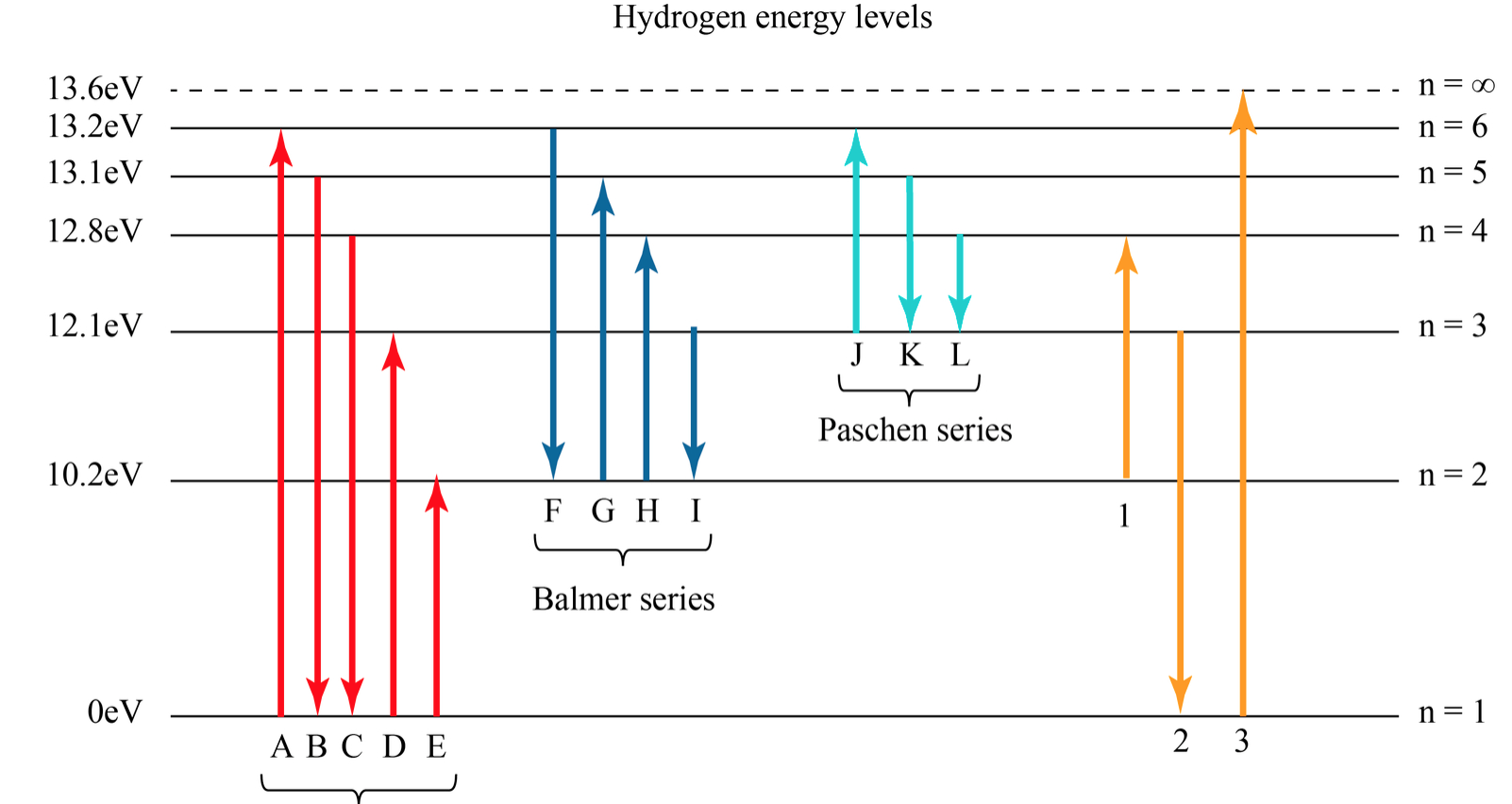
Energy level diagram hydrogen. The fraction of fixed energy a trophic level passes on to the next trophic level is called production. Green plants fix solar energy and accumulate it in organic forms as chemical energy. Since it is the first and basic form of energy storage, the rate at which the energy accumulates in the green plants or producers is known as primary ... In this section we will discuss the energy level of the electron of a hydrogen atom, and how it changes as the electron undergoes transition. According to Bohr's theory, electrons of an atom revolve around the nucleus on certain orbits, or electron shells. Each orbit has its specific energy level, which is expressed as a negative value. This is because the electrons on the orbit are … Hydrogen Spectrum - Wavelength, Diagram, Hydrogen Emission Spectrum. Hydrogen Spectrum introduction. We all know that electrons in an atom or a molecule absorb energy and get excited, they jump from a lower energy level to a higher energy level, and they emit radiation when they come back to their original states. Energy level diagrams and the hydrogen atom. It's often helpful to draw a diagram showing the energy levels for the particular element you're interested in. The diagram for hydrogen is shown above. The n = 1 state is known as the ground state, while higher n states are known as excited states. If the electron in the atom makes a transition from a particular state to a lower state, it is losing energy.
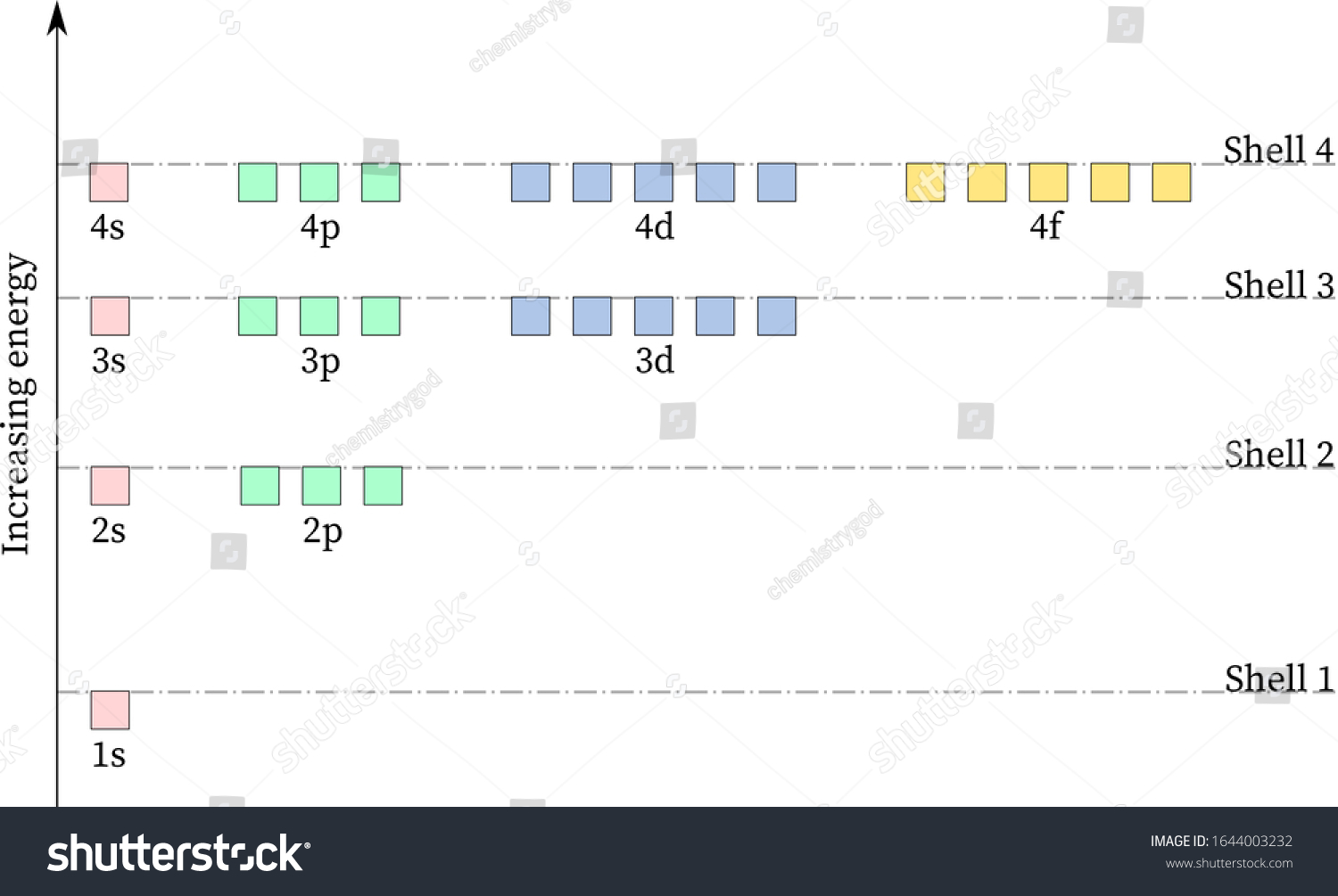
Energy level diagrams are the representation of placements or arrangements of orbitals (also known as subshells) according to their increasing energy levels. Above is the blank energy level diagram which can be used to represent the electrons for any atom under study. Energy level diagrams are known as Grotrian diagrams. The first element of the periodic table is hydrogen and its position at the beginning of the periodic table. Hydrogen is a s-block element. This article gives an idea about the electron configuration of hydrogen, period and groups, valency and valence electrons of hydrogen, bond formation, compound formation, application of different principles.The electron configuration of each atom … Nov 21, 2010 · The ionization energy of an atom is the energy required to remove the electron completely from the atom.(transition from ground state n = 0 to infinity n = ∞). For hydrogen, the ionization energy = 13.6eV; When an excited electron returns to a lower level, it loses an exact amount of energy by emitting a photon. The Lyman(ultraviolet) series of spectral lines corresponds to electron transitions from higher energy levels to level n = 1. The electrolyzer, being the conversion-in component, converts electrical energy into oxygen, low level heat, and hydrogen, which acts as the energy storage medium. Depending on the volume of gas to be stored, and the local conditions, gas storage is either on the surface, such as tube storage, or underground, preferably in salt caverns.
Energy level diagram . The energy of the electron in the n th orbit of the hydrogen atom is given by, En = -13.6 /n 2 eV. Energy associated with the first orbit of the hydrogen atom is, E 1 = -13.6 /1 2 = -13.6 eV. It is called ground state energy of the hydrogen atom. Energy associated with the second orbit is given by, E 2 = -13.6 /2 2 = -3.4 eV The energy levels agree with the earlier Bohr model, and agree with experiment within a small fraction of an electron volt. If you look at the hydrogen energy levels at extremely high resolution, you do find evidence of some other small effects on the energy. The 2p level is split into a pair of lines by the spin-orbit effect.





















0 Response to "40 energy level diagram hydrogen"
Post a Comment