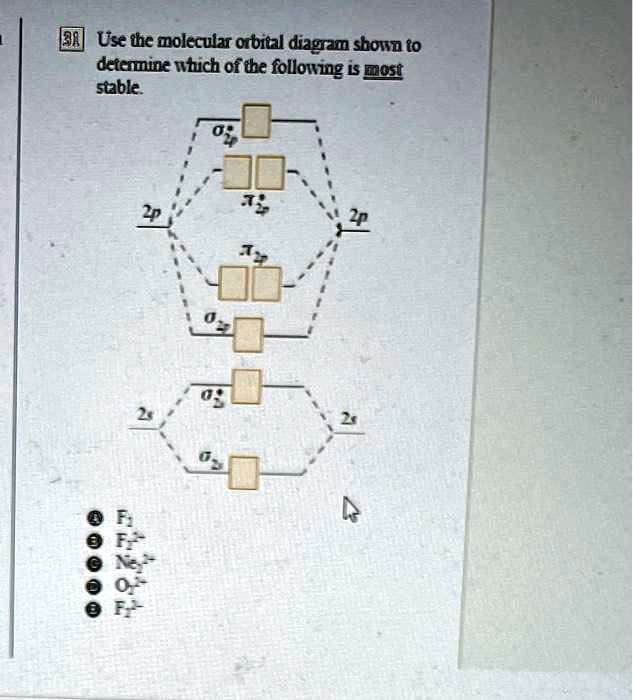
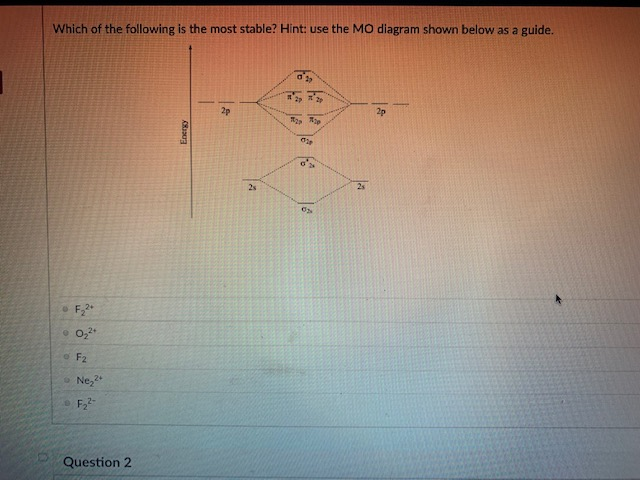
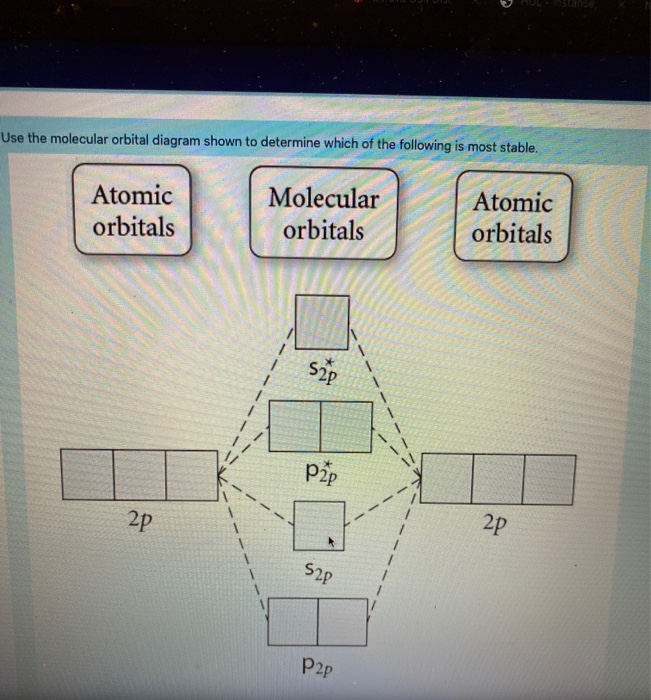
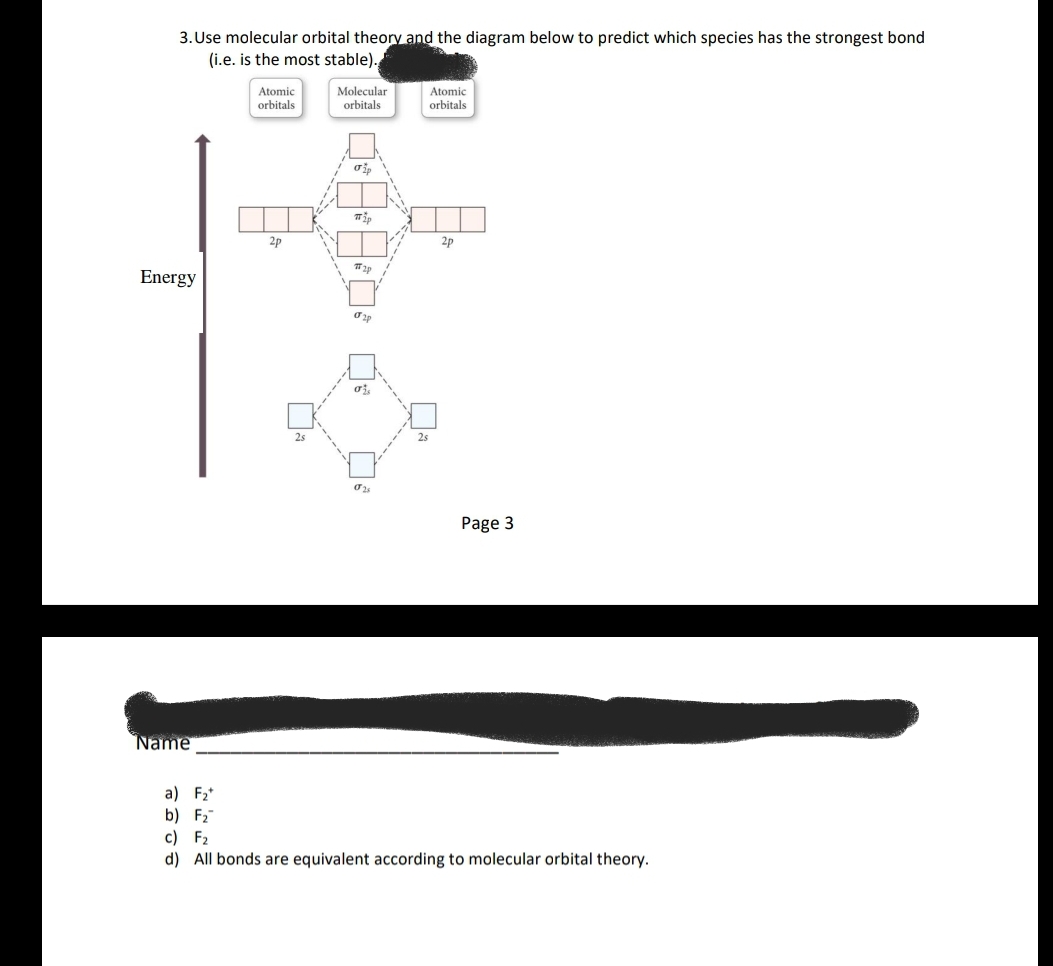
37 use the molecular orbital diagram shown to determine which of the following is most stable
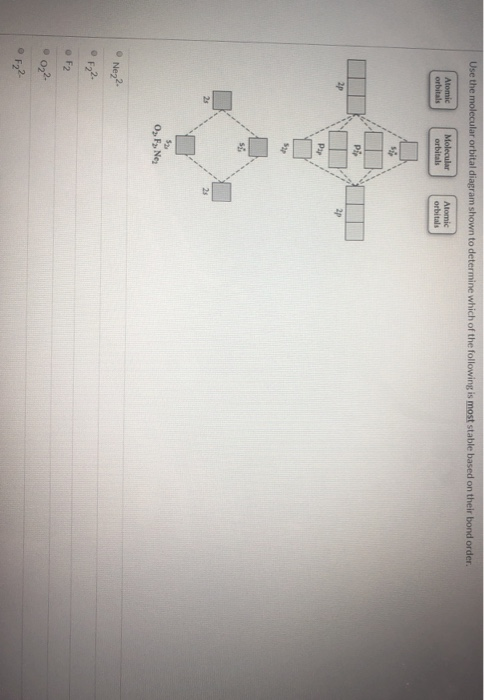
Solved Use the molecular orbital diagram shown to ... Chemistry questions and answers. Use the molecular orbital diagram shown to determine which of the following is most stable based on their bond order Atomic orbitals Molecular orbitals Atomic orbitals O, F2, Nez • Nez2 • F₂2. • F2 022 • F22. Arrange the following in order of decreasing stability. a ... Arrange the following in order of decreasing stability. a blank molecular orbital diagram (part a 1 figure) has been provided to you. rank the fluorine species from most to least stable. to rank items as equivalent, overlap them. f2, f2+, f2-
Use the molecular orbital diagram shown to determine which ... Refer to the information provided in Figure 12.4 below to answer the question(s) that follow. Figure 12.4There are two sectors in the economy, X and Y, and both are in long-run, zero-profit equilibrium at the intersections of S0 and D0.Refer to Figure 12.4.

Use the molecular orbital diagram shown to determine which of the following is most stable
chemistry 1a Chapter 10 Flashcards | Quizlet Use the molecular orbital diagram shown to determine which of the following is most stable A) Ne2^2⁺ B) F2^2⁺ C) F2^2⁻ D) F2 E) O2^2⁺ E). O2^2⁺ Use the molecular orbital diagram shown to determine which of the following are paramagnetic. A) O2 2⁺ ... Use the molecular orbital diagram shown to determine ... Problem: Use the molecular orbital diagram shown to determine which of the following is most stable.A) N22+ B) B2C) B22+D) C22-E) C22+ FREE Expert Solution Show answer Answer: Quiz 1 Flashcards | Quizlet Draw the molecular orbital diagram shown to determine which of the following is MOST stable A) B2^2+ B) C2^2+ C) N2^2+ D) C2^2-E) B2. D. Identify the number of electron groups around a molecule with so hybridization. 2. Identify the number of electron groups around a molecule with sp^2 hybridization. 3.
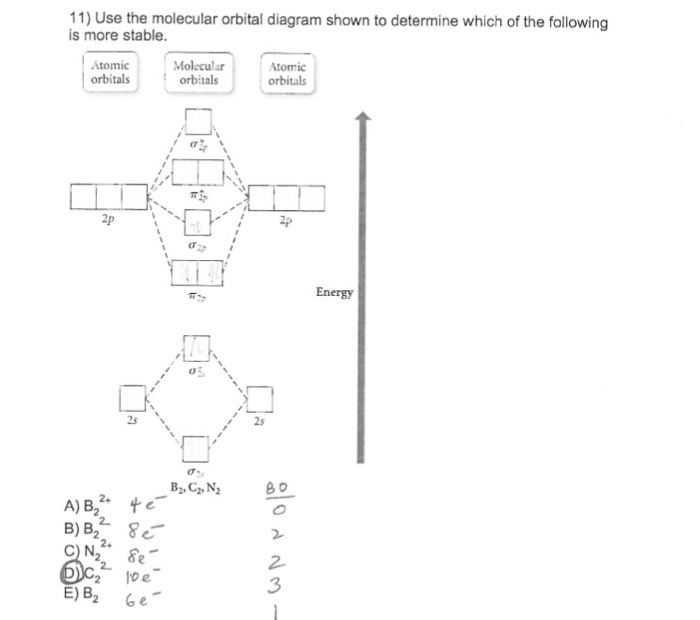
Use the molecular orbital diagram shown to determine which of the following is most stable. Recitation Week 10 (test 3 - Recitation 2) - GitHub Pages 3) Draw the molecular orbital diagram needed, and determine which of the following is paramagnetic. A) B2^2+ B) B2^2-C) N2^2+ D) C2^2-E) B2; 4) Draw the molecular orbital diagram shown to determine which of the following is most stable. A) C2^2+ B) N2^2+ C) B2; D) C2^2-E) B2^2+ 5) Which statement regarding stable heteronuclear diatomic ... Use the molecular orbital diagram shown to determine which ... Use the molecular orbital diagram shown below to determine which of the following molecules/species is most stable (O2, F2 and Ne2) . Explain their magnetic properties using the same diagram. Calculate the bond order in each of the molecule. Use the molecular orbital diagram shown to determine which ... Use the molecular orbital diagram shown to determine which of the following is most stable. Place the species B2+, B2, and B2- in orde ... - Clutch Prep Q. Use the molecular orbital diagram shown to determine which of the following is MOST stable. In molecular orbital names, s = sigma and p = pi.a. F2b.... In molecular orbital names, s = sigma and p = pi.a. F2b....
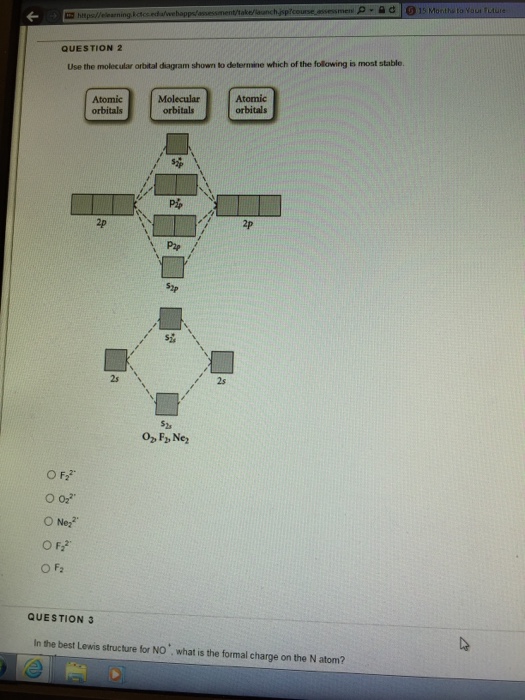
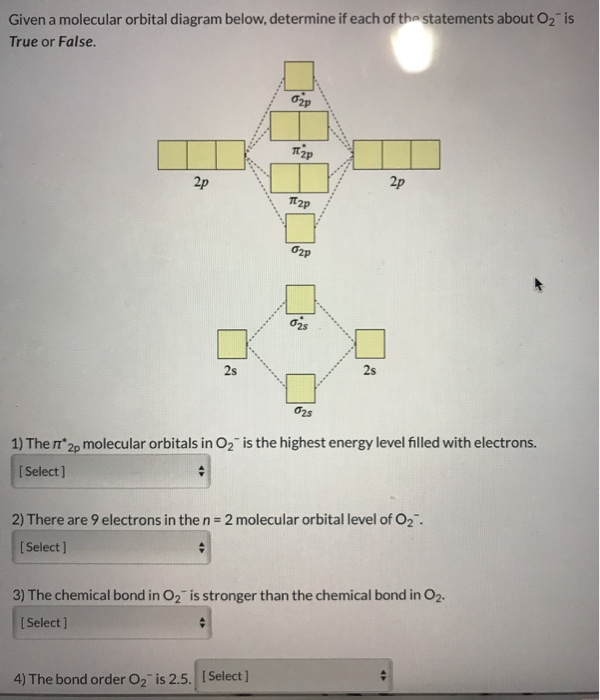
3 13 13 Use the molecular orbital diagram shown to ... 13) 13) Use the molecular orbital diagram shown to determine which of the following are paramagnetic. A) O 2 2 B) Ne 2 2 C) O 2 2 D) F 2 2 E) None of the above are paramagnetic. 4. 14) 14) Use the molecular orbital diagram shown to determine which of the following is most stable. A) C 2 2 B) B 2 2 C) C 2 2 D) N 2 2 E) B 2. Use the molecular orbital diagram shown to... | Clutch Prep Use the molecular orbital diagram shown to determine which of the following are paramagnetic. A. Ne 22+ B. O 22+ C. F 22+ D. O 22- E. None of the above are paramagnetic. Learn this topic by watching MO Theory: Homonuclear Diatomic Molecules Concept Videos. Solved Use the molecular orbital diagram shown to ... Use the molecular orbital diagram shown to determine which of the following is most stable based on their bond order. Atomic orbitals Molecular orbitals Atomic orbitals O, F, Ne Ne22 F₂2. F2 . 022- • F22. Question: Use the molecular orbital diagram shown to determine which of the following is most stable based on their bond order. chem test 3 (ch 6&7) Flashcards - Quizlet Use the molecular orbital diagram shown to determine which of the following is most stable. C) O2^2+ Use the molecular orbital diagram shown to determine which of the following are paramagnetic. B) B2. Identify the number of bonding pairs and lone pairs of electrons in water.
C22- Molecular Orbital Diagram Solution: Use the molecular orbital diagram shown to determine which of the following is most stable. A) N22+ B) B2 C) B22+ D) C E) C22+. Sketch the molecular orbital energy level diagram for the ion. How many net σ and π bonds does the ion have? What is the carbon-carbon bond order? How has the bond order changed on adding electrons to C 2 ... Use the molecular orbital diagram shown to... | Clutch Prep Construct the molecular orbital diagram for each molecule and determine which is most stable by counting the electrons in anti-bonding MOs. Recall that as the electrons in the anti-bonding MO (denoted by *) increases, the more unstable the compound is. Solved Use the molecular orbital diagram shown to ... Solved Use the molecular orbital diagram shown to determine | Chegg.com. Science. Chemistry. Chemistry questions and answers. Use the molecular orbital diagram shown to determine which of the following is most stable. Atomic orbitals Molecular orbitals Atomic orbitals 7 Sp 2p 2p 2p TT 3 2s 25 B, C, N, 2 2 N2 B2 C2 2 B2 C22. Chemistry II Kinetics Test Flashcards - Quizlet b. When two atomic orbitals come together to form two molecular orbitals, one molecular orbital will be lower in energy than the two separate atomic orbitals and one molecular orbital will be higher in energy than the separate atomic orbitals. c. Electrons placed in anti-bonding orbitals stabilize the ion/molecule. d.
[Answered] Use the molecular orbital diagram shown to ... Apr 28, 2017 · The most stable element based on the molecular orbital diagram is . Explanation: Molecular Orbital Theory (MOT) was proposed by Hund and Mulliken. The theory describes the bonding in molecules, elements, or atoms. The theory uses Molecular Orbital (MO) diagram to explain the bonding between atoms. For example, to determine the stability of an ...
Use the molecular orbital diagram shown to determine which ... SOMEONE ASKED 👇. Use the molecular orbital diagram shown to determine which of the following is most stable.
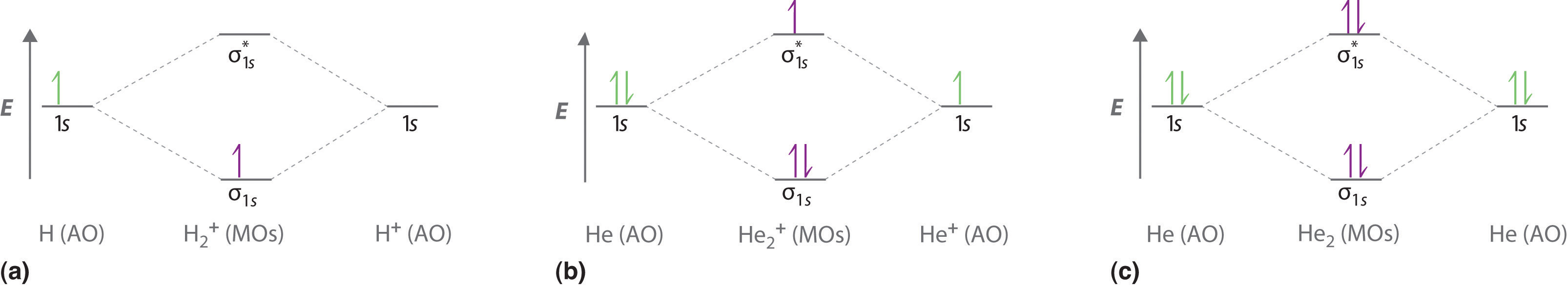
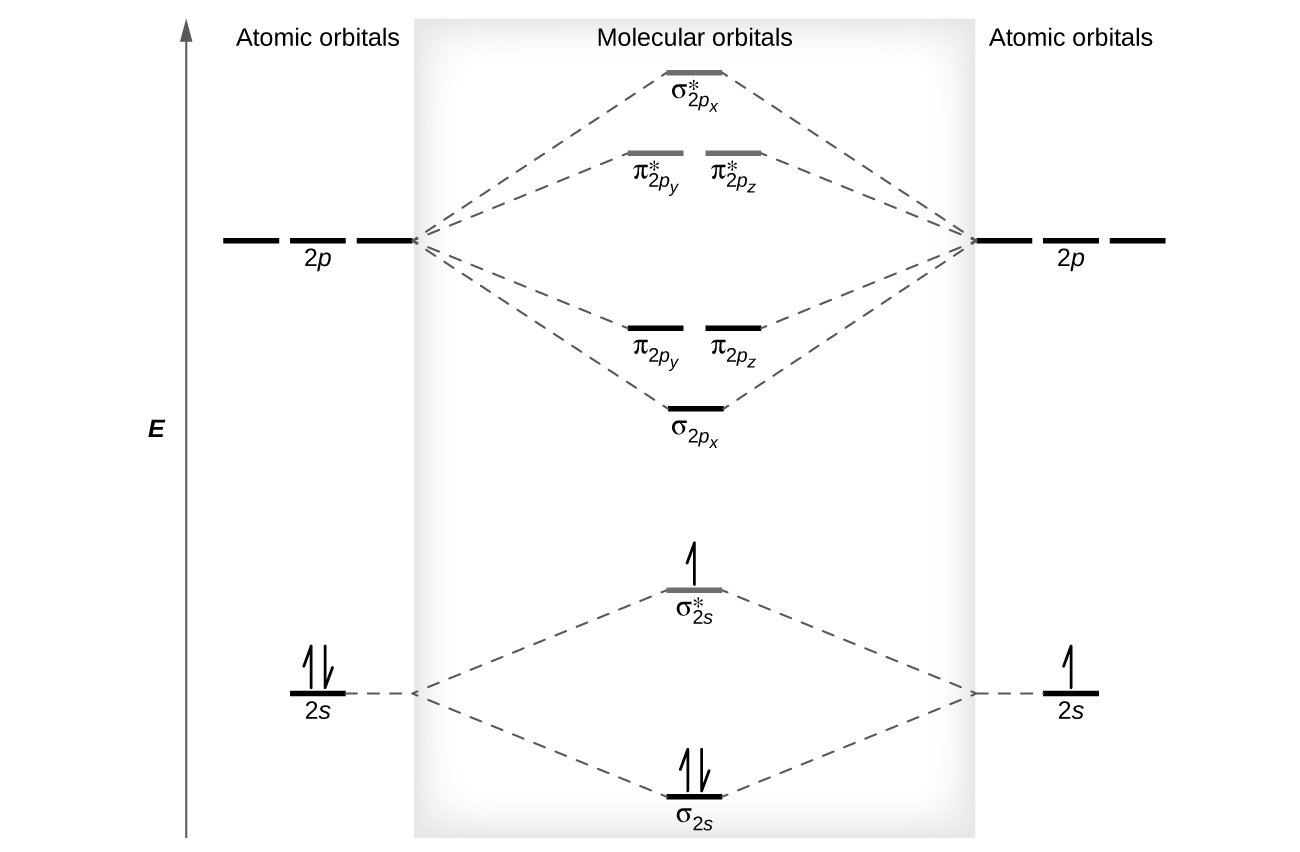
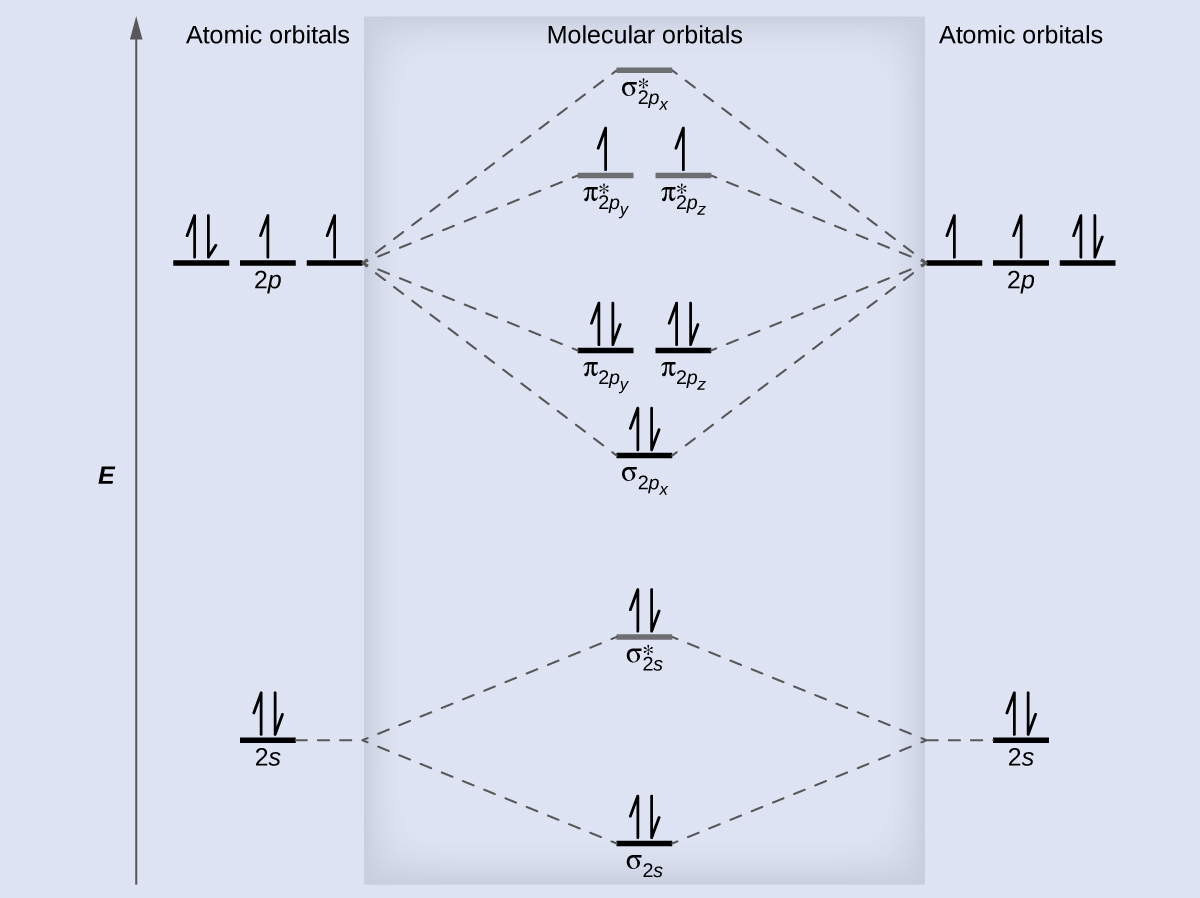
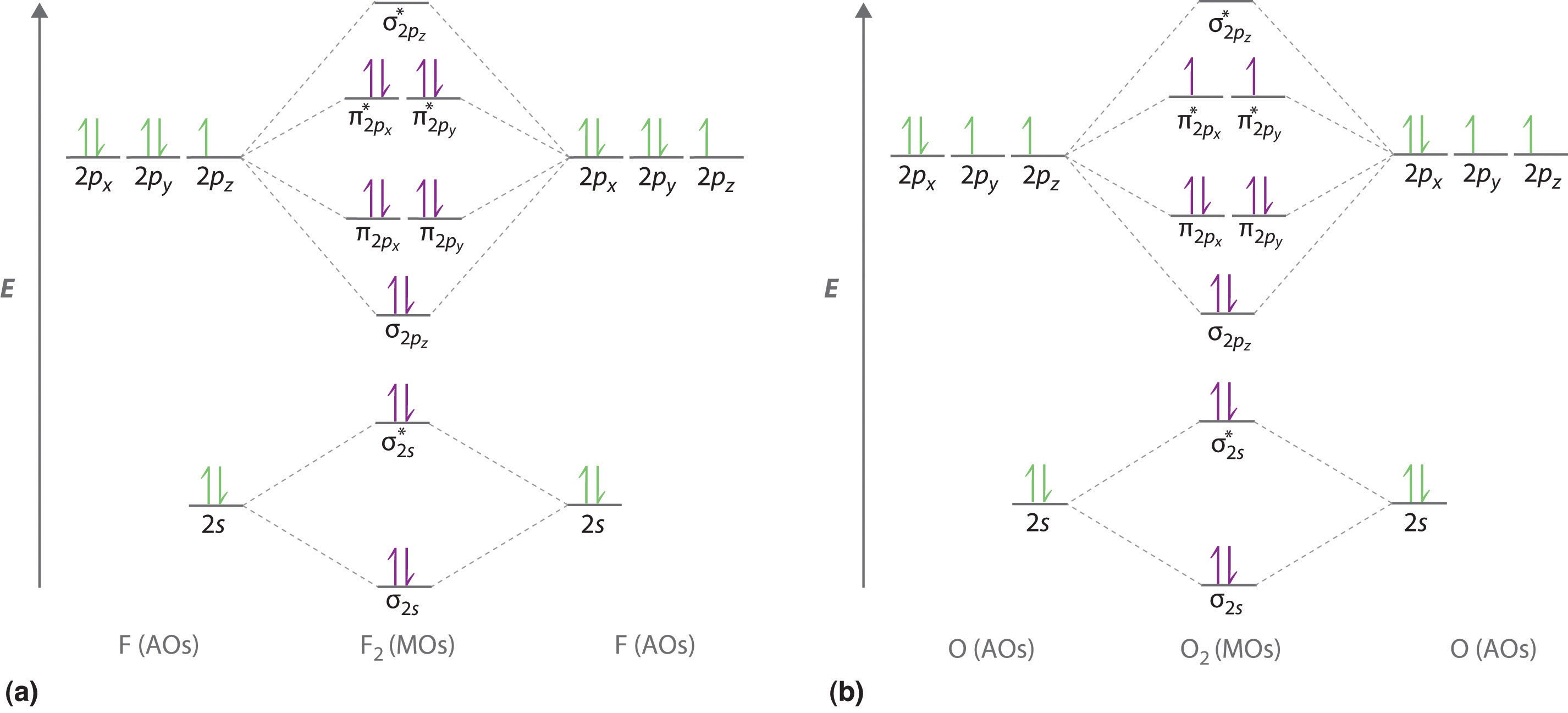
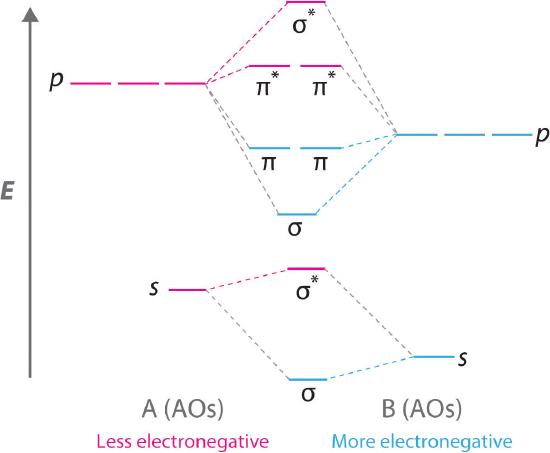
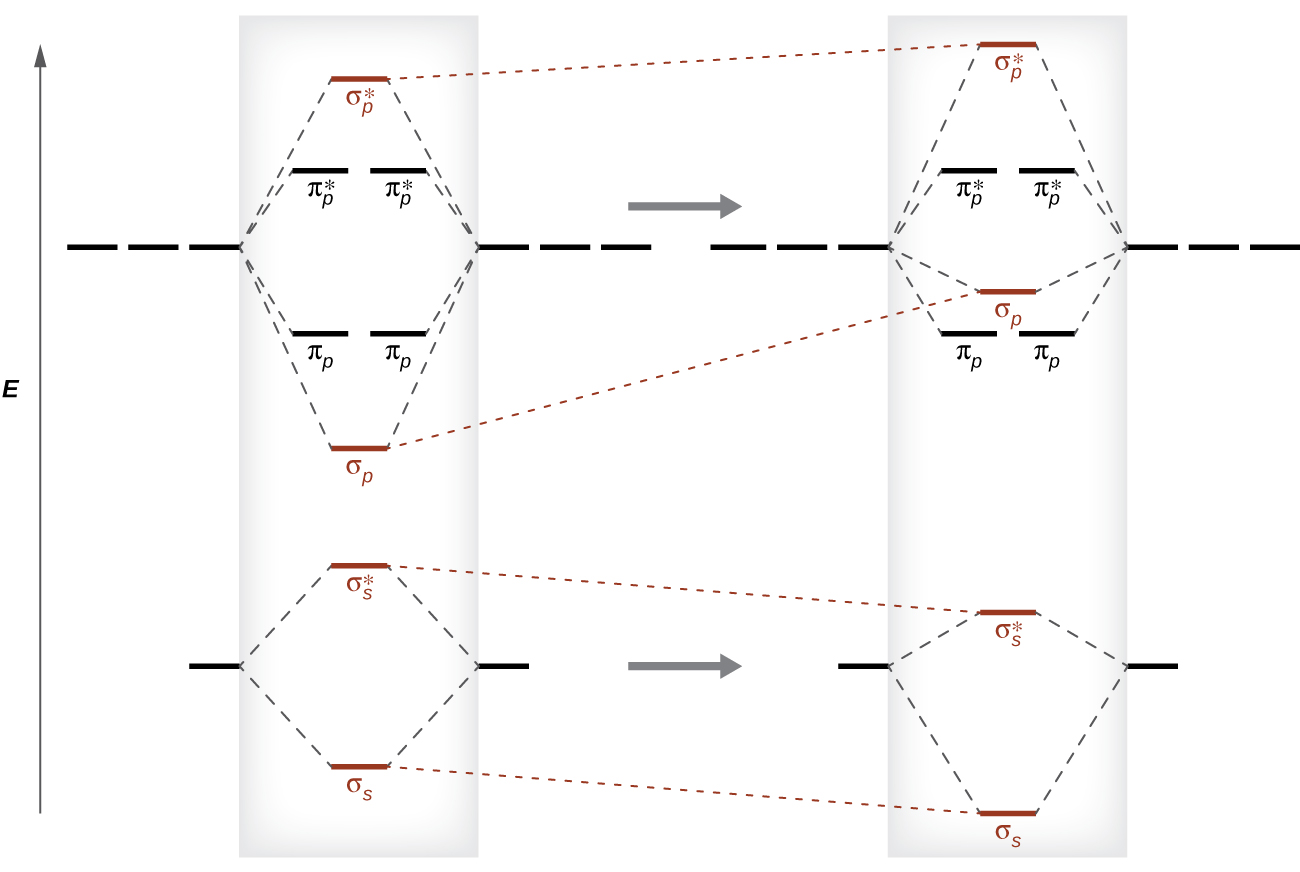
7.7 Molecular Orbital Theory - Chemistry Fundamentals The relative energy levels of atomic and molecular orbitals are typically shown in a molecular orbital diagram (Figure 7.7.9). For a diatomic molecule, the atomic orbitals of one atom are shown on the left, and those of the other atom are shown on the right. Each horizontal line represents one orbital that can hold two electrons.
Solved 15.) Use the molecular orbital diagram shown to ... 15.) Use the molecular orbital diagram shown to determine which of the following is most stable. Atomic orbitals Molecular orbitals orbitals einding -anti s尋 2p 2p Z t Pzp It 2s 1レ 2s B2, C2, N2 b. N22 2- d. C2 e. Question: 15.) Use the molecular orbital diagram shown to determine which of the following is most stable.
Chem Flashcards - Quizlet Use molecular orbital diagram shown to determine which is most stable a) O22-b)F2 c) F22+ d) F22-e) Ne22-a. Use the molecular orbital diagram shown to determine which of the following is most stable. A) F2 B) F22⁺ ...
Use the molecular orbital diagram shown to determine which ... 56) Use the molecular orbital diagram shown to determine which of the following is most stable. A) C 2 2 ⁺ B) N 2 2 ⁺ C) B 2 D) C 2 2 ⁻ E) B 2 2 ⁺ Answer: D. The species C 22- is the most stable. The electronic configuration of C 22- is KK ( σ 2s) 2 ( σ∗ 2s) 2 ( π 2px) 2 ( π∗ 2py) 2 ( σ 2pz) 2.
C22- Molecular Orbital Diagram - schematron.org It is stable. In fact, it's the perioxide ion. Check me out. The problem provides you with the MO diagram for the C2 molecule, so all you really have to do here is add an electron to that diagram. This is the molecular orbital diagram for the homonuclear diatomic Be2+, showing the molecular orbitals of the valence shell only. The molecular ...
Use the molecular orbital diagram shown to determine which ... Oct 02, 2021 · Use the molecular orbital diagram shown to determine which of the following is most stable. a. f22+ b. ne22+ c. f22- d. o22+ e. f2
Mock Final - GitHub Pages 31) Use the molecular orbital diagram shown to determine which of the following is most stable. A) C2^2⁺ B) N2^2⁺ C) B2; D) C2^2⁻ E) B2^2⁺ 32) How many moles of oxygen are formed when 58.6 g of KNO3 decomposes according to the following reaction? The molar mass of KNO3 is 101.11 g/mol. 4 KNO3(s) → 2 K2O(s) + 2 N2(g) + 5 O2(g)
Quiz 1 Flashcards | Quizlet Draw the molecular orbital diagram shown to determine which of the following is MOST stable A) B2^2+ B) C2^2+ C) N2^2+ D) C2^2-E) B2. D. Identify the number of electron groups around a molecule with so hybridization. 2. Identify the number of electron groups around a molecule with sp^2 hybridization. 3.
Use the molecular orbital diagram shown to determine ... Problem: Use the molecular orbital diagram shown to determine which of the following is most stable.A) N22+ B) B2C) B22+D) C22-E) C22+ FREE Expert Solution Show answer Answer:
chemistry 1a Chapter 10 Flashcards | Quizlet Use the molecular orbital diagram shown to determine which of the following is most stable A) Ne2^2⁺ B) F2^2⁺ C) F2^2⁻ D) F2 E) O2^2⁺ E). O2^2⁺ Use the molecular orbital diagram shown to determine which of the following are paramagnetic. A) O2 2⁺ ...


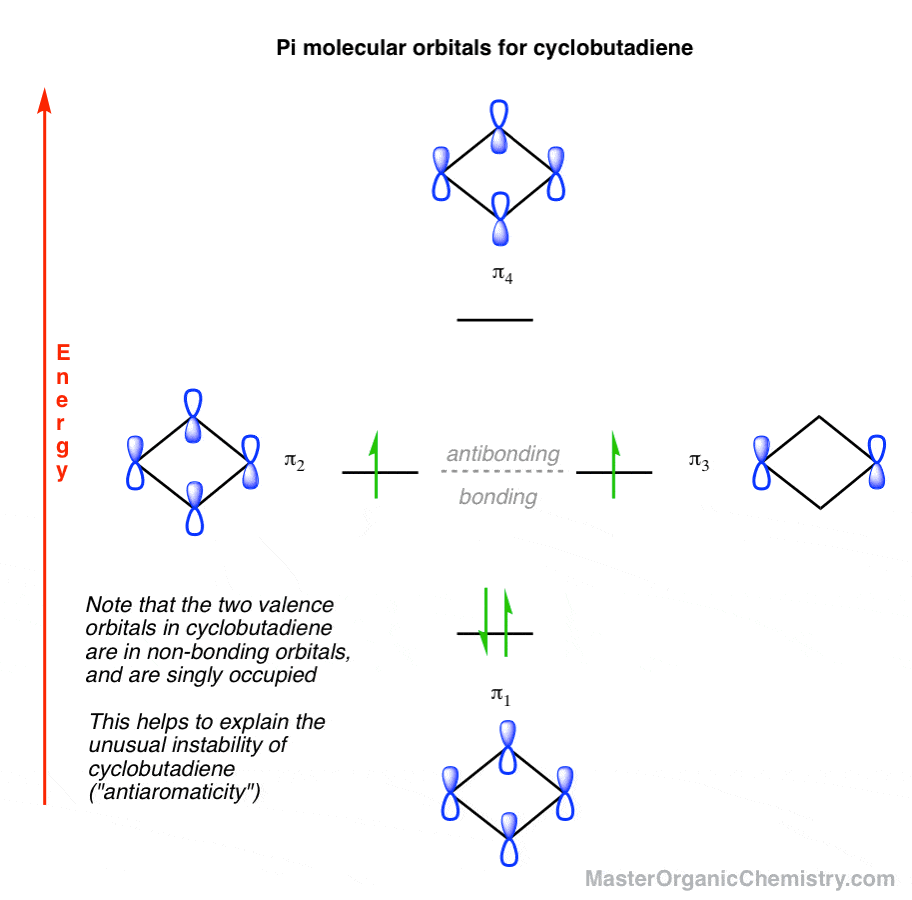
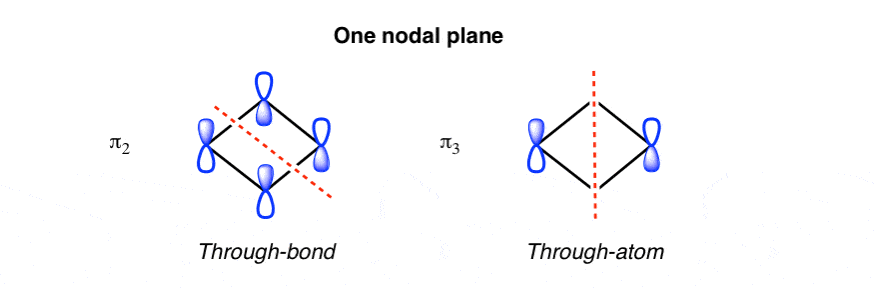




![Expert Answer] Draw the molecular orbital diagram for F2 and ...](https://hi-static.z-dn.net/files/d7a/d377148aa5469a29270f608aa4d77ef6.png)
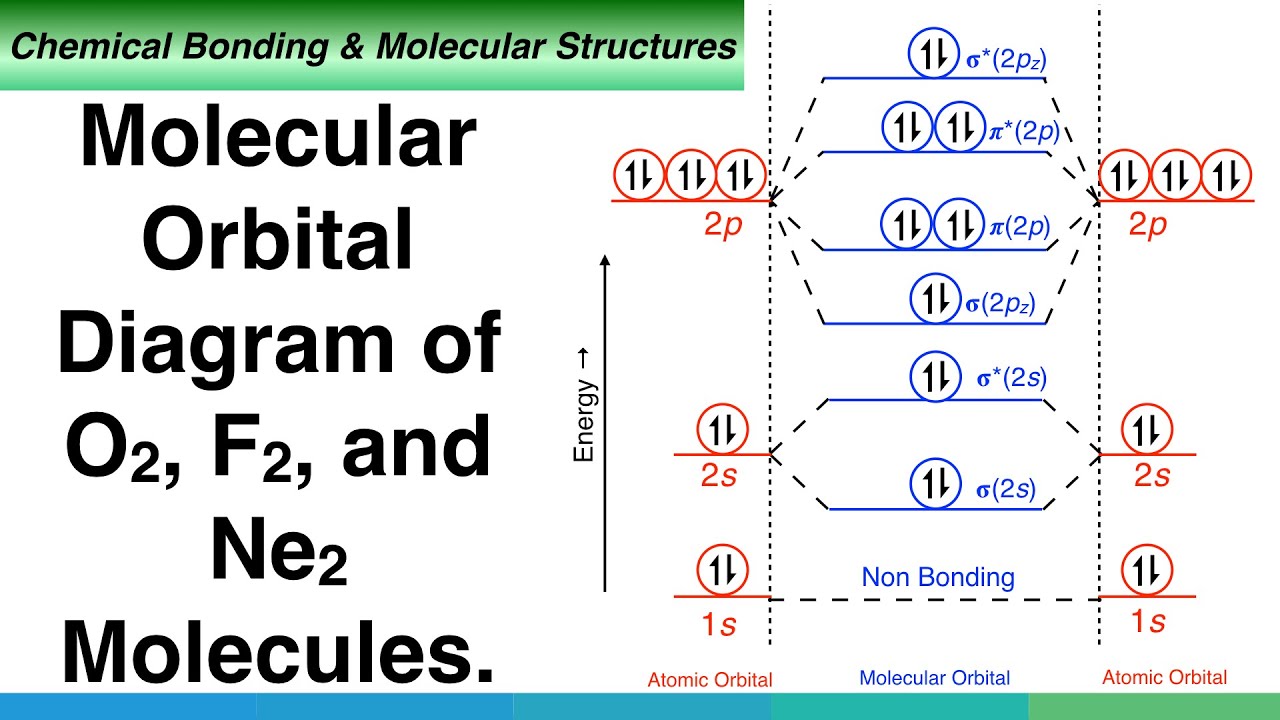
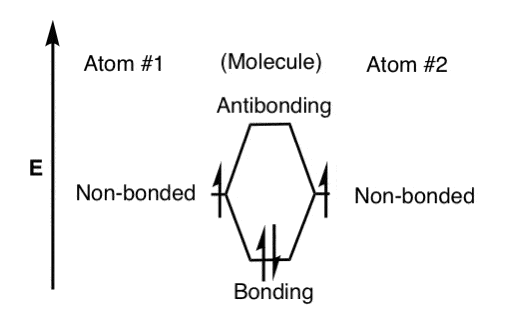




0 Response to "37 use the molecular orbital diagram shown to determine which of the following is most stable"
Post a Comment