36 bohr diagram for sulfur
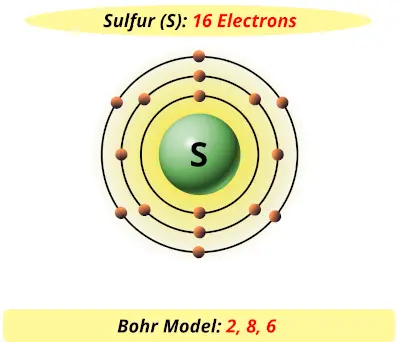
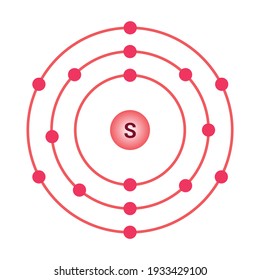
Sulfur / Sulphur has 2 electrons in its first shell, 8 in its second, 6 in its third.Check me out: http://www.chemistnate.com Jul 04, 2019 · Sulfur – 4. Fluorine – 5. Calcium – 6. Argon – Name ANSWER KEY Period Date Bohr Model Diagrams. Use the information provided for each element to draw Bohr Model diagrams. Label how many of each there are in the nucleus (e.g. He: 2p, 2n). They should show all the electrons, 16 electrons in total. There are three shells containing the electrons.
11. Draw Bohr diagrams for sulfur and chlorine. Draw a Bohr diagram and use it to explain your answer. 2. Using the concepts of shielding and attraction, explain why sulfur is smaller in radius than silicon.

Bohr diagram for sulfur
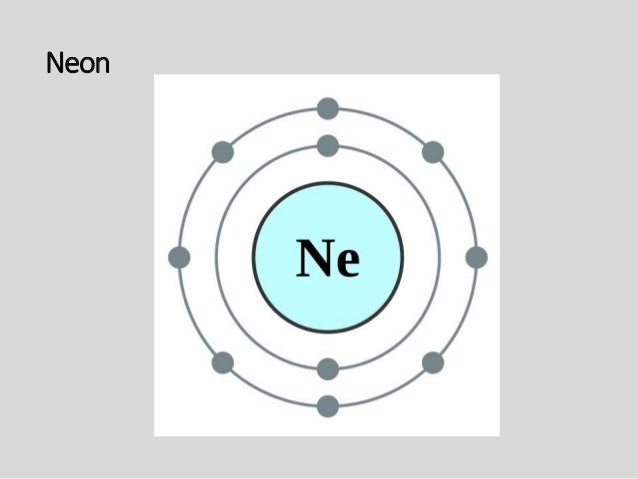
Bohr Diagram Questions 1. The sentences below contain errors or are incomplete. Write complete, correct versions. a) Negative particles called neutrons circle the 3. For the metallic elements sodium, magnesium, and aluminum, answer the following questions: a) Draw a Bohr diagram for each element. Bohr model of Nitrogen (N) 2, 5: 8: Bohr model of Oxygen (O) 2, 6: 9: Bohr model of Fluorine (F) 2, 7: 10: Bohr model of Neon (Ne) 2, 8: 11: Bohr model of Sodium (Na) 2, 8, 1: 12: Bohr model of Magnesium (Mg) 2, 8, 2: 13: Bohr model of Aluminum (Al) 2, 8, 3: 14: Bohr model of Silicon (Si) 2, 8, 4: 15: Bohr model of Phosphorus (P) 2, 8, 5: 16 ... In order to make a Bohr diagram, you need to know the number of protons, neutrons, and electrons the element has. The Bohr model was an advancement to an earlier atomic model, the Rutherford Model. Key features of the Bohr model include set orbits for electrons around the nucleus, as well as...
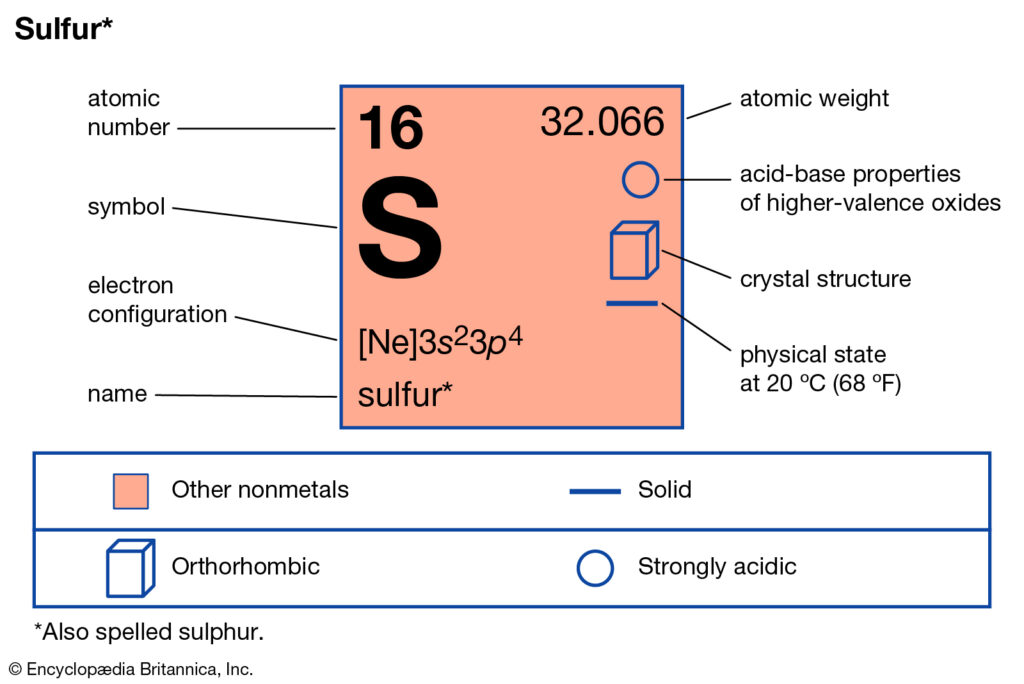
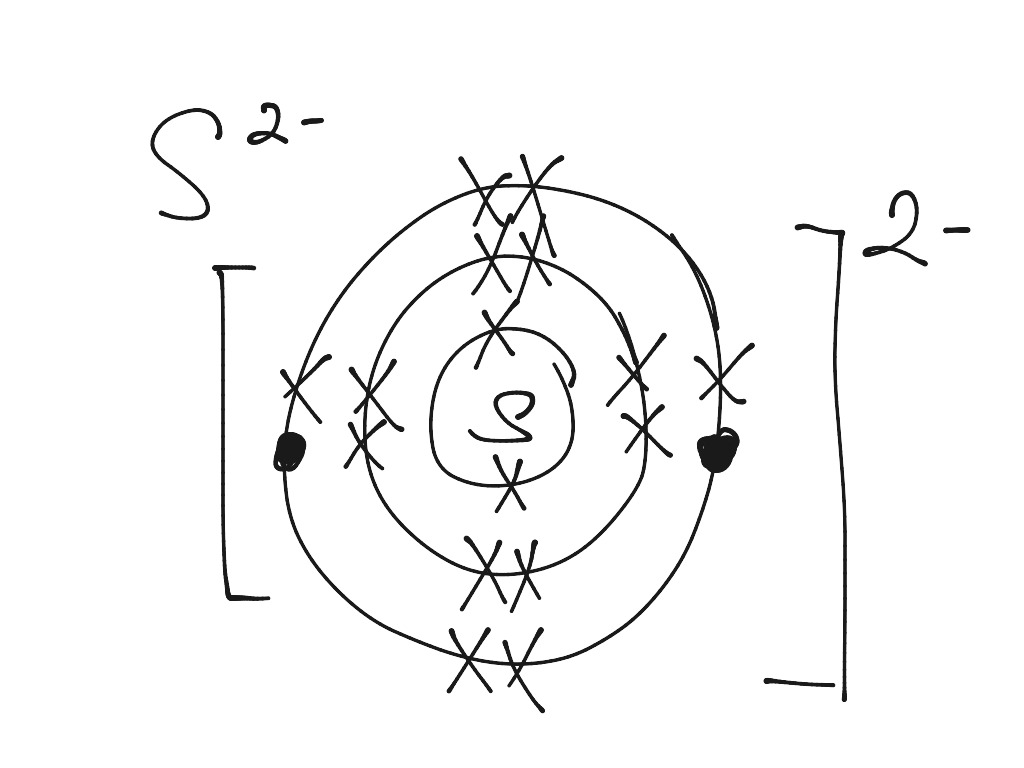
Bohr diagram for sulfur. The bohr diagram is the diagram of the electrons on the orbital layers of the nucleus of an atom. for potassium, you would put 2 electrons on the first layer, 8 on the second layer, and 9 on the third layer. This is because the atomic number of Potassium (K) is 19, therefore has 19 protons and 19 electrons. How to Make a Model of a Sulfur Atom. Just Now Sulfur Bohr Model Diagram. Sulfur at Chemical schematron.org Basic Information Atomic Basic Information. 1 hours ago Bohr Diagram Sulfur Shows all electrons Atomic # 16 Atomic Mass — -32 Protons = I (O Neutrons Electron = 15. What is a Bohr model for sulfur? The sulfur atom has 16 protons, 16 neutrons and 16 electrons in three different energy levels, or orbits. Physics suggests that ... Sulfur has 16 electrons, 16 protons and considering the 3216S 16 32 S isotope, 16 neutrons. Shell n=1 has 2 electrons and shells n=2 has 8...
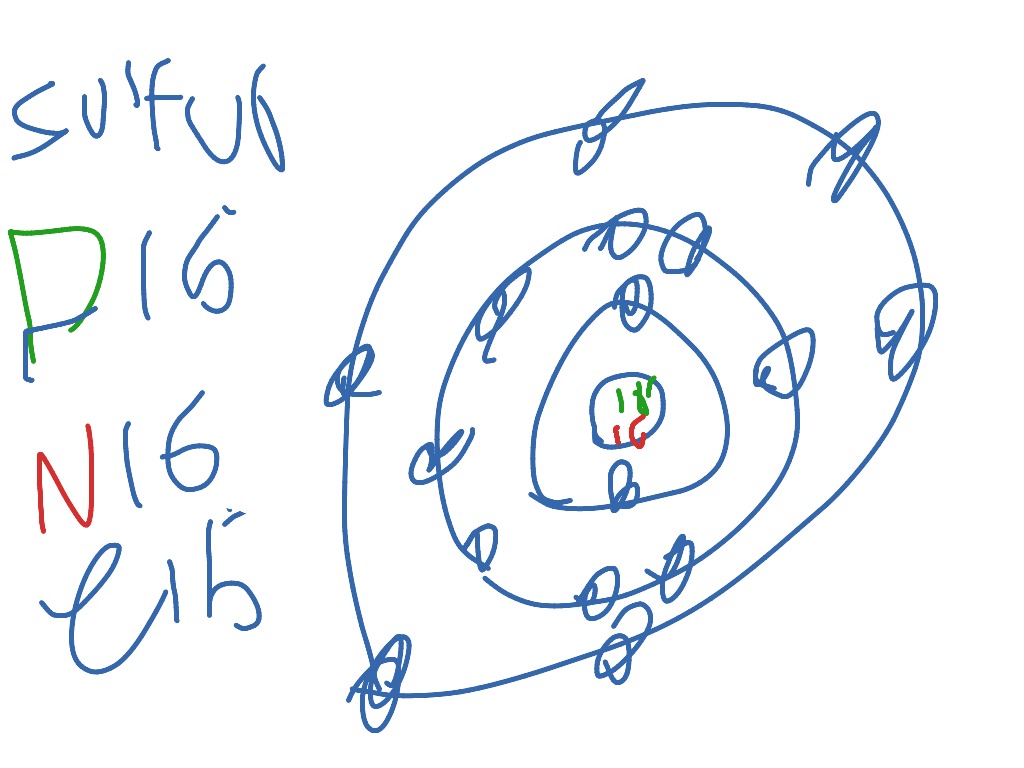
So the fourth energy level needs to have electrons? I noticed I was missing two since I only added 14 to my diagram, but I just put them on the third energy But there's 16 electrons in sulfur. Since it's an atom, the proton and the electron are the same. I am so confused, I'll message my teacher and wait... 14. Scientists use two types of diagrams to show the electron configuration for atoms. Follow your teacher's directions to complete the diagrams. Sulfur Atomic # = 16. Bohr Diagram Shows all electrons. Lewis Structure Shows valence electrons. How to Draw the Bohr-Rutherford Diagram for Boron. A Bohr diagram shows the distribution of an atom's electrons among different energy levels, or electron shells. Each diagram also features the For example, a Bohr diagram of the element boron shows five protons and five electrons. A Bohr diagram starts with a simple circle to represent the...
Outline. IUPAC Bohr diagrams for atoms types of chemical bonds molecular compounds types simple covalent common name ionic compounds simple ionic multivalent polyatomic/complex hydrated ionic acids. Bohr diagrams for a variety of elements are illustrated in chart below . When determining the number of electrons of an atom, remember to refer to the atomic number (the whole Bohr model of sulfur: (CC BY-SA 2.0 uk; Greg Robson). Valence electrons are located in the highest energy level of an atom. Bohr Diagram - The Element Sulfur. The Element Sulfur. All About Sulfur! Bohr Diagram. Bohr model diagram of sulfur S in atomic physics.
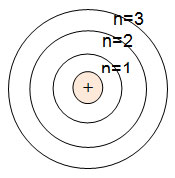
In atomic physics, the Bohr model or Rutherford-Bohr model, presented by Niels Bohr and Ernest Rutherford in 1913, is a system consisting of a small, dense nucleus surrounded by orbiting electrons—similar to the structure of the Solar System...
Electrons are always moving around the nucleus and so possess potential and kinetic energy. But they can only possess certain values of energy, or specific energy levels. (Credit should be given to Niels Bohr for proposing this theory.)
All About Sulfur! Bohr Diagram; All About Sulfur! When: Around 1777 Who Discovered it: Antoine Lavoisier Atomic Number: 16 Atomic Mass: 32.065 Physical properties: Density: 2.067 grams per cubic centimeter, Sulfur is odorless, yellow, brittle, and solid
Number of Protons/Electrons: 16. Number of Neutrons: 16 ... [Bohr Model of Sulfur] ... Name Origin: From the Latin word sulfur (brimstone)
Bohr diagram is very interesting and easy to draw. Here, we will draw the Bohr diagram of the Sulfur atom with some simple steps. The number of neutrons for the Bohr diagram of Sulfur can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole).
Get the detailed answer: sulfur bohr diagram.
12. Bohr Diagrams Find your element on the periodic table. Determine the number of electrons - it is the same as the atomic number. This is how many electrons you will draw. Draw a Bohr diagram for sulfur.
Created with TouchCast https://itunes.apple.com/us/app/touchcast/id603258418For the interactive version visit:http://touchcast.com/msperrotti/bohr_model_sulfur
A Bohr diagram is a simplified visual representation of an atom that was developed by Danish physicist Niels Bohr in 1913. Bohr diagrams are used to introduce students to quantum mechanics because of their simplicity, and are a good way to show students how electrons are organized into discrete...
1501709 3d models found related to bohr diagram for sulfur. Diagram of connection of the capacitive sensor, for the autolevel function, with an relay module. I drew this diagram, because an Internet friend, has this module, common in Arduino projects or as controller of a 220V silicone hotbed.
What Is The Bohr Diagram For Sulfur Study Com. Bohr Diagram Of Aluminium 6 Stromoeko De. How To Draw Bohr Diagrams Slideshare. Bohr Diagram For Sulfur Sodium Diagram Octet Great Installation Of.
Diagram Strontium Bohr Diagram Full Version Hd Quality Bohr. Bohr Diagram For Cl Awesome 27 Inspirational Bohr Diagram Sulfur. Bohr Diagram For Sodium And Chlorine. Chemical Elements Com Sulfur S. 3 9 Atomic Models Of The Twentieth Century Chemistry Libretexts.
Do Now: Draw a Bohr diagram for sulfur Do Now: Draw a Bohr diagram for sulfur. They Bond Beryllium = Group 2 = 2 valance electrons Sulfur = Group 16 = 6 valance electrons. 12 Chemical Bonds Atoms with unfilled valence shells are considered unstable.
Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. In the Bohr model, electrons are. Sulfur (S). Diagram of the nuclear composition, electron configuration, chemical data, and valence orbitals of an atom of sulfur (atomic number: 16), the most. In the Bohr model, electrons are confined to concentric spheres around the nucleus numbered as n=1, 2, 3,.
Information: Phase diagrams. Figure 1: Phase diagram for water. Note that the unit for pressure on the diagram is atmospheres (atm). Thus, the attraction from the nucleus is +4. 11. Draw Bohr diagrams for sulfur and chlorine. Nucleus Nucleus has a +16 has a +17 charge charge.
Bohr Diagrams 1) Check your work. 2) You should have 6 total electrons for Carbon. 3) Only two electrons can fit in the 1st shell. 4) The 2nd shell can hold up to 8 electrons. 5) The 3rd shell can hold 18, but the elements in the first few periods only use 8 electrons. 6p 6n. Bohr Diagrams Try the following elements one at a time: a) H b) He
Draw a Bohr Diagram for Sulfur. Draw a Bohr Diagram for Neon. List the number of protons, neutrons and electrons in 48 Ca? (use periodic table for protons/electrons, do the math for neutrons).
Example: Draw a Bohr diagram for a Li atom: Example: Draw a Bohr diagram for a Ne atom: Example: Draw a Bohr diagram for a S 7 TABLE A. Writing the Formula from the Name - Molecular Compounds Name Formula Name 1. chlorine monoxide 11. sulfur dioxide 2. oxygen difluoride 12...
Bohr Model Diagrams and Lewis Dot Structures. Use the information provided for each element to draw Bohr Model diagrams. Rather than drawing individual protons and neutrons, you may simply label how many of each there are in the nucleus (e.g. He: 2p, 2n). Then, draw the individual electrons...
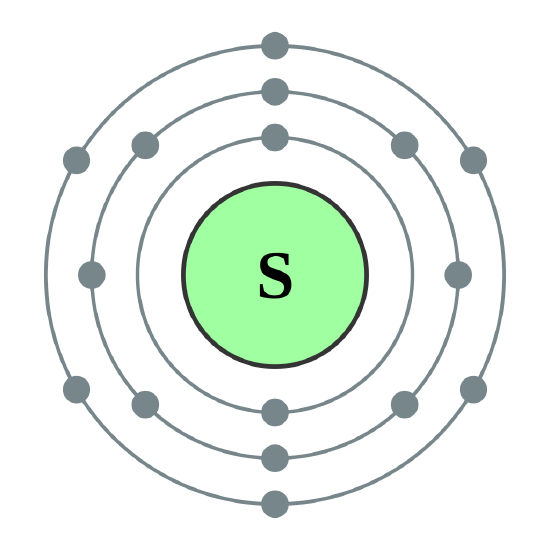
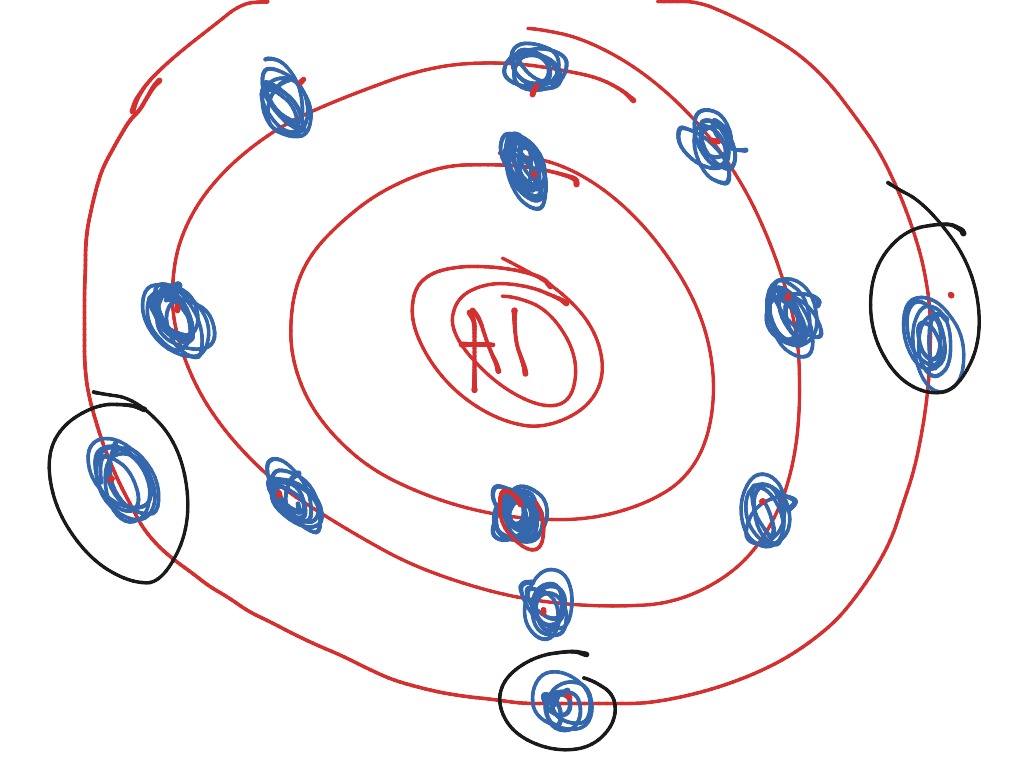
Name: Sulfur Symbol: S [Bohr Model of Sulfur], Number of Energy Levels: 3. In the Bohr model, electrons are confined to concentric spheres around the nucleus numbered as n=1, 2, 3,. The sphere n = 1 can accommodate two, the n = Model sulfur atoms are complex, containing nearly 50 parts. the Bohr atom model with fixed electrons as a way to simplify atomic structure. Sulfur (S).
The element you're looking for is sulfur, or "S". Here's how you can use an element's electron configuration to determine its identity. This means that the element you're looking for will have its atomic number equal to 16. According to the periodic table, this element is sulfur.
Bohr-Rutherford Diagrams. We have looked at atomic models and the structure of atoms. Today we will practice drawing those models for the elements on the periodic table. Using the whiteboards, practice drawing the atomic model (called a Bohr-Rutherford diagram) for the elements listed.
Valence electrons are electrons in the outermost energy level of an atom. More. Presentation on theme: "Q2 : Draw a Bohr model of Sulfur. Draw a ...
15 Aug 2020 — Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. In the Bohr model, electrons are ...
Earn +20 pts. Q: What is the Bohr diagram of sulfur? What does a Bohr model of sulfur look like? The electron configuration of sulfur is: [Ne]3s23p4.
Sulfur Bohr Model - How to draw Bohr diagram for Sulfur (S ... The Bohr model of Sulfur (S) is drawn with three electron shells, the first shell contains 2 electrons, the second shell contains 8 electrons and the third shell contains 6 electrons.
In order to make a Bohr diagram, you need to know the number of protons, neutrons, and electrons the element has. The Bohr model was an advancement to an earlier atomic model, the Rutherford Model. Key features of the Bohr model include set orbits for electrons around the nucleus, as well as...
Bohr model of Nitrogen (N) 2, 5: 8: Bohr model of Oxygen (O) 2, 6: 9: Bohr model of Fluorine (F) 2, 7: 10: Bohr model of Neon (Ne) 2, 8: 11: Bohr model of Sodium (Na) 2, 8, 1: 12: Bohr model of Magnesium (Mg) 2, 8, 2: 13: Bohr model of Aluminum (Al) 2, 8, 3: 14: Bohr model of Silicon (Si) 2, 8, 4: 15: Bohr model of Phosphorus (P) 2, 8, 5: 16 ...
Bohr Diagram Questions 1. The sentences below contain errors or are incomplete. Write complete, correct versions. a) Negative particles called neutrons circle the 3. For the metallic elements sodium, magnesium, and aluminum, answer the following questions: a) Draw a Bohr diagram for each element.
















![How to-bohr-diagram[1]](https://image.slidesharecdn.com/how-to-bohr-diagram1-121113154654-phpapp01/95/how-tobohrdiagram1-14-638.jpg?cb=1352821646)


0 Response to "36 bohr diagram for sulfur"
Post a Comment