39 diagram of exothermic reaction
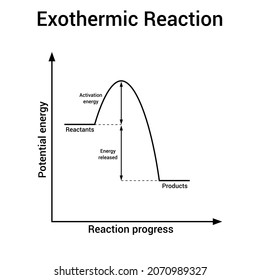
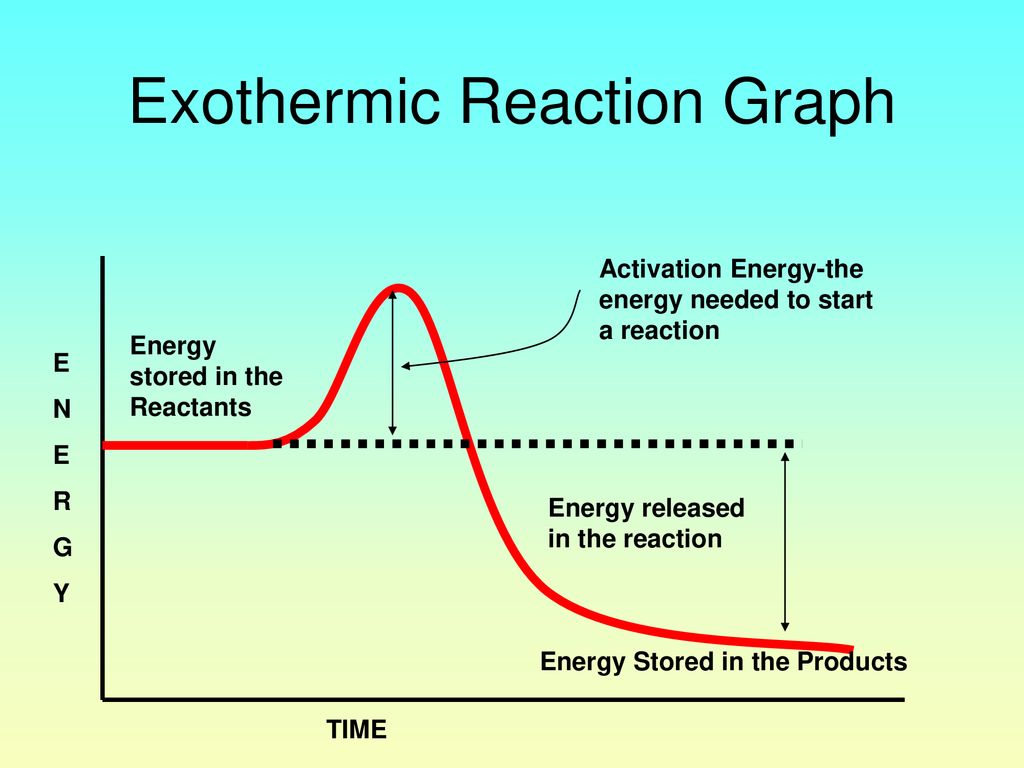
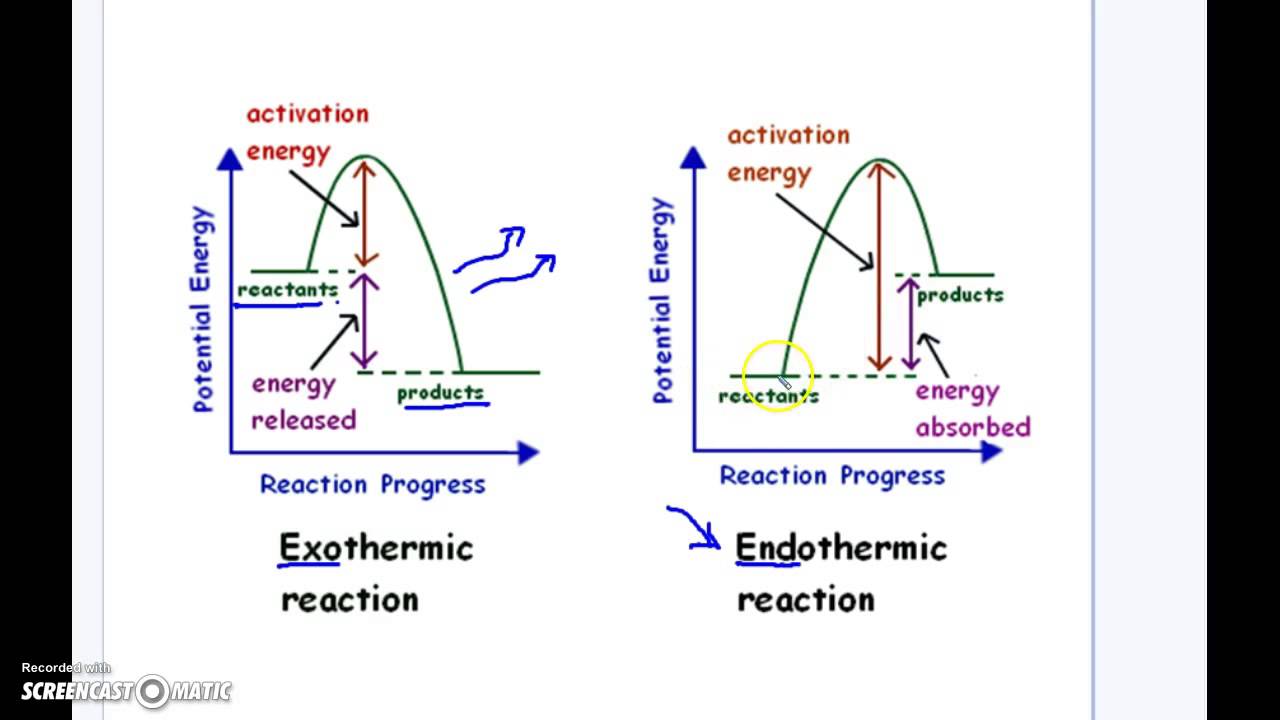
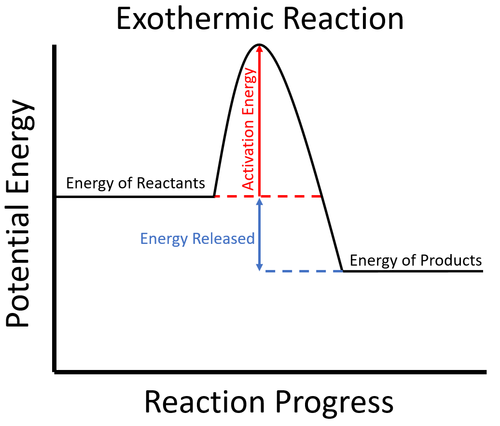
Reaction profiles - Exothermic and endothermic reactions ... An energy level diagram shows whether a reaction is exothermic or endothermic. It shows the energy in the reactants and products, and the difference in energy between them. Exothermic reaction The... What are Exothermic Reactions? (with Examples and Video) An Exothermic reaction is a chemical reaction that involves the release of energy in the form of heat or light. These reactions are the opposite of endothermic reactions and can be expressed in a chemical equation as follows: Reactants → Products + Energy What is an Exothermic Reaction?
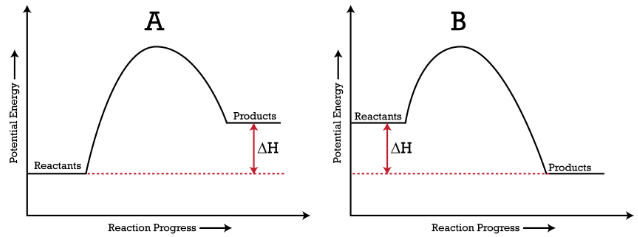
Energy level diagrams - Why are there energy changes in ... The reaction shown by the second diagram is more exothermic. There is a greater difference in energy between the reactants and products. The green arrow is longer.

Diagram of exothermic reaction
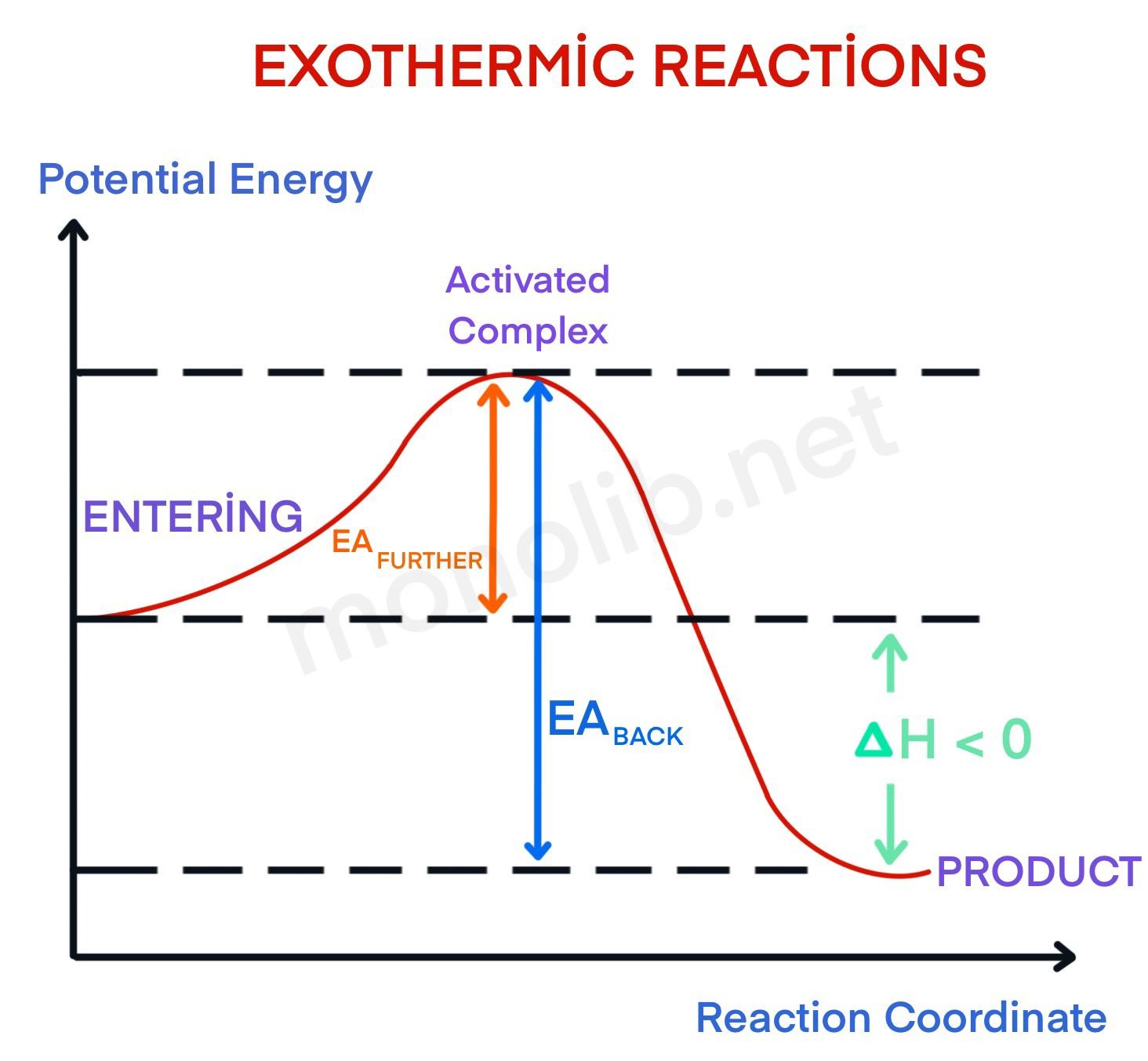
7.01_chemistry.docx - Endothermic and Exothermic Activity ... Endothermic and Exothermic Activity For this assignment, you will create your own potential energy diagrams for each of the three chemical reactions. Then you will analyze the data and your diagrams for each reaction. Generic Reactions Reactants Products Transition State Synthesis A + B → AB A + B −15 kJ AB 20 kJ 30 kJ Single Replacement C + AB → CB + A C + AB 65 kJ CB + A 30 kJ 85 kJ ... PDF Exhothermic and Endothermic Reactions / Activation Energy ... • In the graphs that follow, I have drawn diagrams for both Exhothermic and Endothermic reactions showing their uncatalysed Activation Energy - and I have also shown the NEW Activation Energy that would pertain to the situation if a catalyst were introduced to the reaction. Potential Energy Diagrams - Chemistry - Catalyst ... This chemistry video tutorial focuses on potential energy diagrams for endothermic and exothermic reactions. It also shows the effect of a catalyst on the f...
Diagram of exothermic reaction. Difference between Exothermic and Endothermic Reactions ... A chemical reaction that involves the release of energy in the form of heat or light is known as an exothermic reaction. When carbon burns in oxygen to make carbon dioxide, for example, a large amount of heat is produced. C + O2 → CO2 (Carbon) (Oxygen) (Carbon dioxide) Endothermic Reaction Coordinate Diagram - schematron.org A typical reaction coordinate diagram for a mechanism with a single step is shown below: Below is a reaction coordinate diagram for an endothermic reaction. The fully filled in reaction coordinate diagram is displayed below. This reaction is also exothermic because the energy of the products is lower than that of the. Chemistry H, Venn Diagram {Exothermic vs. Endothermic ... Start studying Chemistry H, Venn Diagram {Exothermic vs. Endothermic} :). Learn vocabulary, terms, and more with flashcards, games, and other study tools. PDF Exothermic vs endothermic reaction graphs Given the reaction: A + B --> Ca) Does the diagram illustrate an exothermic or an endothermic reaction? State one reason, in terms of energy, to support your answer.Answer-->Endothermic, the products have more energy than the reactants.b) On the diagram provided in your answer booklet, draw a dashed line to indicate a potential energy curve for ...
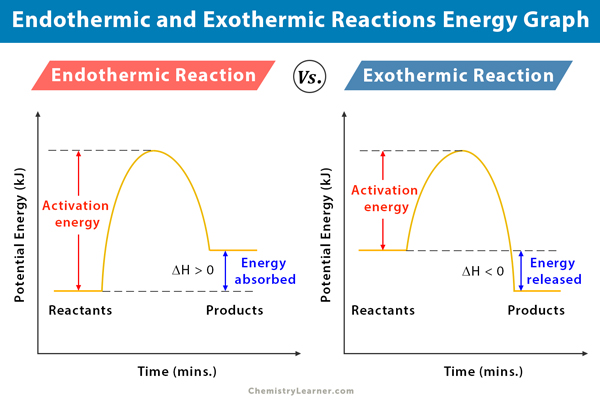
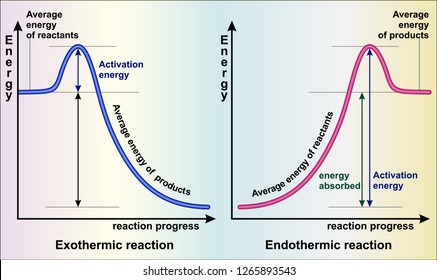
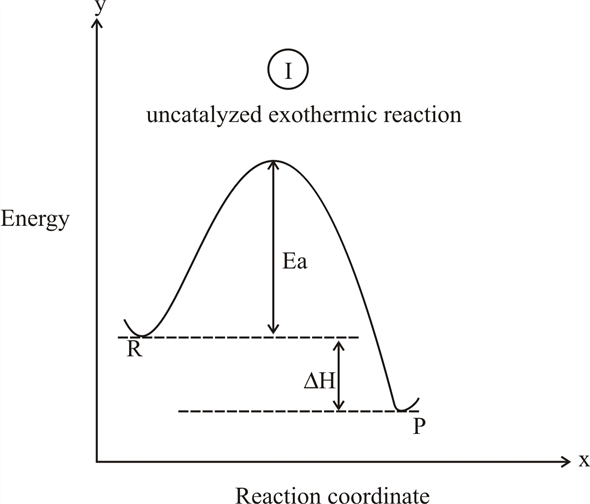
PDF Topic 5.1 Exothermic and Endothermic Reactions Heat and ... a) Draw a diagram of the energy profile for this reaction. Label the diagram. b) State whether the reaction is endothermic or exothermic. c) Calculate the energy difference between the reactants and the products. d) Deduce the sign of the enthalpy change. e) Identify with a reason, which is more stable, the reactants of products. 8. (N04/S/2) Exothermic Reaction - Definition and Examples | Properties ... Above is an energy level diagram that describes an exothermic reaction. The difference in intrinsic energy of the products and the reactants showcase how much energy is given out to the surroundings. Examples of Exothermic Reactions Following is a list of examples of endothermic reactions that are found in day to day life: Energy Diagrams of Reactions | Fiveable Energy Diagrams. Exothermic Reactions. Endothermic Reactions. Example. 6.3 Kinetic Energy, Heat Transfer, and Thermal Equilibrium. 6.4 Heat Capacity and Coffee-Cup Calorimetry. 6.5 Phase Changes and Energy. 6.6 Introduction to Enthalpy of Reaction. 6.7 Bond Enthalpy and Bond Dissociation Energy. Exothermic And Endothermic Reaction | Kofa Study (b) Neutralization reaction (c) Displacement reaction (d) Addition of concentrated acid to water (e) All combustion reactions (f) Passing SO 2 gas into water In the energy profile diagram of an exothermic reaction, note that the product is formed at lower energy than the reactant. ∆H = negative, since Hr ≥ Hpr.
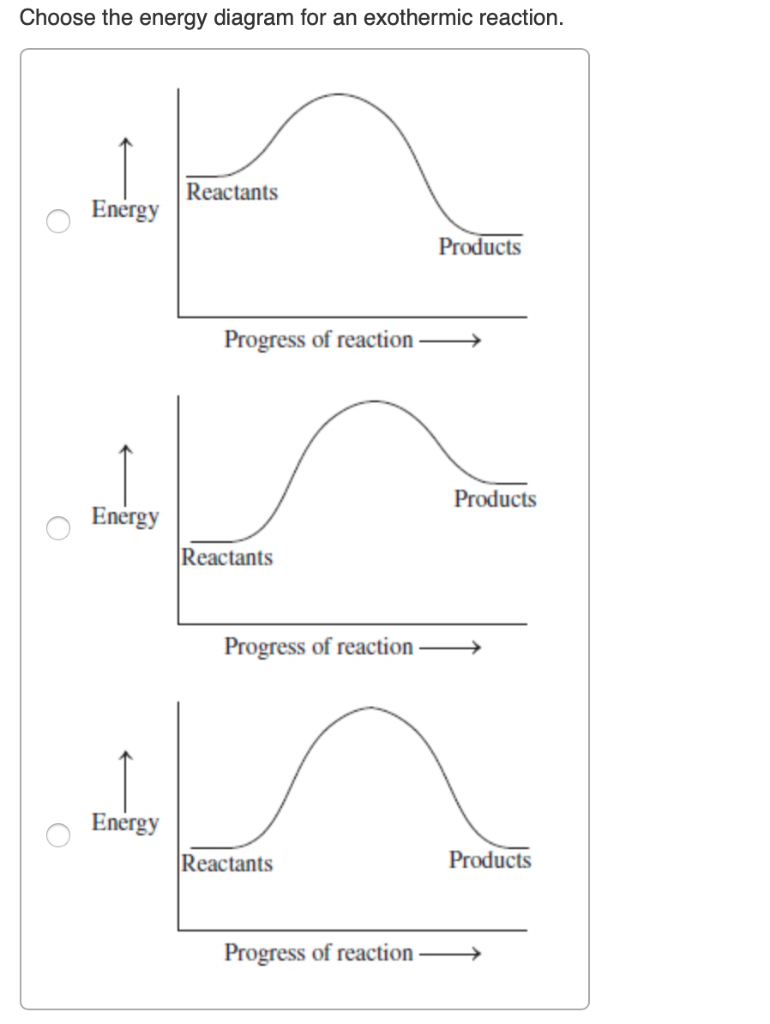
Endothermic vs. exothermic reactions (article) | Khan Academy An exothermic process releases heat, causing the temperature of the immediate surroundings to rise. An endothermic process absorbs heat and cools the surroundings.". Based on the above definition, let's pick a few examples from our daily lives and categorize them as endothermic or exothermic. Exothermic and Endothermic Reactions Venn Diagram ... The exothermic and endothermic reactions Venn diagram graphic organiser is a great way for students to compare and contrast these two types of chemical reactions. Includes two versions: Version 1. Students cut and paste labels onto the correct part of the Venn diagram. Version 2. Students write the correct statement into boxes on the Venn diagram. How do you know if a diagram is endothermic or exothermic ... The diagram shows a reaction profile for an exothermic reaction. What are 3 differences between an endothermic and exothermic reaction? Similar is the case with the endothermic and exothermic reactions in Chemistry. These release energy in the form of sound, light, cold or heat….Exothermic Reactions. Which Potential Energy Diagram Represents An Exothermic ... Which Potential Energy Diagram Represents An Exothermic Reaction? Potential Energy Potential Energy Non Reaction Coordinate A) Reaction Coordinate B) Reaction Coordinate C) Reaction Coordinate D) Select An Answer And Submit. For Keyboard Navigation, Use The Up/Down Arrow Keys To Select An Answer. A...
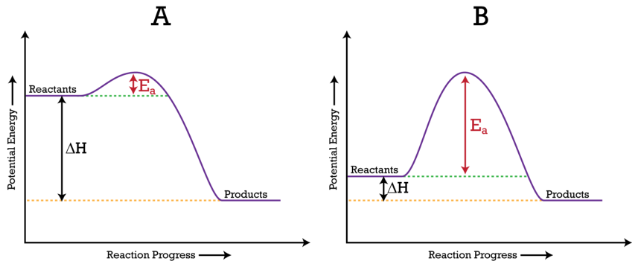
Reaction Coordinate Diagrams The diagram below is called a reaction coordinate diagram. It shows how the energy of the system changes during a chemical reaction. In this example, B is at a lower total energy than A. This is an exothermic reaction(heat is given off) and should be favorable from an energy standpoint. The energy difference between A and B is E in the diagram.
Answered: In an energy diagram for an exothermic… | bartleby In an energy diagram for an exothermic chemical reaction which of the following is true? The energy of the products is higher than that of the reactants. The energy of the transition state is lower than that of the reactants. The energy of the products is lower than that of the reactants. an exothermic reaction requires energy to proceed.
Endothermic and Exothermic Reactions Diagram | Quizlet Diagram of endothermic and exothermic reactions. Terms in this set (5) Exothermic Reaction. In this type of reaction, energy (in the form of heat, sound or light) is released when the reactants break apart. Heat energy can be picked up by the area surrounding the products. ... In exothermic reactions, there is more energy in the reactants than ...
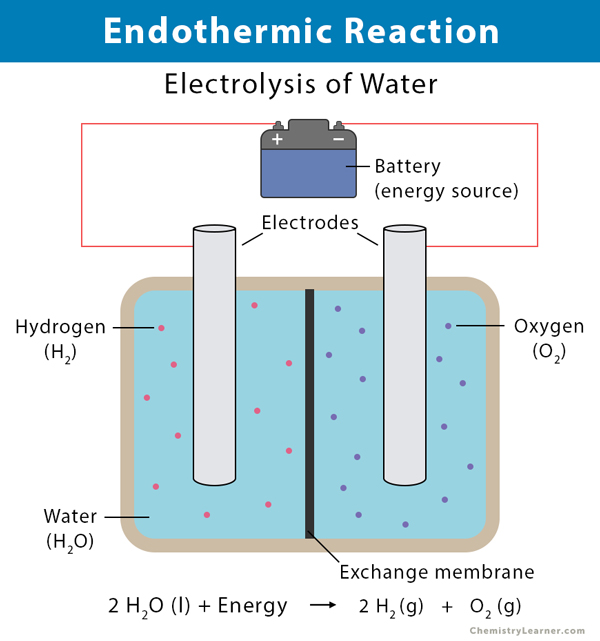
Exothermic and Endothermic Processes | Introduction to ... Exothermic reaction In an exothermic reaction, the total energy of the products is less than the total energy of the reactants. Therefore, the change in enthalpy is negative, and heat is released to the surroundings. Endothermic Reactions. Endothermic reactions are reactions that require external energy, usually in the form of heat, for the ...
PDF Endothermic and Exothermic reaction Worksheet Name Date Block Is the reaction exothermic or endothermic? Use ideas about bonds to explain why. _exo because two bonds are broken from 4 bonds b) Draw diagrams to show what happens when hydrogen reacts with oxygen. Mark the bonds broken in blue and the new bonds formed in red. The equation is: 2H 2 bonds hydrogen Blue + O 2 oxygen 2H 2 Ored side water
Creative Diagram Of Exothermic Reaction - Glaucoma Template The reaction diagrams of exothermic reactions and endothermic reactions are as follows. Endothermic and exothermic reactions can be visually represented by energy-level diagrams like the ones in Figure PageIndex2. As we can see and examine from the graph in an exothermic reaction the reactants are usually at a higher.
How does the energy level diagram show this reaction is ... Figure shows the energy level diagram for the reaction between methane and oxygen. Based on Figure, the following information can be obtained. (a) The reaction between methane and oxygen to form carbon dioxide and water is an exothermic reaction. (b) During the reaction, the temperature of the mixture increases.
Exothermic and Endothermic Reactions - Energy Level Diagram Exothermic and Endothermic Reactions - Energy Level DiagramForm 5 Chemistry Chapter 4 ThermochemistryThis video is created by ...
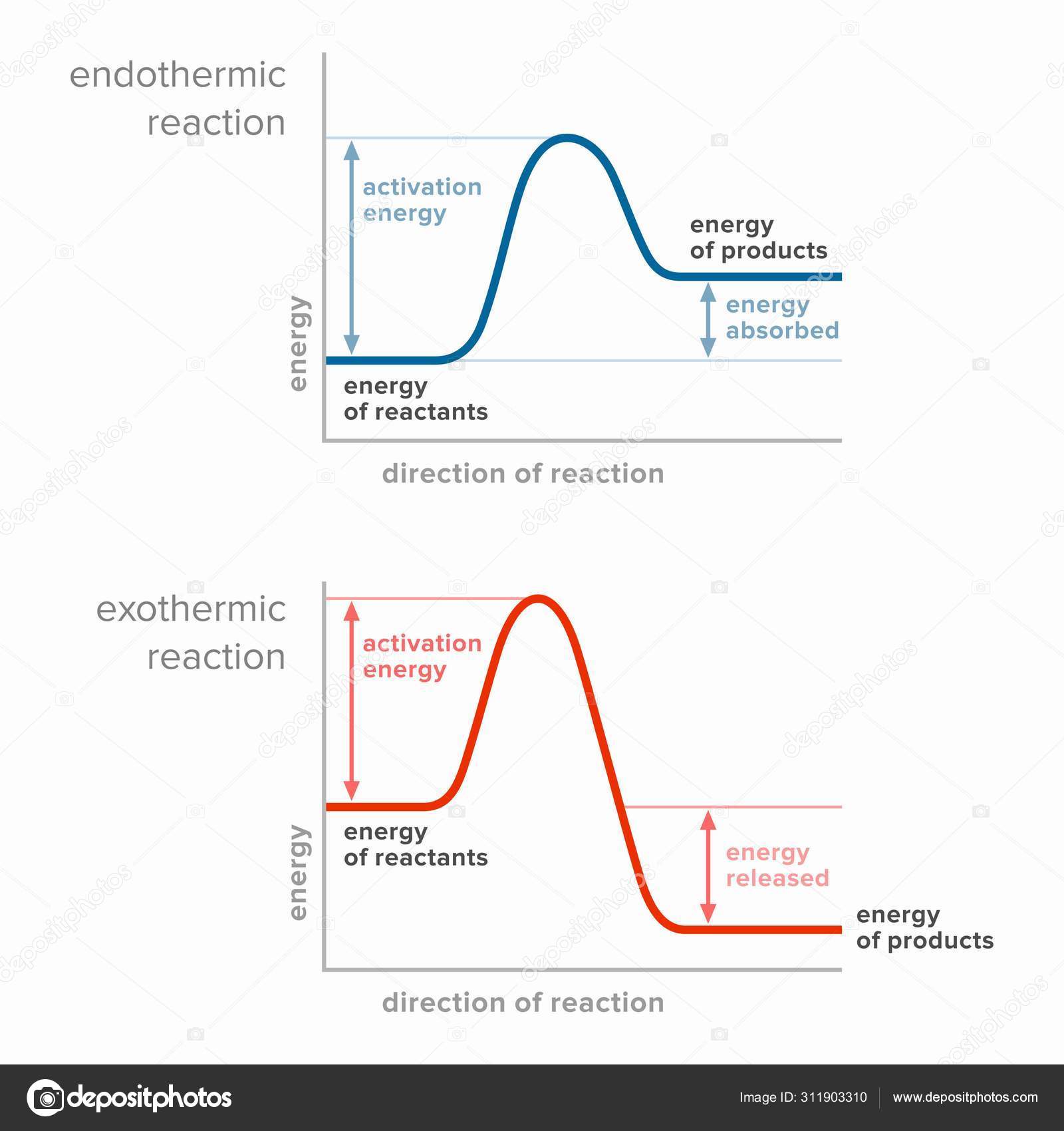
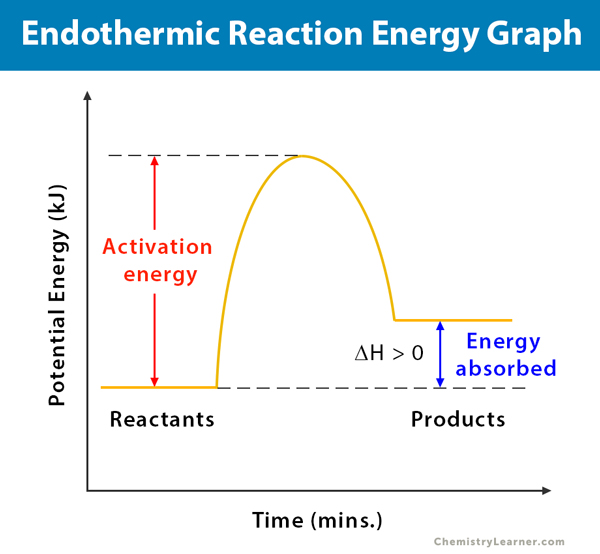
Endothermic Reactions: Definition, Example, Diagram and ... Endothermic Reaction Energy Level Diagram: Endothermic reactions are depicted in a basic energy level diagram below. The activation energy is the amount of energy that must be delivered to the reactants for them to break through the energy barrier and react. In an endothermic reaction, the result has higher potential energy than the reactants.
Exothermic Reactions: An Overview, Formula, Examples ... Exothermic reactions are opposed to endothermic reactions which involve the absorption of energy from the surrounding in the form of heat. Exothermic chemical reactions transfer the energy in the surrounding area. In the presence of water, a strong acid will dissociate quickly and release heat, therefore it is an exothermic reaction.
Consider the Reaction Energy diagram below: Group of ... Chemistry questions and answers. Consider the Reaction Energy diagram below: Group of answer choices The reaction is exothermic. The reaction can not occur as written. The reaction is endothermic. The reaction produces energy. Question: Consider the Reaction Energy diagram below: Group of answer choices The reaction is exothermic.
Potential Energy Diagrams - Chemistry - Catalyst ... This chemistry video tutorial focuses on potential energy diagrams for endothermic and exothermic reactions. It also shows the effect of a catalyst on the f...
PDF Exhothermic and Endothermic Reactions / Activation Energy ... • In the graphs that follow, I have drawn diagrams for both Exhothermic and Endothermic reactions showing their uncatalysed Activation Energy - and I have also shown the NEW Activation Energy that would pertain to the situation if a catalyst were introduced to the reaction.
7.01_chemistry.docx - Endothermic and Exothermic Activity ... Endothermic and Exothermic Activity For this assignment, you will create your own potential energy diagrams for each of the three chemical reactions. Then you will analyze the data and your diagrams for each reaction. Generic Reactions Reactants Products Transition State Synthesis A + B → AB A + B −15 kJ AB 20 kJ 30 kJ Single Replacement C + AB → CB + A C + AB 65 kJ CB + A 30 kJ 85 kJ ...













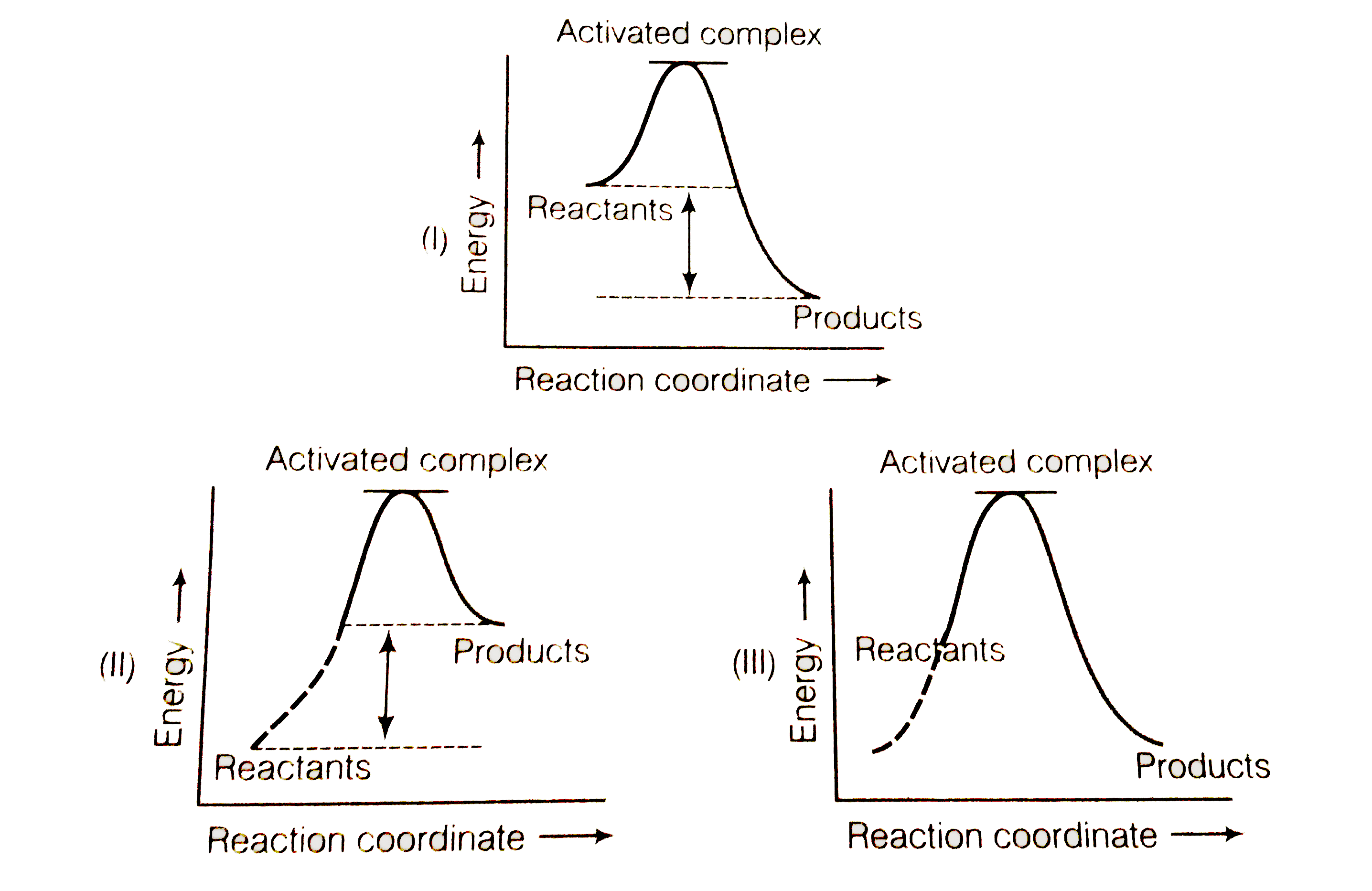

0 Response to "39 diagram of exothermic reaction"
Post a Comment